Abstract
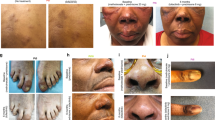
Alefacept (Amevive) is an immunosuppressive dimeric fusion protein that is used for psoriasis control. We recently showed its effect in acute steroid-resistant/dependent GVHD. In this study, we describe the effect of alefacept treatment on chronic extensive GVHD (cGVHD). Twelve patients were included in this study; of these 8 (9 of 13 episodes) showed response. The median time to initial response was 2.25 weeks and the response was marked (n=3), moderate (n=2) or minimal (n=4). In two responding patients, the response was only temporary. Complications that appeared during treatment included infection, pericarditis and squamous cell carcinoma of the lip. All these events may be related to other drugs given simultaneously. With a 30-month median follow-up, 6 of 12 patients are alive, with all but one with stable or improved cGVHD. Six patients died because of GVHD progression, whereas none of the patients experienced relapse of the disease for which the transplantation was done. As reported earlier in psoriatic patients treated with alefacept, we found a consistent increase in the percentage of naive T cells as a consequence of treatment. In conclusion, alefacept is effective for the treatment of cGVHD, and dose and time intervals of treatment should be explored further.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout


Similar content being viewed by others
References
Lebwohl M, Christophers E, Langley R, Ortonne JP, Roberts J, Griffiths CE . An international, randomized, double-blind, placebo-controlled phase 3 trial of intramuscular alefacept in patients with chronic plaque psoriasis. Arch Dermatol 2003; 139: 719–727.
Ellis CN, Krueger GG, Alefacept Clinical Study Group. Treatment of chronic plaque psoriasis by selective targeting of memory effector T lymphocytes. N Engl J Med 2001; 345: 248–255.
Graves JE, Nunley K, Heffernan MP . Off-label uses of biologics in dermatology: rituximab, omalizumab, infliximab, etanercept, adalimumab, efalizumab, and alefacept. J Am Acad Dermatol 2007; 56: e55–e79.
Chamian F, Lin SL, Lee E, Kikuchi T, Gilleaudeau P, Sullivan-Whalen M et al. Alefacept (anti-CD2) causes a selective reduction in circulating effector memory T cells (Tem) and relative preservation of central memory T cells (Tcm) in psoriasis. J Transl Med 2007; 5: 27–33.
Larsen R, Ryder LP, Svejgaard A, Gniadecki R . Changes in circulating lymphocyte subpopulations following administration of the leucocyte function-associated antigen-3 (LFA-3)/IgG1 fusion protein alefacept. Clin Exp Immunol 2007; 149: 23–30.
Fraser CJ, Scott Baker K . The management and outcome of chronic graft-versus-host disease. Br J Haematol 2007; 138: 131–145.
Shapira MY, Resnick IB, Bitan M, Ackerstein A, Tsirigotis P, Gesundheit B et al. Rapid response to alefacept given to patients with steroid resistant or steroid dependent acute graft-versus-host disease: a preliminary report. Bone Marrow Transplant 2005; 36: 1097–1101.
Toor AA, Stiff PJ, Nickoloff BJ, Rodriguez T, Klein JL, Gordon KB . Alefacept in corticosteroid refractory graft versus host disease: early results indicate promising activity. J Dermatolog Treat 2007; 18: 13–18.
Pavletic SZ, Martin P, Lee SJ, Mitchell S, Jacobsohn D, Cowen EW et al. Measuring therapeutic response in chronic graft-versus-host disease: National Institutes of Health Consensus Development Project on Criteria for Clinical Trials in Chronic Graft-versus-Host Disease: IV. Response Criteria Working Group report. Biol Blood Marrow Transplant 2006; 12: 252–266.
Pugatsch T, Oppenheim A, Slavin S . Improved single-step PCR assay for sex identification post-allogeneic sex-mismatched BMT. Bone Marrow Transplant 1996; 17: 273–275.
Nakamura Y, Leppert O, O'Connel P, Wolff R, Holm T, Culver M et al. Variable number of tandem repeats (VNTR) markers for human gene mapping. Science 1987; 235: 1616–1622.
Atkinson K . Chronic graft-versus-host disease. Bone Marrow Transplant 1990; 5: 69–82.
Deeg HJ, Leisenring W, Storb R, Nims J, Flowers ME, Witherspoon RP et al. Long-term outcome after marrow transplantation for severe aplastic anemia. Blood 1998; 91: 3637–3645.
Kulkarni S, Powles R, Treleaven J, Riley U, Singhal S, Horton C et al. Chronic graft versus host disease is associated with long-term risk for pneumococcal infections in recipients of bone marrow transplants. Blood 2000; 95: 3683–3686.
Atkinson K, Horowitz MM, Gale RP, van Bekkum DW, Gluckman E, Good RA et al. Risk factors for chronic graft-versus-host disease after HLA-identical sibling bone marrow transplantation. Blood 1990; 75: 2459–2464.
Ochs LA, Miller WJ, Filipovich AH, Haake RJ, McGlave PB, Blazar BR et al. Predictive factors for chronic graft-versus-host disease after histocompatible sibling donor bone marrow transplantation. Bone Marrow Transplant 1994; 13: 455–460.
Carlens S, Ringdén O, Remberger M, Lönnqvist B, Hägglund H, Klaesson S et al. Risk factors for chronic graft-versus-host disease after bone marrow transplantation: a retrospective single centre analysis. Bone Marrow Transplant 1998; 22: 755–761.
Marks DI, Cullis JO, Ward KN, Lacey S, Syzdlo R, Hughes TP . Allogeneic bone marrow transplantation for chronic myeloid leukemia using sibling and volunteer unrelated donors: a comparison of complications in the first 2 years. Ann Intern Med 1993; 119: 207–214.
Cutler C, Giri S, Jeyapalan S, Paniagua D, Viswanathan A, Antin JH . Acute and chronic graft-versus-host disease after allogeneic peripheral-blood stem-cell and bone marrow transplantation: a meta-analysis. J Clin Oncol 2001; 19: 3685–3691.
Przepiorka D, Anderlini P, Saliba R, Cleary K, Mehra R, Khouri I et al. Chronic graft-versus-host disease after allogeneic blood stem cell transplantation. Blood 2001; 98: 1695–1700.
Shlomchik WD, Couzens MS, Tang CB, McNiff J, Robert ME, Liu J et al. Prevention of graft-versus-host disease by inactivation of host antigen-presenting cells. Science 1999; 285: 412–415.
Martin PJ, Schoch G, Fisher L, Byers V, Anasetti C, Appelbaum FR et al. A retrospective analysis of therapy for acute graft versus host disease: initial treatment. Blood 1990; 76: 1464–1472.
Weisdorf D, Haake R, Blazar B, Miller W, McGlave P, Ramsay N et al. Treatment of moderate/severe acute graft versus host disease after allogeneic bone marrow transplantation: an analysis of clinical risk features and outcome. Blood 1990; 75: 1024–1030.
Chatenoud L . Monoclonal antibody-based strategies in autoimmunity and transplantation. Methods Mol Med 2005; 109: 297–328.
Zeiser R, Marks R, Bertz H, Finke J . Immunopathogenesis of acute graft-versus-host disease: implications for novel preventive and therapeutic strategies. Ann Hematol 2004; 83: 551–565.
Ghoreschi K, Weigert C, Röcken M . Immunopathogenesis and role of T cells in psoriasis. Clin Dermatol 2007; 25: 574–580.
Bashir SJ, Maibach HI . Alefacept (Biogen). Curr Opin Investig Drugs 2001; 2: 631–634.
Chamian F, Lowes MA, Lin SL, Lee E, Kikuchi T, Gilleaudeau P et al. Alefacept reduces infiltrating T cells, activated dendritic cells, and inflammatory genes in psoriasis vulgaris. Proc Natl Acad Sci USA 2005; 102: 2075–2080.
Cooper JC, Morgan G, Harding S, Subramanyam M, Majeau GR, Moulder K et al. Alefacept selectively promotes NK cell-mediated deletion of CD45R0+ human T cells. Eur J Immunol 2003; 33: 666–675.
Acknowledgements
Dr Michael Shapira's study is supported by the Dr Sima Lior Fund and by the Zaltzberg Fund.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Shapira, M., Abdul-Hai, A., Resnick, I. et al. Alefacept treatment for refractory chronic extensive GVHD. Bone Marrow Transplant 43, 339–343 (2009). https://doi.org/10.1038/bmt.2008.324
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/bmt.2008.324
Keywords
This article is cited by
-
Activation and regulation of alloreactive T cell immunity in solid organ transplantation
Nature Reviews Nephrology (2022)
-
Steroid-refractory chronic graft-versus-host disease: treatment options and patient management
Bone Marrow Transplantation (2021)
-
Keratinocytes costimulate naive human T cells via CD2: a potential target to prevent the development of proinflammatory Th1 cells in the skin
Cellular & Molecular Immunology (2020)
-
More than skin deep? Emerging therapies for chronic cutaneous GVHD
Bone Marrow Transplantation (2013)
-
Advances in immunosuppression for renal transplantation
Nature Reviews Nephrology (2010)