Abstract
Aim:
The present study aimed to explore the protective effect of endogenous sulfur dioxide (SO2) in the development of monocrotaline (MCT)-induced pulmonary hypertension (PH) in rats.
Methods:
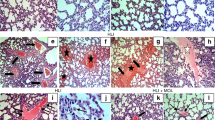
Forty Wistar rats were randomly divided into the MCT group receiving MCT treatment, the MCT+L-aspartate-β-hydroxamate (HDX) group receiving MCT plus HDX treatment, the MCT+SO2 group receiving MCT plus SO2 donor treatment, and the control group. Mean pulmonary artery pressure (mPAP) and structural changes in pulmonary arteries were evaluated. SO2 content, aspartate aminotransferase activity, and gene expression were measured. Superoxide dismutase (SOD), glutathione peroxidase (GSH-Px), catalase (CAT), reduced glutathione (GSH), oxidized glutathione, and malondialdehyde (MDA) levels were assayed.
Results:
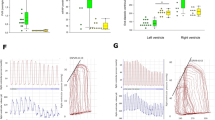
In the MCT-treated rats, mPAP and right ventricle/(left ventricle+septum) increased significantly (P<0.01), pulmonary vascular structural remodeling developed, and SOD, GSH-Px, CAT, GSH, and MDA levels of lung homogenates significantly increased (P<0.01) in association with the elevated SO2 content, aspartate aminotransferase activity, and gene expression, compared with the control rats. In the MCT+HDX-treated rats, lung tissues and plasma SO2 content and aspartate aminotransferase activities decreased significantly, whereas the mPAP and pulmonary vascular structural remodeling were markedly aggravated with the decreased SOD, CAT, and GSH levels of lung tissue homogenates compared with the MCT-treated rats (P<0.01). In contrast, with the use of a SO2 donor, the pulmonary vascular structural remodeling was obviously lessened with elevated lung tissue SOD, GSH-Px, and MDA content, and plasma SOD, GSH-Px, and CAT levels.
Conclusion:
Endogenous SO2 might play a protective role in the pathogenesis of MCT-induced PH and promote endogenous antioxidative capacities.
Similar content being viewed by others
Article PDF
References
McLaughlin VV, McGoon MD . Primary pulmonary hypertension. Circulation 2006; 114: 1417–31.
Hoeper MM, Rubin LJ . Update in pulmonary hypertension 2005. Am J Respir Crit Care Med 2006; 173: 499–505.
Granton JT, Rabinovitch M . Pulmonary arterial hypertension in congenital heart disease. Cardiol Clin 2002; 20: 441–57.
Tuder RM, Groves B, Badesch DB, Voelkel NF . Exuberant endothelial cell growth and elements of inflammation are present in plexiform lesions of pulmonary hypertension. Am J Pathol 1994; 144: 275–85.
Wu LY, Wang R . Carbon monoxide: endogenous production, physiological functions, and pharmacological applications. Pharmacol Rev 2005; 57: 585–630.
Yanagisawa M, Kurihara H, Kimura S, Tomobe Y, Kobayashi M, Mitsui Y, et al. A novel potent vasoconstrictor peptide produced by vascular endothelial cells. Nature 1988; 332: 411–5.
Shimokawa H, Tomoike H, Nabeyama S, Yamamoto H, Araki H, Nakamura M, et al. Coronary artery spasm induced in atherosclerotic miniature swine. Science 1983; 221: 560–2.
Vroomen M, Takahashi Y, Gournay V, Roman C, Rudolph AM, Heymann MA . Adrenomedulin increases pulmonary blood flow in fetal sheep. Pediatr Res 1997; 41: 493–7.
Du JB, Jia JF, Li WZ, Zhao B, Zeng HP . Nitric oxide impacts endothelin-1 gene expression in intrapulmonary arteries of chronically hypoxic rats. Angiology 1999; 50: 479–85.
Shi Y, Du JB, Gong LM, Zeng CM, Tang XY, Tang CS . The regulating effect of heme oxygenase/carbon monoxide on hypoxic pulmonary vascular structural remodeling. Biochem Biophys Res Commun 2003; 306: 523–9.
Li XH, Du JB, Shi L, Li J, Tang XY, Qi JG, et al. Down-regulation of endogenous hydrogen sulfide pathway in pulmonary hypertension and pulmonary vascular structural remodeling induced by high pulmonary blood flow in rats. Circ J 2005; 69: 1418–24.
Ignarro LJ . Biological actions and properties of endothelium-derived nitric oxide formed and released from artery and vein. Circ Res 1989; 65: 1–2.
Garg UC, Hassid A . Nitric oxide-generating vasodilators and 8-bro-mo-cyclic guanosine monophosphate inhibit mitogenesis and proliferation of cultured rat vascular smooth cells. J Clin Invest 1989; 83: 1774–7.
Zhen G, Zhang Z, Xu Y . The role of endogenous carbon monoxide in the hypoxic vascular remodeling of rat model of hypoxic pulmonary hypertension. J Huazhong Univ Sci Tech Med Sci 2003; 23: 356–68.
Tang CS, Li XH, Du JB . Hydrogen sulfide as a new endogenous gaseous transmitter in the cardiovascular system. Curr Vasc Pharmacol 2006; 4: 17–22.
Zhang CY, Du JB, Bu DF, Yan H, Tang XY, Tang CS . The regulatory effect of hydrogen sulfide on hypoxic pulmonary hypertension in rats. Biochem Biophys Res Commun 2003; 302: 810–6.
Meng ZQ, Zhang H . The vasodilator effect and its mechanism of sulfur dioxide-derivatives on isolated aortic rings of rats. Inhal Toxicol 2007; 19: 979–86.
Meng ZQ, Geng HF, Bai JL, Yan G . Blood pressure of rats lowered by sulfur dioxide and its derivatives. Inhal Toxicol 2003; 15: 951–9.
Mitsuhashi H, Ota F, Ikeuchi K, Kaneko Y, Kuroiwa T, Ueki K, et al. Sulfite is generated from PAPS by activated neutrophils. Tohoku J Exp Med 2002; 198: 125–32.
Shigehara T, Mitsuhashi H, Ota F, Kuroiwa T, Kaneko Y, Ueki K, et al. Sulfite induces adherence of polymorphonuclear neutrophils to immobilized fibrinogen through activation of Mac-1 beta2-integrin (CD11b/CD18). Life Sci 2002; 70: 2225–32.
Huxtable RJ . Activation and pulmonary toxicity of pyrrolizidine alkaloids. Pharmacol Ther 1990; 47: 371–89.
Rosenberg HC, Rabinovitch M . Endothelial injury and vascular reactivity in monocrotaline pulmonary hypertension. Am J Physiol 1988; 255: H1484–91.
Aziz SM, Toborek M, Hennig B, Endean E, Lipke DW . Polyamine regulatory processes and oxidative stress in monocrotaline-treated pulmonary artery endothelial cells. Cell Biol Int 1997; 21: 801–12.
Kamezaki F, Tasaki H, Yamashita K, Tsutsui M, Koide S, Nakata S, et al. Gene transfer of extracellular superoxide dismutase ameliorates pulmonary hypertension in rats. Am J Respir Crit Care Med 2008; 177: 219–26.
Meng Z, Qin G, Zhang B, Geng H, Bai Q, Bai W, et al. Oxidative damage of sulfur dioxide inhalation on lungs and hearts of mice. Environ Res 2003; 93: 285–92.
Barth PJ, Kimpel CH, Roy S, Wagner U . An improved mathematical approach for the assessment of the medial thickness of pulmonary arteries. Pathol Res Pract 1993; 189: 567–76.
Mitsuhashi H, Ikeuchi H, Yamashita S, Kuroiwa T, Kaneko Y, Hiromura K, et al. Increased levels of serum sulfite in patients with acute pneumonia. Shock 2004; 21: 99–102.
Ji AJ, Savon SR, Jacobsen DW . Determination of total serum sulfite by HPLC with fluorescence detection. Clin Chem 1995; 41: 897–903.
Stipanuk MH . Metabolism of sulfur containing amino acids. Annu Rev Nutr 1986; 6: 179–209.
Griffith OW . Cysteinesulfinate metabolism altered partitioning between transamination and decarboxylation following administration of beta-methylene aspartate. J Biol Chem 1983; 258: 1591–8.
Hironaka E, Hongo M, Sakai A, Mawatari E, Terasawa F, Okumura N, et al. Serotonin receptor antagonist inhibits monocrotaline-induced pulmonary hypertension and prolongs survival in rats. Cardiovasc Res 2003; 60: 692–9.
Versluis JP, Heslinga JW, Sipkema P, Westerhof N . Contractile reserve but not tension is reduced in monocrotaline-induced right ventricular hypertrophy. Am J Physiol Heart Circ Physiol 2004; 286: H979–87.
Ecarnot-Laubriet A, Rochette L, Vergely C, Sicard P, Teyssier JR . The activation pattern of the antioxidant enzymes in the right ventricle of rat in response to pressure overload is of heart failure type. Heart Dis 2003; 5: 308–12.
Farahmand F, Hill MF, Singal PK . Antioxidant and oxidative stress changes in experimental cor pulmonale. Mol Cell Biochem 2004; 260: 21–9.
Ceballos-Picot I, Nicole A, Clément M, Bourre JM, Sinet PM . Age-related changes in antioxidant enzymes and lipid peroxidation in brains of control and transgenic mice overexpressing copper-zinc superoxide dismutase. Mutat Res 1992; 275: 281–93.
Gupta A, Hasan M, Chander R, Kapoor NK . Age-related elevation of lipid peroxidation products: diminution of superoxide dismutase activity in the central nervous system of rats. Gerontology 1991; 37: 305–9.
Singh R, Pathak DN . Lipid peroxidation and glutathione peroxidase, glutathione reductase, superoxide dismutase, catalase, and glucose-6-phosphate dehydrogenase activities in FeCl3-induced epileptogenic foci in the rat brain. Epilepsia 1990; 31: 15–26.
Yargicoglu P, Sahin E, Gümüşlü S, Aǧar A . The effect of sulfur dioxide inhalation on active avoidance learning, antioxidant status and lipid peroxidation during aging. Neurotoxicol Teratol 2007; 29: 211–8.
Meng Z, Liu Y, Wu D . Effect of sulfur dioxide inhalation on cytokine levels in lungs and serum of mice. Inhal Toxicol 2005; 17: 303–7.
Meng Z . Oxidative damage of sulfur dioxide on various organs of mice: sulfur dioxide is a systemic oxidative damage agent. Inhal Toxicol 2003; 15: 181–95.
Meng Z, Bai W . Oxidation damage of sulfur dioxide on testicles of mice. Environ Res 2004; 96: 298–304.
Xie J, Fan R, Meng Z . Protein oxidation and DNA-protein crosslink induced by sulfur dioxide in lungs, livers, and hearts from mice. Inhal Toxicol 2007; 19: 759–65.
Author information
Authors and Affiliations
Corresponding authors
Additional information
This work was supported by the Major Basic Research Program of China (No 2006CB503807), the National Natural Science Foundation of China (No 30630031), the Beijing Natural Science Foundation (No 7072082 and 7082095), the Research Fund for the Doctoral Program of Ministry of Education of China (20070001702, and the Cheung Kong Scholars Program (No 985-2-087–111).
Rights and permissions
About this article
Cite this article
Jin, Hf., Du, Sx., Zhao, X. et al. Effects of endogenous sulfur dioxide on monocrotaline-induced pulmonary hypertension in rats. Acta Pharmacol Sin 29, 1157–1166 (2008). https://doi.org/10.1111/j.1745-7254.2008.00864.x
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1111/j.1745-7254.2008.00864.x
Keywords
This article is cited by
-
Sulfur dioxide acts as an antioxidant and delays programmed cell death in wheat aleurone layers upstream of H2S and NO signaling pathways
Biologia Plantarum (2018)
-
The PI3K/Akt, p38MAPK, and JAK2/STAT3 signaling pathways mediate the protection of SO2 against acute lung injury induced by limb ischemia/reperfusion in rats
The Journal of Physiological Sciences (2016)
-
Endogeous sulfur dioxide protects against oleic acid-induced acute lung injury in association with inhibition of oxidative stress in rats
Laboratory Investigation (2015)
-
Sulfur dioxide promotes germination and plays an antioxidant role in cadmium-stressed wheat seeds
Plant Growth Regulation (2015)
-
Sulfur dioxide attenuates LPS-induced acute lung injury via enhancing polymorphonuclear neutrophil apoptosis
Acta Pharmacologica Sinica (2012)