Abstract
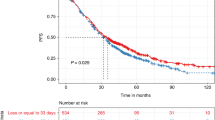
Reported results of high-dose therapy (HDT) reflect the combined effect of initial therapy and HDT. The incremental contribution of HDT is often difficult to analyze with varying degrees of response pre-HDT. Here we analyze results of HDT in patients with measurable disease at transplant, defined as a serum or 24 h urine M protein of >1.0 g per 100 ml and >200 mg per day, respectively. Paraprotein responses were calculated using measurements prior to HDT and the lowest subsequent measurement. A total of 431 patients were studied; 264 (61.3%) transplanted within 1-year of diagnosis. An additional reduction in paraprotein by 50% following HDT was seen in 86% patients; with 129 patients (30%) obtaining a 90% reduction. Patients with at least a 90% reduction had longer time to progression with no overall survival advantage and this was independent of other prognostic factors for decreased risk of progression. This study provides an estimate of the degree of tumor reduction provided by HDT, in addition to that provided by the initial therapy. In this group of patients with measurable disease after initial therapy, HDT therapy leads to complete responses in nearly a quarter of the patients and a 90% reduction in another 7%, an outcome associated with better progression-free survival.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout



Similar content being viewed by others
References
Attal M, Harousseau JL, Stoppa AM, Sotto JJ, Fuzibet JG, Rossi JF et al. A prospective, randomized trial of autologous bone marrow transplantation and chemotherapy in multiple myeloma. Intergroupe Francais du Myelome. N Engl J Med 1996; 335: 91–97.
Child JA, Morgan GJ, Davies FE, Owen RG, Bell SE, Hawkins K et al. High-dose chemotherapy with hematopoietic stem-cell rescue for multiple myeloma. N Engl J Med 2003; 348: 1875–1883.
Fermand JP, Katsahian S, Divine M, Leblond V, Dreyfus F, Macro M et al. High-dose therapy and autologous blood stem-cell transplantation compared with conventional treatment in myeloma patients aged 55–65 years: long-term results of a randomized control trial from the Group Myelome-Autogreffe. J Clin Oncol 2005; 23: 9227–9233.
Fermand JP, Ravaud P, Chevret S, Divine M, Leblond V, Belanger C et al. High-dose therapy and autologous peripheral blood stem cell transplantation in multiple myeloma: up-front or rescue treatment? Results of a multicenter sequential randomized clinical trial. Blood 1998; 92: 3131–3136.
Moreau P, Milpied N, Mahe B, Juge-Morineau N, Rapp MJ, Bataille R et al. Melphalan 220 mg/m2 followed by peripheral blood stem cell transplantation in 27 patients with advanced multiple myeloma. Bone Marrow Transplant 1999; 23: 1003–1006.
Moreau P, Kergueris MF, Milpied N, Le Tortorec S, Mahe B, Bulabois CE et al. A pilot study of 220 mg/m2 melphalan followed by autologous stem cell transplantation in patients with advanced haematological malignancies: pharmacokinetics and toxicity. Br J Haematol 1996; 95: 527–530.
Giralt S, Bensinger W, Goodman M, Podoloff D, Eary J, Wendt R et al. 166Ho-DOTMP plus melphalan followed by peripheral blood stem cell transplantation in patients with multiple myeloma: results of two phase 1/2 trials. Blood 2003; 102: 2684–2691.
Attal M, Harousseau JL, Facon T, Guilhot F, Doyen C, Fuzibet JG et al. Single versus double autologous stem-cell transplantation for multiple myeloma. N Engl J Med 2003; 349: 2495–2502.
Barlogie B, Kyle RA, Anderson KC, Greipp PR, Lazarus HM, Hurd DD et al. Standard chemotherapy compared with high-dose chemoradiotherapy for multiple myeloma: final results of phase III US Intergroup Trial S9321. J Clin Oncol 2006; 24: 929–936.
Greipp PR, Lust JA, O'Fallon WM, Katzmann JA, Witzig TE, Kyle RA . Plasma cell labeling index and beta 2-microglobulin predict survival independent of thymidine kinase and C-reactive protein in multiple myeloma. Blood 1993; 81: 3382–3387.
Blade J, Samson D, Reece D, Apperley J, Bjorkstrand B, Gahrton G et al. Criteria for evaluating disease response and progression in patients with multiple myeloma treated by high-dose therapy and haemopoietic stem cell transplantation. Myeloma Subcommittee of the EBMT. European Group for Blood and Marrow Transplant. Br J Haematol 1998; 102: 1115–1123.
Kaplan E, Meier P . Non-parametric estimation from incomplete observations. J Am Stat Assoc 1958; 53: 457–481.
Cox D . Regression models and life tables. J R Stat Soc 1972; 34: 187–202.
Dingli D, Nowakowski GS, Dispenzieri A, Lacy MQ, Hayman S, Litzow MR et al. Cyclophosphamide mobilization does not improve outcome in patients receiving stem cell transplantation for multiple myeloma. Clin Lymphoma Myeloma 2006; 6: 384–388.
McElwain TJ, Powles RL . High-dose intravenous melphalan for plasma-cell leukaemia and myeloma. Lancet 1983; 2: 822–824.
Barlogie B, Hall R, Zander A, Dicke K, Alexanian R . High-dose melphalan with autologous bone marrow transplantation for multiple myeloma. Blood 1986; 67: 1298–1301.
McElwain TJ, Selby PJ, Gore ME, Viner C, Meldrum M, Millar BC et al. High-dose chemotherapy and autologous bone marrow transplantation for myeloma. Eur J Haematol Suppl 1989; 51 (Suppl 1989): 152–156.
Singhal S, Powles R, Sirohi B, Treleaven J, Kulkarni S, Mehta J . Response to induction chemotherapy is not essential to obtain survival benefit from high-dose melphalan and autotransplantation in myeloma. Bone Marrow Transplant 2002; 30: 673–679.
Alexanian R, Dimopoulos MA, Delasalle KB, Hester J, Champlin R . Myeloablative therapy for primary resistant multiple myeloma. Stem Cells 1995; 13 (Suppl 2): 118–121.
Kumar S, Lacy MQ, Dispenzieri A, Rajkumar SV, Fonseca R, Geyer S et al. High-dose therapy and autologous stem cell transplantation for multiple myeloma poorly responsive to initial therapy. Bone Marrow Transplant 2004; 34: 161–167.
Blade J, Rosinol L, Garcia-Sanz R, Lahuerta J, Hernandez M, Sureda A et al. A PETHEMA study of high-dose therapy/stem cell support (HDT), including tandem transplant, in primary refractory multiple myeloma (MM): identification of two populations with different outcomes. J Clin Oncol (Meeting Abstracts) 2007; 25 (18 Suppl): 8021.
Rajkumar SV, Fonseca R, Dispenzieri A, Lacy MQ, Witzig TE, Lust JA et al. Effect of complete response on outcome following autologous stem cell transplantation for myeloma. Bone Marrow Transplant 2000; 26: 979–983.
Harousseau J-L, Attal M, Moreau P, Garban F, Facon T, Avet-Loiseau H . The prognostic impact of complete remission (CR) plus very good partial remission (VGPR) in a double-transplantation program for newly diagnosed multiple myeloma (MM). Combined results of the IFM 99 trials. ASH Annual Meeting Abstracts 2006; 108: 3077.
Dingli D, Pacheco JM, Nowakowski GS, Kumar SK, Dispenzieri A, Hayman SR et al. Relationship between depth of response and outcome in multiple myeloma. J Clin Oncol 2007; 25: 4933–4937.
Lenhoff S, Hjorth M, Turesson I, Westin J, Gimsing P, Wisloff F et al. Intensive therapy for multiple myeloma in patients younger than 60 years. Long-term results focusing on the effect of the degree of response on survival and relapse pattern after transplantation. Haematologica 2006; 91: 1228–1233.
Facon T, Mary JY, Hulin C, Benboubker L, Attal M, Pegourie B et al. Melphalan and prednisone plus thalidomide versus melphalan and prednisone alone or reduced-intensity autologous stem cell transplantation in elderly patients with multiple myeloma (IFM 99–06): a randomised trial. Lancet 2007; 370: 1209–1218.
Moreau P, Facon T, Attal M, Hulin C, Michallet M, Maloisel F et al. Comparison of 200 mg/m(2) melphalan and 8 Gy total body irradiation plus 140 mg/m(2) melphalan as conditioning regimens for peripheral blood stem cell transplantation in patients with newly diagnosed multiple myeloma: final analysis of the Intergroupe Francophone du Myelome 9502 randomized trial. Blood 2002; 99: 731–735.
Acknowledgements
This study was supported in part by Hematologic Malignancies Program, Mayo Clinic CR20 program, ASCO Young Investigator Award, Amgen Oncology Institute Junior Faculty Award (SK) and grants CA93842 and CA10080 from the National Cancer Institute; National Institutes of Health and the Department of Health and Human Services.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Kumar, S., Dingli, D., Dispenzieri, A. et al. Impact of additional cytoreduction following autologous SCT in multiple myeloma. Bone Marrow Transplant 42, 259–264 (2008). https://doi.org/10.1038/bmt.2008.166
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/bmt.2008.166
Keywords
This article is cited by
-
Impact of the time interval between end of induction and autologous hematopoietic transplantation in newly diagnosed patients with multiple myeloma
Bone Marrow Transplantation (2023)
-
Value of novel agents and intensive therapy for patients with multiple myeloma
Bone Marrow Transplantation (2014)
-
Attainment of at least a very good partial response after induction treatment is an important surrogate of longer survival for multiple myeloma
Bone Marrow Transplantation (2010)
-
CR represents an early index of potential long survival in multiple myeloma
Bone Marrow Transplantation (2010)