Abstract
Aim:
The aim of the present study was to design, synthesize, and evaluate novel antibacterial agents, derivatives of aryl-4-guanidinomethylbenzoate and N-aryl-4-guanidinomethylbenzamide.
Methods:
A total of 44 derivatives of aryl-4-guanidinomethylbenzoate (series A) and N-aryl-4-guanidinomethylbenzamide (series B) were synthesized and their antibacterial activities were assessed in vitro against a variety of Gram-positive and Gram-negative bacteria by an agar dilution method.
Results:
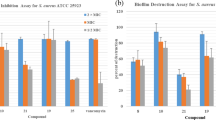
Twelve compounds showed potent bactericidal effects against a panel of Gram-positive germs, including methicillin-resistant Staphylococcus aureus (MRSA), vancomycin-resistant enterococci (VRE), vancomycin-intermediate Staphylococcus aureus (VISA), and methicillin-resistant coagulase-negative staphylococci (MRCNS), with minimum inhibitory concentrations (MIC) ranging between 0.5 and 8 μg/mL, which were comparable to the MIC values of several marketed antibiotics. They exhibited weak or no activity on the Gram-negative bacteria tested. In addition, these compounds displayed high inhibitory activities towards oligopeptidase B of bacterial origin.
Conclusion:
In comparison with the previously reported MIC values of several known antibiotics, the derivatives of aryl-4-guanidinomethylbenzoate and N-aryl-4-guanidinomethylbenzamide showed comparable in vitro bactericidal activities against VRE and VISA as linezolid. Their growth inhibitory effects on MRSA were similar to vancomycin, but were less potent than linezolid and vancomycin against MRCNS. This class of compounds may have the potential to be developed into narrow spectrum antibacterial agents against certain drug-resistant strains of bacteria.
Similar content being viewed by others
Article PDF
References
Fisher JF, Meroueh SO, Mobashery S . Bacterial resistance to β-lactam antibiotics: compelling opportunism, compelling opportunity. Chem Rev 2005; 105: 395–424.
Sandhya RG, McColm J, Clements JM, Taupin P, Barrowcliffe S, Hevizi S, et al. Pharmacokinetics in animals and humans of a first-in-class peptide deformylase inhibitor. Antimicrob Agents Chemother 2004; 48: 4835–42.
Jarvest RL, Berge JM, Berry V, Boyd HF, Brown MJ, Elder JS, et al. Nanomolar inhibitors of Staphylococcus aureus methionyl tRNA synthetase with potent antibacterial activity against Gram-positive pathogens. J Med Chem 2002; 45: 1959–62.
Dyatkina NB, Roberts CD, Keicher JD, Dai Y, Nadherny JP, Zhang W, et al. Minor groove DNA binders as antimicrobial agents. Pyrrole tetraamides are potent antibacterials against vancomycin resistant Enteroccoci and methicillin resistant Staphylococcus aureus. J Med Chem 2002; 45: 805–17.
Stefano D, Letizia B, Stefania S, Margherita S, Sofia S . Discovering novel antibacterial agents by high throughput screening. Front Drug Des Discov 2005; 1: 3–16.
Ko KS, Kim YS, Song JH, Yeom JS, Lee H, Jung SI, et al. Genotypic diversity of methicillin-resistant Staphylococcus aureus isolates in Korean hospitals. Antimicrob Agents Chemother 2005; 49: 3583–5.
Raad II, Hanna HA, Hachem RY, Dvorak T, Arbuckle RB Chaiban G, et al. Clinical-use-associated decrease in susceptibility of vancomycin-resistant Enterococcus faecium to linezolid: a comparison with quinupristin-dalfopristin. Antimicrob Agents Chemother 2004; 48: 3583–5.
Greenhill J V, Lue P . Amidines and guanidines in medicinal chemistry. Prog Med Chem 1993; 30: 203–326.
Berlinck RG, Kossuga MH . Natural guanidine derivatives. Nat Prod Rep 2005; 22: 516–50.
Berlinck RG . Natural guanidine derivatives. Nat Prod Rep 2002; 19: 617–49.
Berlinck RG . Natural guanidine derivatives. Nat Prod Rep 1999; 16: 339–65.
Berlinck RG . Natural guanidine derivatives. Nat Prod Rep 1996; 13: 377–409.
Bycroft BW, Cameron D, Johnson AW . Synthesis of capreomycidine and epicapreomycidine, the epimers of alpha-(2-iminohexahydropyrimid-4-yl)glycine. J Chem Soc C 1971; 30: 40–7.
Bycroft BW, Croft LR, Johnson AW, Webb T . Viomycin. Part I. The structure of the guanidine-containing unit. J Chem Soc Perkin Trans 1 1972; 820–7.
Yoshioka H, Aoki T, Goko H, Nakatsu K, Noda T, Sakakibara H, et al. Chemical studies on tuberactinomycin. II. The structure of tuberactinomycin 0. Tetrahedron Lett 1971; 12: 2043–6.
Schwab LE, Garner OB, Schuksz M, Crawford BE, Esko JD, Tor Y . Guanidinylated neomycin delivers large, bioactive cargo into cells through a heparan sulfate-dependent pathway. J Biol Chem 2007; 282: 13585–91.
Kato M, Irisawa T, Ohtani M, Muramatu M . Purification and characterization of proteinase In: a trypsin-like proteinase, in Escherichia coli. Eur J Biochem 1992; 210: 1007–14.
Kato M, Irisawa T, Morimoto Y, Muramatu M . A trypsin inhibitor trans-4-guanidinomethylcyclohexanecarboxylic acid 4-tert-butylphenyl ester suppresses the onset of DNA synthesis in Escherichia coli cells synchronized by phosphate starvation. Biol Pharm Bull 1993; 16: 552–7.
Kato M, Irisawa T, Morimoto Y, Muramatu M . Effects of synthetic trypsin inhibitors on the growth of Escherichia coli. Biol Pharm Bull 1993; 16: 120–4.
Irisawa T, Kato M, Moroishi J, Muramatu M . Effect of trans-4-guanidinomethylcyclohexanecarboxylic acid 4-tert-butylphenyl ester, a trypsin inhibitor, on the growth of various strains of Escherichia coli. Biol Pharm Bull 1993; 16: 621–6.
Kamoda O, Anzai K, Mizoguchi J, Shiojiri M, Yanagi T, Nishino T, et al. In vitro activity of a novel antimicrobial agent, TG44, for treatment of Helicobacter pylori infection. Antimicrob Agents Chemother 2006; 50: 3062–9.
Dai GF, Cheng N, Dong L, Muramatsu M, Xiao SD, Wang MW, et al. Bactericidal and morphological effects of NE-2001, a novel synthetic agent directed against Helicobacter pylori. Antimicrob Agents Chemother 2005; 49: 3468–73.
Zhu DX, Muramatsu M, Xie JS, Cheng N, Wang MW . Methods and compositions for treating or preventing bacterial infection. US Patent 6 734 212. 2004; May 11.
Yamashiro D, Blake J, Li CH . Use of N. alpha., Nim-bis(tert-butyloxycarbonyl)histidine and N.alpha.-2-(p-biphenylyl) isopropyloxycarbonyl-Nim-tert-butyloxycarbonylhistidine in the solid-phase synthesis of histidine-containing peptides. J Am Chem Soc 1972; 94: 2855–9.
Johnstone RW, Wilby AH, Entwistle ID . Heterogeneous catalytic transfer hydrogenation and its relation to other methods for reduction of organic compounds. Chem Rev 1985; 85: 129–70.
Pews RG, Tsuno Y, Taft RW . A fluorine nuclear magnetic resonance shielding study of substituent effects on charge distributions in benzophenone and its Lewis acid adducts. J Am Chem Soc 1967; 89: 2391–6.
Kubo K, Ohyama SI, Shimizu T, Takami A, Murooka H, Nishitoba T, et al. Synthesis and structure-activity relationship for new series of 4-phenoxyquinoline derivatives as specific inhibitors of platelet-derived growth factor receptor tyrosine kinase. Bioorg Med Chem 2003; 11: 5117–33.
Lemenovskii DA, Makarov MV, Dyadchenko VP, Bruce AE, Bruce MR, Larkin SA, et al. Syntheses and crystal structures of ferrocenyl derivatives of biphenyl. Rus Chem Bull 2003; 52: 607–15.
Belcher R, Nutten AJ, Stephen WI . Substituted benzidines and related compounds as reagents in analytical chemistry. Part XII. Reagents for the precipitation of sulphate. J Chem Soc 1953; 1334–8.
Roe A, Fleishman HL . The synthesis of some fluorohydroxy-biphenyls. J Am Chem Soc 1947; 69: 509–10.
Laeckmann D, Rogister F, Dejardin JV, Christelle PM, Géczy J, Delarge J, et al. Synthesis and biological evaluation of aroylguanidines related to amiloride as inhibitors of the human platelet Na+/H+ exchanger. Bioorg Med Chem 2002; 10: 1793–804.
Scott FL, O'Donovan DG, Reilly J . Studies in the pyrazole series III. Substituted guanidines. J Am Chem Soc 1953; 75: 4053–4.
Miller AE, Bischoff JJ . A facile conversion of amino acids to guanidine acids. Synthesis 1986; 9: 777–9.
National Committee for Clinical Laboratory Standards. Performance standards for antimicrobial susceptibility testing and approved standard M7-A6. Informational supplement M100-S15 (2005). National Committee for Clinical Laboratory Standards: Wayne, PA, USA; 2003.
Yan JB, Wang GQ, Du P, Zhu DX, Wang MW, Jiang XY . High-level expression and purification of Escherichia coli oligopeptidase B. Protein Expr Purif 2006; 47: 645–50.
Nikaido H . Molecular basis of bacterial outer membrane permeability revisited. Microbiol Mol Biol Rev 2003; 67: 593–656.
Fritsche TR, Stephen JS, Jones RN . Changing patterns of bacterial pathogens and their antimicrobial susceptibility profiles in patients hospitalized with pneumonia. Report from the SENTRY Antimicrobial Surveillance Program, 2002. Program and abstracts of the 43rd Annual ICAAC; Sep 14-17 2003, Illinois, Chicago, USA; Abstract C-2191.
Ueda H, Ikeda T, Kakegawa H, Miyataka H, Matsumoto H, Satoh T . Syntheses and inhibitory effects on gastric lesions of 4-guanidinomethylbenzoic acid arylamides. Chem Pharm Bull 1993; 41: 1387–90.
Seedat J, Zick G, Klare I, Konstabel C, Weiler N, Sahly H . Rapid emergence of resistance to linezolid during linezolid therapy of an Enterococcus faecium infection. Antimicrob Agents Chemother 2006; 50: 4217–9.
Rybak MJ, Cappelletty DM, Moldovan T, Aeschlimann JR, Kaatz GW . Comparative in vitro activities and postantibiotic effects of the oxazolidinone compounds eperezolid (PNU-100592) and linezolid (PNU-100766) versus vancomycin against staphylococcus aureus, coagulase-negative staphylococci, Enterococcus faecalis, and Enterococcus faecium. Antimicrob Agents Chemother 1998; 42: 721–4.
Howe RA, Wootton M, Noel AR, Bowker KE, Walsh TR, MacGowan AP . Activity of AZD2563, a novel oxazolidinone, against staphylococcus aureus strains with reduced susceptibility to vancomycin or linezolid. Antimicrob Agents Chemother 2003; 47: 3651–2.
Tanaka M, Yamazaki E, Chiba M, Yoshihara K, Akasaka T, Takemura M . In vitro antibacterial activities of DQ-113, a potent quinolone, against clinical isolates. Antimicrob Agents Chemother 2002; 46: 904–8.
Bogdanovich T, Esel D, Kelly LM, Bozdogan B, Credito K, Lin G, et al. Antistaphylococcal activity of DX-619, a new des-F(6)-quinolone, compared to those of other agents. Antimicrob Agents Chemother 2005; 49: 3325–33.
Park HS, Kim HJ, Seol MJ, Choi DR, Choi EC, Kwak JH . In vitro and in vivo antibacterial activities of DW-224a, a new fluoronaphthyridone. Antimicrob Agents Chemother 2006; 50: 2261–4.
Novak P, Ray PH, Dev IK . Localization and purification of two enzymes from Escherichia coli capable of hydrolyzing a signal peptide. J Biol Chem 1986; 261: 420–7.
Conlin CA, Vimr ER, Miller CG . Escherichia coli prlC encodes an endopeptidase and is homologous to the Salmonella typhimurium opdA gene. J Bacteriol 1992; 174: 5869–87.
Novak P, Dev IK . Degradation of a signal peptide by protease IV and oligopeptidase A. J Bacteriol 1988; 170: 5067–75.
Conlin CA, Miller CG . Dipeptidyl carboxypeptidase and oligopeptidase A from Escherichia coli and Salmonella typhimurium. Methods Enzymol 1995; 248: 567–79.
Morty RE, Fulop V, Andrews NW . Substrate recognition properties of oligopeptidase B from Salmonella enterica serovar typhimurium. J Bacteriol 2002; 184: 3329–37.
Tsuru D, Yoshimoto T . Oligopeptidase B: protease II from Escherichia coli. Methods Enzymol 1994; 244: 201–15.
Tsuji A, Yuasa K, Matsuda Y . Identification of oligopeptidase B in higher plants. Purification and characterization of oligopeptidase B from quiescent wheat embryo, triticum aestivum. J Biochem 2004; 136: 673–81.
Tsuji A, Yoshimoto T, Yuaask K, Matsuda Y . Protamine: a unique and potent inhibitor of oligopeptidase B. J Peptide Sci 2006; 12: 65–71.
Tünde J, Zoltán S, Veronika R, László P . Role of the oxyanion binding site and subsites S1 and S2 in the catalysis of oligopeptidase B, a novel target for antimicrobial chemotherapy. Biochemistry 2002; 41: 4096–106.
Morty RE, Authíe E, Troeberg L, Lonsdale-Eccles JD, Coetzer THT . Purification and characterization of a trypsin-like serine oligopeptidase from Trypanosoma congolense. Mol Biochem Parasitol 1999; 102: 145–55.
Morty RE, Lonsdale-Eccles JD, Mentele R, Auerswald EA, Coetzer THT . Trypanosoma-derived oligopeptidase B is released into the plasma of infected rodents, where its persists and retains full catalytic activity. Infect Immun 2001; 69: 2757–61.
Morty RE, Troeberg L, Pike RN, Jones R, Nickel P, Lonsdale-Eccles JD, et al. A trypanosome oligopeptidase as a target for the trypanocidal agents pentamidine, diminazone and suramin. FEBS Lett 1998; 433: 251–6.
Author information
Authors and Affiliations
Corresponding author
Additional information
Project supported in part by grants from the Ministry of Science and Technology of China (No 2004CB518902) and Shanghai Municipality Government (No 30219228 and 05dz22914).
Rights and permissions
About this article
Cite this article
Yu, Wy., Yang, Lx., Xie, Js. et al. Derivatives of aryl-4-guanidinomethylbenzoate and N-aryl-4-guanidinomethylbenzamide as new antibacterial agents: synthesis and bioactivity. Acta Pharmacol Sin 29, 267–277 (2008). https://doi.org/10.1111/j.1745-7254.2008.00720.x
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1111/j.1745-7254.2008.00720.x