Abstract
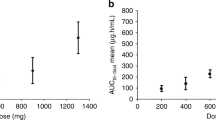
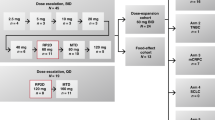
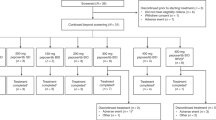
3,5-Dichloro-2,4-dimethoxy-6-(trichloromethyl)pyridine (penclomedine, NSC 338720, CRC 88-04) is an alpha-picoline derivative with anti-tumour activity in preclinical models. Penclomedine administration by 1-h intravenous infusion on 5 consecutive days was repeated 3 weekly in the absence of dose-limiting toxicity (DLT) or disease progression. Five dose levels were investigated (22.5-340 mg m(-2) day[-1]). Eight men and eight women were entered, median age 59 years (range 39-73 years), with good performance status (ECOG 0/1) in 11 patients. A total of 13 out of 16 patients had received previous chemotherapy. Common toxicity criteria grade (CTCg) II vomiting was recorded at all dose levels. Neurotoxicity (cerebellar ataxia and dizziness) was the DLT, CTCg III toxicity occurring in three out of three patients treated at 340 mg m(-2) day(-1). CTCg III dizziness was noted in one out of three patients at 250 mg m(-2) day(-1). Neurotoxicity developed during the 1-h infusion and persisted for a variable period (maximum 5 h) after infusion. Prophylactic antiemetic drugs appeared to reduce associated vomiting but did not prevent ataxia. No antiproliferative toxicities were noted and no anti-tumour responses were documented. Penclomedine pharmacokinetic studies confirmed preclinical evidence of extensive apparent distribution (93 l m[-2]) and rapid clearance (41 l h[-1] m[-2]). Purkinje cell loss has been identified in preclinical models after intraperitoneal administration (O'Reilly et al, 1996a) and further clinical development of penclomedine will focus on oral administration.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 24 print issues and online access
$259.00 per year
only $10.79 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout
Similar content being viewed by others
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Jodrell, D., Bowman, A., Stewart, M. et al. Dose-limiting neurotoxicity in a phase I study of penclomedine (NSC 388720, CRC 88-04), a synthetic α-picoline derivative, administered intravenously. Br J Cancer 77, 808–811 (1998). https://doi.org/10.1038/bjc.1998.131
Issue Date:
DOI: https://doi.org/10.1038/bjc.1998.131
This article is cited by
-
Carbonate and carbamate derivatives of 4-demethylpenclomedine as novel anticancer agents
Cancer Chemotherapy and Pharmacology (2009)
-
Thiolo-, thiono- and dithiocarbonate and thiocarbamate derivatives of demethylpenclomedine as novel anticancer agents
Cancer Chemotherapy and Pharmacology (2006)
-
Evaluation of rodent-only toxicology for early clinical trials with novel cancer therapeutics
British Journal of Cancer (1999)