Abstract
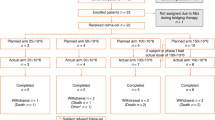
We investigated efficacy and toxicity of lenalidomide in 24 heavily pretreated myeloma patients with a median age of 59 years (range: 37–70) and relapse after allo-SCT. Lenalidomide was given at a dose of 15 mg (n=4), or 25 mg (n=20), orally once daily on day 1 to day 1 every 28 days, with (n=20) or without (n=4) DHAP. The median number of lenalidomide cycles was five (range: 2–17). Major side effects were leukopenia (grade 4: 4%, grade 3: 21% and grade 2: 17%) and thrombocytopenia (grade 3: 17% and grade 2: 29%); infectious complications were observed in 50%. Non-hematological toxicity consisted of muscle cramps (n=9), fatigue (n=5) and constipation (n=2). Mild grade I–II GVHD was seen in three patients. Response was achieved in 66%: CR in 8%, VGPR in 8%, PR in 50% and SD in 13%. The median time to progression was 9.7 months (95% confidence interval (CI): 7.5–11.9), and median OS was 19.9 months (95% CI: 17.3–22.5). Immunomonitoring after lenalidomide showed significant increase of activated NK (NKp44+) and T (HLA-DR+) cells, as well as regulatory T cells (CD4+, CD25+, CD127lo), supporting an immunomodulating anti-myeloma effect of lenalidomide.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout

Similar content being viewed by others
References
Maloney DG, Molina AJ, Sahebi F, Stockerl-Goldstein KE, Sandmaier BM, Bensinger W et al. Allografting with nonmyeloablative conditioning following cytoreductive autografts for the treatment of patients with multiple myeloma. Blood 2003; 102: 3447–3454.
Kröger N, Schwerdtfeger R, Kiehl M, Sayer HG, Renges H, Zabelina T et al. Autologous stem cell transplantation followed by a dose-reduced allograft induces high complete remission rate in multiple myeloma. Blood 2002; 100: 755–760.
Bruno B, Rotta M, Patriarca F, Mordini N, Allione B, Carnevale-Schianca F et al. A comparison of allografting with autografting for newly diagnosed myeloma. N Engl J Med 2007; 356: 1110–1120.
Ayuk F, Shimoni A, Nagler A, Schwerdtfeger R, Kiehl M, Sayer HG et al. Efficacy and toxicity of low-dose escalating donor lymphocyte infusion given after reduced intensity conditioning allograft for multiple myeloma. Leukemia 2004; 18: 659–662.
Kröger N, Krüger W, Renges H, Zabelina T, Stute N, Jung R et al. Donor lymphocyte infusion enhances remission status in patients with persistent disease after allografting for multiple myeloma. Br J Haematol 2001; 112: 421–423.
Kröger N, Zabelina T, Ayuk F, Atanackovic D, Schieder H, Renges H et al. Bortezomib after dose-reduced allogeneic stem cell transplantation for multiple myeloma to enhance or maintain remission status. Exp Hematol 2006; 34: 770–775.
Kröger N, Shimoni A, Zagrivnaja M, Ayuk F, Lioznov M, Schieder H et al. Low-dose thalidomide and donor lymphocyte infusion as adoptive immunotherapy after allogeneic stem cell transplantation in patients with multiple myeloma. Blood 2004; 104: 3361–3363.
van de Donk NW, Kröger N, Hegenbart U, Corradini P, San Miguel JF, Goldschmidt H et al. Remarkable activity of novel agents bortezomib and thalidomide in patients not responding to donor lymphocyte infusions following nonmyeloablative allogeneic stem cell transplantation in multiple myeloma. Blood 2006; 107: 3415–3416.
Mohty M, Attal M, Marit G, Bulabois CE, Garban F, Gratecos N et al. Thalidomide salvage therapy following allogeneic stem cell transplantation for multiple myeloma: a retrospective study from the Intergroupe Francophone du Myélome (IFM) and the Société Française de Greffe de Moelle et Thérapie Cellulaire (SFGM-TC). Bone Marrow Transplant 2005; 35: 165–169.
Bladé J, Samson D, Reece D, Apperley J, Björkstrand B, Gahrton G et al. Criteria for evaluating disease response and progression in patients with multiple myeloma treated by high-dose therapy and haemopoietic stem cell transplantation. Myeloma Subcommittee of the EBMT. European Group for Blood and Marrow Transplant. Br J Haematol 1998; 102: 1115–1123.
Weber DM, Chen C, Niesvizky R, Wang M, Belch A, Stadtmauer EA et al. Multiple Myeloma (009) Study Investigators. Lenalidomide plus dexamethasone for relapsed multiple myeloma in North America. N Engl J Med 2007; 357: 2133–2142.
Dimopoulos M, Spencer A, Attal M, Prince HM, Harousseau JL, Dmoszynska A et al. Multiple Myeloma (010) Study Investigators. Lenalidomide plus dexamethasone for relapsed or refractory multiple myeloma. N Engl J Med 2007; 357: 2123–2132.
Minnema MC, van der Veer MS, Aarts T, Emmelot M, Mutis T, Lokhorst HM . Lenalidomide alone or in combination with dexamethasone is highly effective in patients with relapsed multiple myeloma following allogeneic stem cell transplantation and increases the frequency of CD4(+)Foxp3(+) T cells. Leukemia 2009; 23: 605–607.
Hayashi T, Hideshima T, Akiyama M, Podar K, Yasui H, Raje N et al. Molecular mechanisms whereby immunomodulatory drugs activate natural killer cells: clinical application. Br J Haematol 2005; 128: 192–203.
Davies FE, Raje N, Hideshima T, Lentzsch S, Young G, Tai YT et al. Thalidomide and immunomodulatory derivatives augment natural killer cell cytotoxicity in multiple myeloma. Blood 2001; 98: 210–216.
Beyer M, Kochanek M, Giese T, Endl E, Weihrauch MR, Knolle PA et al. In vivo peripheral expansion of naive CD4+CD25high FoxP3+ regulatory T cells in patients with multiple myeloma. Blood 2006; 107: 3940–3949. E-pub 2006 Jan 12.
Rezvani K, Mielke S, Ahmadzadeh M, Kilical Y, Savani BN, Zeilah J et al. High donor FOXP3-positive regulatory T-cell (Treg) content is associated with a low risk of GVHD following HLA-matched allogeneic SCT. Blood 2006; 108: 1291–1297.
Atanackovic D, Cao Y, Luetkens T, Panse J, Faltz C, Arfsten J et al. CD4+CD25+FOXP3+ T regulatory cells reconstitute and accumulate in the bone marrow of patients with multiple myeloma following allogeneic stem cell transplantation. Haematologica 2008; 93: 423–430.
Acknowledgements
We thank the staff of the BMT unit for providing excellent care of our patients, and the medical technicians for their excellent work in the laboratories. This work was supported in part by a grant from the Deutsche José Carreras Leukämie-Stiftung e.V. (to NK)
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Lioznov, M., El-Cheikh, J., Hoffmann, F. et al. Lenalidomide as salvage therapy after allo-SCT for multiple myeloma is effective and leads to an increase of activated NK (NKp44+) and T (HLA-DR+) cells. Bone Marrow Transplant 45, 349–353 (2010). https://doi.org/10.1038/bmt.2009.155
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/bmt.2009.155
Keywords
This article is cited by
-
Comparison of Outcomes of Donor Lymphocyte Infusions With or Without Lenalidomide in Patients with Hematological Malignancies Post Allogeneic HSCT
Indian Journal of Hematology and Blood Transfusion (2023)
-
Unleashing the power of NK cells in anticancer immunotherapy
Journal of Molecular Medicine (2022)
-
Safety and tolerability of lenalidomide maintenance in post-transplant acute myeloid leukemia and high-risk myelodysplastic syndrome
Bone Marrow Transplantation (2021)
-
A systematic scoping review on natural killer cell function in colorectal cancer
Cancer Immunology, Immunotherapy (2021)
-
Lenalidomide improves the therapeutic effect of an interferon-α-dendritic cell-based lymphoma vaccine
Cancer Immunology, Immunotherapy (2019)