Abstract
Aim:
To develop an efficient RNA interference system using phagemid particles displaying the epidermal growth factor (EGF) ligand.
Methods:
pSilencer1.0-siEGFP and pSilencer4.1-siAkt plasmids were constructed by gene clone technology. The modified helper phage genome (plasmid) M13KO7EGFCT was used to package phagemids, such as pSilencer1.0-siEGFP and pSilencer4.1-siAkt. ELISA was used to quantify the titer of the progeny virus particles. Single-strand DNA was extracted and analyzed by agarose gel electrophoresis to evaluate the percentage of the phagemid particles. The expression level of the reporter gene enhanced green fluorescence protein (EGFP) was determined by transducing phagemid particles packaging pSilencer1.0-siEGFP into cells. The level of Akt gene expression in cells transduced phagemid particles packaging pSilencer4.1-siAkt was examined by Western blotting. Hydroxycamptothecin (HCPT) was used to enhance the gene transduction efficiency.
Results:
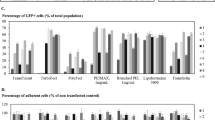
RNAi vectors pSilencer1.0-siEGFP and pSilencer4.1-siAkt were successfully constructed. Phagemid-encoding siRNA can be packaged efficiently. After the cells were infected by EGF displaying phagemid particles in the presence of HCPT, the expression of the target gene EGFP or Akt was substantially downregulated.
Conclusion:
Cell-targeted phagemid particles are efficient siRNA delivery vectors in the presence of HCPT.
Similar content being viewed by others
Article PDF
References
Burg MA, Jensen-Pergakes K, Gonzalez AM, Ravey P, Baird A, Larocca D . Enhanced phagemid particle gene transfer in camptothecin-treated carcinoma cells. Cancer Res 2002; 62: 977–81.
Larocca D, Witte A, Johnson W, Pierce GF, Baird A . Targeting bacteriophage to mammalian cell surface receptors for gene delivery. Hum Gene Ther 1998; 9: 2393–9.
Larocca D, Kassner PD, Witte A, Ladner RC, Pierce GF, Baird A . Gene transfer to mammalian cells using genetically targeted filamentous bacteriophage. FASEB J 1999; 13: 727–34.
Kassner PD, Burg MA, Baird A, Larocca D . Genetic selection of phage engineered for receptor-mediated gene transfer to mammalian cells. Biochem Biophys Res Commun 1999; 264: 921–8.
Li ZH, Zhang J, Zhao RJ, Xu YH, Gu JR . Preparation of peptide-targeted phagemid particles using a protein III-modified helper phage. Biotechniques 2005; 39: 493–7.
Poul MA, Marks JD . Targeted gene delivery to mammalian cells by filamentous bacteriophage. J Mol Biol 1999; 288: 203–11.
Di Giovine M, Salone B, Martina Y, Amati V, Zambruno G, Cundari E, et al. Cell delivery and gene transfer of adenoviral penton base displaying bacteriophage. Virology 2001; 282: 102–12.
Li ZH, Jiang H, Zhang J, Shi BZ, Gu JR . Cell targeted phagemid particles preparation using Ecoli bearing ligand-pIII encoding helper phage genome. Bio Techniques 2006; 41: 706–7.
Sui G, Soohoo C, Affar el B, Gay F, Shi Y, Forrester WC, et al. A DNA vector-based RNAi technology to suppress gene expression in mammalian cells. Proc Natl Acad Sci USA 2002; 99: 5515–20.
Katome T, Obata T, Matsushimal R, Masuyama N, Cantley LC, Gotoh Y, et al. Use of RNA interference-mediated gene silencing and adenoviral overexpression to elucidate the roles of AKT/protein kinase B isoforms in insulin actions. J Biol Chem 2003; 278: 28312–23.
Barbas CF . Phage display: a laboratory manual. CSH Laboratory Press; 2001.
Rondot S, Koch J, Breitling F, Dübel S . A helper phage to improve single-chain antibody presentation in pahge display. Nat Biotechnol 2001; 19: 75–78.
Alexander IE, Russell DW, Miller AD . DNA-damaging agents greatly increase the transduction of nondividing cells by adeno-associated virus vectors. J Virol 1994; 68: 8282–7.
Gao CS, Mao SL, Lo CL, Wirsching P, Lerner RA, Janda KD . Making artificial antibodies: a format for phage display of combinatorial heterodimeric arrays. Proc Natl Acad Sci USA 1999; 96: 6025–30.
Kwaśnikowski P, Kristensen P, Markiewicz WT . Multivalent display system on filamentous bacteriophage pVII minor coat protein. J Immunol Methods 2005; 307: 135–43.
Gao C, Mao S, Ditzel HJ, Farnaes L, Wirsching P, Lerner RA, et al. A cell-penetrating peptide from a novel pVII-pIX phage-displayed random peptide library. Bioorg Med Chem 2002; 10: 4057–65.
Zhang R, Li Y, Cai Q, Liu T, Sun H, Chambless B, et al. Preclinical pharmacology of the nature product anticancer agent 10-hydroxycamptothecin, an inhibitor or topoisomerase I. Cancer Chemother Pharmacol 1998; 41: 257–67.
Zhou JJ, Liu J, Xu B . Relation between lactone ring forms of HCPT and their antitumor activities. Acta Pharmacol Sin 2001; 22: 827–30.
Author information
Authors and Affiliations
Corresponding author
Additional information
Project supported by the National 973 Basic Research Program of China (No 2004CB518802) and the Science and Technology Commission of Shanghai Municipality (No 054119568).
Rights and permissions
About this article
Cite this article
Jiang, H., Cai, Xm., Shi, Bz. et al. Development of efficient RNA interference system using EGF-displaying phagemid particles. Acta Pharmacol Sin 29, 437–442 (2008). https://doi.org/10.1111/j.1745-7254.2008.00768.x
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1111/j.1745-7254.2008.00768.x
Keywords
This article is cited by
-
Pseudovirions as Vehicles for the Delivery of siRNA
Pharmaceutical Research (2010)
-
Inhibition of cell growth and invasion by epidermal growth factor-targeted phagemid particles carrying siRNA against focal adhesion kinase in the presence of hydroxycamptothecin
BMC Biotechnology (2008)