Abstract
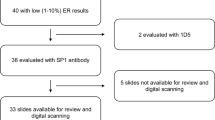
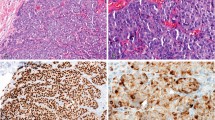
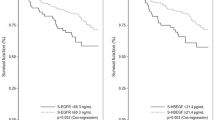
Epidermal growth factor receptor (EGFR) has been the subject of much research since it was first described as a prognostic factor in breast cancer. The assay methods used and results obtained vary widely between studies. In this study 88 primary breast cancers were assayed for EGFR using a novel immunohistochemical assay performed on paraffin-embedded sections. The monoclonal antibody used was raised against purified, denatured EGFR, reacts with an epitope on the external domain and does not interfere with ligand binding. Twenty-two per cent of the tumours were EGFR positive using this assay. The results obtained were significantly correlated with those obtained by ligand-binding assay (r = 0.621, P = 0.011). The concordance rate was 82% (P < 0.001). The majority of discordant results could be explained by the presence of benign breast tissue and other non-malignant elements which could be seen to express EGFR on the immunohistochemical assay and were excluded from the score for this, but would be incorporated into ligand-binding assay results. The well-established inverse relationship between EGFR (as measured by this assay) and oestrogen receptor (ER) was seen (chi 2 = 24.9, P < 0.0001). In addition, in this exploratory study on a limited tumour set, EGFR was a significant adverse prognostic factor (on univariate but not multivariate analysis) for both relapse-free survival (P = 0.02) and overall survival (P = 0.03) when measured by this immunohistochemical assay, but was not significant when measured by ligand-binding assay.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 24 print issues and online access
$259.00 per year
only $10.79 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout
Similar content being viewed by others
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Newby, J., A'Hern, R., Leek, R. et al. Immunohistochemical assay for epidermal growth factor receptor on paraffin-embedded sections: validation against ligand-binding assay and clinical relevance in breast cancer. Br J Cancer 71, 1237–1242 (1995). https://doi.org/10.1038/bjc.1995.239
Issue Date:
DOI: https://doi.org/10.1038/bjc.1995.239
This article is cited by
-
Prognostic and predictive value of p-Akt, EGFR, and p-mTOR in early breast cancer
Strahlentherapie und Onkologie (2014)
-
Overexpression of epithelial growth factor receptor (EGFR) predicts better response to neo-adjuvant chemotherapy in patients with triple-negative breast cancer
Journal of Translational Medicine (2012)
-
Regulation of IMP3 by EGFR signaling and repression by ERβ: implications for triple-negative breast cancer
Oncogene (2012)
-
Additive antitumour effect of the epidermal growth factor receptor tyrosine kinase inhibitor gefitinib (Iressa, ZD1839) and the antioestrogen fulvestrant (Faslodex, ICI 182,780) in breast cancer cells
British Journal of Cancer (2004)
-
Expression of epidermal growth factor receptor in gestational trophoblastic diseases
Journal of Cancer Research and Clinical Oncology (1997)