Abstract
Aim:
To elucidate the role of scaffold protein postsynaptic density (PSD)-95 in the dopamine D1 receptor (D1R)-modulated NR1a/NR2B receptor response.
Methods:
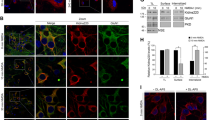
The human embryonic kidney 293 cells expressing D1R (tagged with the enhanced yellow fluorescent protein) and NR1a/NR2B with or without co-expression of PSD-95 were used in the experiments. The Ca2+ influx measured by imaging technique was employed to monitor N -methyl-D-aspartic acid receptors (NMDAR) function.
Results:
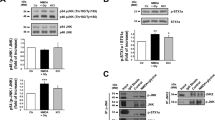
The application of dopamine (DA, 100 μmol/L) did not alter glutamate/glycine (Glu/Gly)-induced NMDAR-mediated Ca2+ influx in cells only expressing the D1R/NR1a/NR2B receptor. However, DA increased Glu/Gly-induced Ca2+ influx in a concentration-dependent manner while the cells were co-expressed with PSD-95. D1R-stimulated Ca2+ influx was inhibited by a selective D1R antagonist SCH23390. Moreover, pre-incubation with either the protein kinase A (PKA) inhibitor H89, or the protein kinase C (PKC) inhibitor chelerythrine attenuated D1R-enhanced Ca2+ influx induced by the N-methyl-D-aspartic acid (NMDA) agonist. The results clearly indicate that D1R-modulated NR1a/NR2B receptor function depends on PSD-95 and is subjected to the regulation of PKA and PKC.
Conclusion:
The present study provides the first evidence that PSD-95 is essential in D1R-regulated NR1a/NR2B receptor function.
Similar content being viewed by others
Article PDF
References
Dingledine R, Borges K, Bowie D, Traynelis SF . The glutamate receptor ion channels. Pharmacol Rev, 1999; 51: 7–61.
Cull-Candy S, Brickley S, Farrant M . NMDA receptor subunits: diversity, development and disease. Curr Opin Neurobiol, 2001; 11: 327–35.
Gracy KN, Pickel VM . Ultrastructural immunocytochemical localization of the N-methyl-D-aspartate receptor and tyrosine hydroxylase in the shell of the rat nucleus accumbens. Brain Res, 1996; 739: 169–81.
Cepeda C, Levine MS . Dopamine and N-methyl-D-aspartate receptor interactions in the neostriatum. Dev Neurosci, 1998; 20: 1–18.
Sesack SR, Carr DB, Omelchenko N, Pinto A . Anatomical substrates for glutamate-dopamine interactions: evidence for specificity of connections and extrasynaptic actions. Ann N Y Acad Sci, 2003; 1003: 36–52.
Fiorentini C, Gardoni F, Spano P, Di Luca M, Missale C . Regulation of dopamine D1 receptor trafficking and desensitization by oligomerization with glutamate N-methyl-D-aspartate receptors. J Biol Chem, 2003; 278: 20196–202.
Lee FJ, Xue S, Pei L, Vukusic B, Chery N, Wang Y, et al. Dual regulation of NMDA receptor functions by direct protein-protein interactions with the dopamine D1 receptor. Cell, 2002; 111: 219–30.
Snyder GL, Fienberg AA, Huganir RL, Greengard P . A dopamine/ D1 receptor/protein kinase A/dopamine- and cAMP-regulated phosphoprotein (Mr 32 kDa)/protein phosphatase-1 pathway regulates dephosphorylation of the NMDA receptor. J Neurosci, 1988; 18: 10297–303.
Wang J, O'Donnell P . D(1) dopamine receptors potentiate NMDA-mediated excitability increase in layer V prefrontal cortical pyramidal neurons. Cereb Cortex, 2001; 11: 452–62.
Chen G, Greengard P, Yan Z . Potentiation of NMDA receptor currents by dopamine D1 receptors in prefrontal cortex. Proc Natl Acad Sci USA, 2004; 101: 2596–600.
Kennedy MB . Signal-processing machines at the postsynaptic density. Science, 2000; 290: 750–4.
McGee AW, Dakoji SR, Olsen O, Bredt DS, Lim WA, Prehoda KE . Structure of the SH3-guanylate kinase module from PSD-95 suggests a mechanism for regulated assembly of MAGUK scaffolding proteins. Mol Cell, 2001; 8: 1291–301.
Niethammer M, Kim E, Sheng M . Interaction between the C terminus of NMDA receptor subunits and multiple members of the PSD–95 family of membrane-associated guanylate kinases. J Neurosci, 1996; 16: 2157–63.
Xie Z, Gray JA, Compton-Toth BA, Roth BL . A direct interaction of PSD-95 with 5-HT2A serotonin receptors regulates receptor trafficking and signal transduction. J Biol Chem, 2003; 278: 21901–8.
Swayze RD, Lise MF, Levinson JN, Phillips A, El-Husseini A . Modulation of dopamine mediated phosphorylation of AMPA receptors by PSD-95 and AKAP79/150. Neuropharm, 2004; 47: 764–78.
Barton AC, Sibley DR . Agonist-induced desensitization of D1-dopamine receptors linked to adenylyl cyclase activity in cultured NS20Y neuroblastoma cells. Mol Pharmacol, 1990; 38: 531–41.
Westphal RS, Tavalin SJ, Lin JW, Alto NM, Fraser ID, Langeberg LK, et al. Regulation of NMDA receptors by an associated phosphatase-kinase signaling complex. Science, 1999; 285: 93–6.
Posas F, Saito H . Osmotic activation of the HOG MAPK pathway via Ste11p MAPKKK: scaffold role of Pbs2p MAPKK. Science, 1997; 276: 1702–5.
Skeberdis VA, Chevaleyre V, Lau CG, Goldberg JH, Pettit DL, Suadicani SO, et al. Protein kinase A regulates calcium permeability of NMDA receptors. Nat Neurosci, 2006; 9: 501–10.
Author information
Authors and Affiliations
Corresponding authors
Additional information
Project supported by the National Natural Science Foundation of China (No 30572168, 30672448) and the National 973 plan (No 2003CB5154000).
Rights and permissions
About this article
Cite this article
Gu, Wh., Yang, S., Shi, Wx. et al. Requirement of PSD-95 for dopamine D1 receptor modulating glutamate NR1a/NR2B receptor function. Acta Pharmacol Sin 28, 756–762 (2007). https://doi.org/10.1111/j.1745-7254.2007.00557.x
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1111/j.1745-7254.2007.00557.x