Abstract
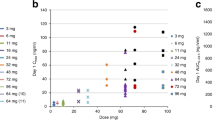
The results of preclinical and clinical studies indicate enhanced antineoplastic activity of topotecan (SKF 104864-A) when administered as a chronic treatment. We determined the apparent bioavailability and pharmacokinetics of topotecan administered orally to 12 patients with solid tumours in a two-part crossover study. The oral dose of 1.5 mg m-2 was administered as a drinking solution of 200 ml on day 1. The i.v. dose of 1.5 mg m-2 was administered as a 30 min continuous infusion on day 2. The bioavailability was calculated as the ratio of the oral to i.v. area under the curve (AUC) calculated up to the last measured time point. The oral drinking solution was well tolerated. The bioavailability revealed moderate inter-patient variation and was 30% +/- 7.7% (range 21-45%). The time to maximum plasma concentration after oral administration (Tmax) was 0.78 h (median; range 0.33-2.5). Total i.v. plasma clearance of topotecan was 824 +/- 154 ml min-1 (range 535-1068 ml min(-1)). The AUC ratio of topotecan and the lactone ring-opened hydrolysis product (hydroxy acid) was of the same order after oral (0.34-1.13) and i.v. (0.47-0.98) administration. The bioavailability of topotecan after oral administration illustrates significant systemic exposure to the drug which may enable chronic oral treatment.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 24 print issues and online access
$259.00 per year
only $10.79 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout
Similar content being viewed by others
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Schellens, J., Creemers, G., Beijnen, J. et al. Bioavailability and pharmacokinetics of oral topotecan: a new topoisomerase I inhibitor. Br J Cancer 73, 1268–1271 (1996). https://doi.org/10.1038/bjc.1996.243
Issue Date:
DOI: https://doi.org/10.1038/bjc.1996.243
This article is cited by
-
Pharmacokinetics of metronomic chemotherapy: a neglected but crucial aspect
Nature Reviews Clinical Oncology (2016)
-
5,7-Dimethoxyflavone and Multiple Flavonoids in Combination Alter the ABCG2-Mediated Tissue Distribution of Mitoxantrone in Mice
Pharmaceutical Research (2011)
-
Involvement of P-glycoprotein, Multidrug Resistance Protein 2 and Breast Cancer Resistance Protein in the Transport of Belotecan and Topotecan in Caco-2 and MDCKII Cells
Pharmaceutical Research (2008)
-
Phase-I study of a new schedule based on increasing days of topotecan administration associated with dose individualisation
Cancer Chemotherapy and Pharmacology (2006)
-
Pharmacokinetics, pharmacodynamics and adherence to oral topotecan in myelodysplastic syndromes: a Cancer and Leukemia Group B study
Cancer Chemotherapy and Pharmacology (2006)