Abstract
Aim:
Spermatogonial stem cells can initiate the process of cellular differentiation to generate mature spermatozoa, but whether it possess the characteristic of pluripotency and plasticity, similar to embryonic stem cells, has not been elucidated. This study was designed to evaluate the differentiation potential of spermatogonial stem cells into renal cells in vivo.
Methods:
Neonatal mouse spermatogonial stem cells were transplanted into mature male mice lacking endogenous spermatogenesis. The restoration of fertility in recipient males was observed. Spermatogonial stem cells were then injected into renal parenchyma of mature female mice to make a new extracellular environment for differentiation. Fluorescence in situ hybridization technology (FISH) was used to detect the expression of chromosome Y in recipient renal tissues. To determine the type of cells differentiated from spermatogonial stem cells, the expression of ricinus communis agglutinin, vimentin, CD45, and F4/80 proteins were examined in the renal tissues by immunohistochemistry.
Results:
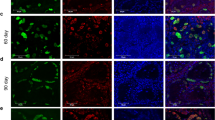
The proliferation of seminiferous epithelial cells was distinctly observed in seminiferous tubules of transplanted testes, whereas no regeneration of spermatogenesis was observed in non-transplanted control testes. In transplanted female renal tissues, FISH showed a much stronger immuno-fluorescence signal of chromosome Y in the nucleolus of epithelial cells of the renal tubule and podocytes of the glomerulus.
Conclusion:
The spermatogonial stem cells were successfully purified from mouse testicles. This finding demonstrated that spermatogonial stem cells could not only restore damaged spermatogenesis, but were also capable of differentiating into mature renal parenchyma cells in vivo.
Similar content being viewed by others
Article PDF
References
Watt FM, Hogan BL . Out of Eden: stem cells and their niches. Science 2000; 287: 1427–30.
Morrison SJ, Shah NM, Anderson DJ . Regulatory mechanisms in stem cell biology. Cell 1997; 88: 287–98.
Gage FH, Ray J, Fisher LJ . Isolation, characterization, and use of stem cells from the CNS. Annu Rev Neurosci 1995; 18: 159–92.
Zhao GQ, Garbers DL . Male germ cell specification and differentiation. Dev Cell 2002; 2: 537–47.
Reya T, Morrison SJ, Clarke MF, Weissman IL . Stem cells, cancer, and cancer stem cells. Nature 2001; 414: 105–11.
Donovan PJ, Stott D, Cairns LA, Heasman J, Wylie CC . Migratory and postmigratory mouse primordial germ cells behave differently in culture. Cell 1986; 44: 831–8.
Rochette-Egly C, Chambon P . F9 embryo carcinoma cells: a cell autonomous model to study the functional selectivity of RARs and RXRs in retinoid signaling. Histol Histopathol 2001; 16: 909–22.
Lehtonen E, Laasonen A, Tienari J . Teratocarcinoma stem cells as a model for differentiation in the mouse embryo. Int J Dev Biol 1989; 33: 105–15.
McLaren A, Durcova-Hills G . Germ cells and pluripotent stem cells in the mouse. Reprod Fertil Dev 2001; 13: 661–4.
Beddington RS, Robertson EJ . An assessment of the developmental potential of embryonic stem cells in the midgestation mouse embryo. Development 1989; 105: 733–7.
Spradling A, Drummond-Barbosa D, Kai T . Stem cells find their niche. Nature 2001; 414: 98–104.
Guan K, Nayernia K, Maier LS, Wagner S, Dressel R, Lee JH, et al. Pluripotency of spermatogonial stem cells from adult mouse testis. Nature 2006; 440: 1199–203.
Izadyar F, Spierenberg GT, Creemers LB, den Ouden K, de Rooij DG . Isolation and purification of type A spermatogonia from the bovine testis. Reproduction 2002; 124: 85–94.
Boettger-Tong HL, Johnston DS, Russell LD, Griswold MD, Bishop CE . Juvenile spermatogonial depletion (jsd) mutant seminiferous tubules are capable of supporting transplanted spermatogenesis. Biol Reprod 2000; 63: 1185–91.
Gordeeva O, Zinovieva R, Smirnova Y, Payushina O, Nikonova T, Khrushchov N . Differentiation of embryonic stem cells after transplantation into peritoneal cavity of irradiated mice and expression of specific germ cell genes in pluripotent cells. Transplant Proc 2005; 37: 295–8.
McLaren A . Germ and somatic cell lineages in the developing gonad. Mol Cell Endocrinol 2000; 163: 3–9.
de Rooij DG, Grootegoed JA . Spermatogonial stem cells. Curr Opin Cell Biol 1998; 10: 694–701.
Eglitis MA, Mezey E . Hematopoietic cells differentiate into both mi-croglia and macroglia in the brains of adult mice. Proc Natl Acad Sci USA 1997; 94: 4080–5.
Gussoni E, Soneoka Y, Strickland CD, Buzney EA, Khan MK, Flint AF, et al. Dystrophin expression in the mdx mouse restored by stem cell transplantation. Nature 1999; 401: 390–4.
Pittenger MF, Mackay AM, Beck SC, Jaiswal RK, Douglas R, Mosca JD, et al. Multilineage potential of adult human mesenchymal stem cells. Science 1999; 284: 143–7.
Nakano K, Migita M, Mochizuki H, Shimada T . Differentiation of transplanted bone marrow cells in the adult mouse brain. Transplantation 2001; 71: 1735–40.
Jackson KA, Mi T, Goodell MA . Hematopoietic potential of stem cells isolated from murine skeletal muscle. Proc Natl Acad Sci USA 1999; 96: 14482–6.
Bjornson CR, Rietze RL, Reynolds BA, Magli MC, Vescovi AL . Turning brain into blood: a hematopoietic fate adopted by adult neural stem cells in vivo. Science 1999; 283: 534–7.
Poulsom R, Forbes SJ, Hodivala-Dilke K, Ryan E, Wyles S, Navarat-narasah S, et al. Bone marrow contributes to renal parenchymal turnover and regeneration. J Pathol 2001; 195: 229–35.
Guan K, Wagner S, Unsöld B, Maier LS, Kaiser D, Hemmerlein B, et al. Generation of functional cardiomyocytes from adult mouse spermatogonial stem cells. Circ Res 2007; 100: 1615–25.
Kanatsu-Shinohara M, Lee J, Inoue K, Ogonuki N, Miki H, Toyokuni S, et al. Pluripotency of a single spermatogonial stem cell in mice. Biol Reprod 2008; 78: 681–7.
Author information
Authors and Affiliations
Corresponding author
Additional information
Project supported by the National Natural Science Foundation of China (No 30200132)
Rights and permissions
About this article
Cite this article
Wu, Dp., He, Dl., Li, X. et al. Differentiations of transplanted mouse spermatogonial stem cells in the adult mouse renal parenchyma in vivo. Acta Pharmacol Sin 29, 1029–1034 (2008). https://doi.org/10.1111/j.1745-7254.2008.00836.x
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1111/j.1745-7254.2008.00836.x