Abstract
Aim:
To characterize the molecular mechanisms of nitrofen-induced pulmonary hypoplasia.
Methods:
After administration of nitrofen to cultured type IIA549 pneumocytes, cell proliferation and DNA synthesis were investigated by 3- (4, 5-dimethylthiazol-2-yl)-2, 5-diphenyl tetrazolium bromide colorimetry, colony formation assay, flow cytometry and [3H]-thymidine incorporation assay. Apoptosis was measured by terminal transferase-mediated dUTP nick-end-labeling, acridine orange-ethidium bromide staining and flow cytometry. Expression of proliferating cell nuclear antigen (PCNA) and apoptosis-related genes was assayed by immunofluorescence, RT-PCR and Western blot.
Results:
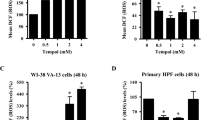
Nitrofen inhibited the cell proliferation of A549 cells in a dose- and time-dependent manner, accompanied by downregulation of PCNA. As a result, the DNA synthesis of nitrofentreated A549 cells decreased, while cell cycle was arrested at G0/G1 phase. Moreover, nitrofen induced apoptosis of A549 cells, which was not abolished by Z-Val-Ala-Asp(OCH3)- fluoromethylketone. In addition, nitrofen decreased the expression of Bcl-xL, but not of Bcl-2, Bax, and Bak, resulting in a loss of mitochondrial membrane potential and the nuclear translocation of apoptosis-inducing factor (AIF). Meanwhile, nitrofen strongly activated the p38 mitogen-activated protein kinase (p38-MAPK). Pretreatment of cells with SB203580 (5 μmol/L) blocked nitrofen-induced phosphorylation of p38-MAPK and abolished nitrofen-induced AIF translocation and apoptosis in A549 cells.
Conclusion:
Nitrofen suppresses the proliferation of cultured type II pneumocytes accompanied by the downregulation of PCNA, and induces mitochondria-mediated apoptosis involving the activation of p38-MAPK.
Similar content being viewed by others
Article PDF
References
Hurt SS, Smith JM, Hayes AW . Nitrofen: a review and perspective. Toxicology 1983; 29: 1–37.
Manson JM . Mechanism of nitrofen teratogenesis. Environ Health Perspect 1986; 70: 137–47.
Islam S, Narra V, Cote GM, Manganaro TF, Donahoe PK, Schnitzer JJ . Prenatal vitamin E treatment improves lung growth in fetal rats with congenital diaphragmatic hernia. J Pediatr Surg 1999; 34: 172–6.
Alfonso LF, Vilanova J, Aldazabal P, Lopez de Torre B, Tovar JA . Lung growth and maturation in the rat model of experimentally induced congenital diaphragmatic hernia. Eur J Pediatr Surg 1993; 3: 6–11.
Suen HC, Catlin EA, Ryan DP, Wain JC, Donahoe PK . Biochemical immaturity of lungs in congenital diaphragmatic hernia. J Pediatr Surg 1993; 28: 471–7.
Alfanso LF, Arnaiz A, Alvarez FJ, Qi B, Diez-Pardo JA, Vallis-i-Soler A, et al. Lung hypoplasia and surfactant system immaturity induced in the fetal rat by prenatal exposure to nitrofen. Biol Neonate 1996; 69: 94–100.
Coleman C, Zhao J, Gupta M, Buckley S, Tefft JD, Wuenschell CW, et al. Inhibition of vascular and epithelial differentiation in murine nitrofen-induced diaphragmatic hernia. Am J Physiol Lung Cell Mol Physiol 1998; 274: L636–46.
Cilley RE, Zgleszewski SE, Krummel TM, Chinoy MR . Nitrofen dose-dependent gestational day-specific murine lung hypoplasia and left-sided diaphragmatic hernia. Am J Physiol Lung Cell Mol Physiol 1997; 272: L362–71.
Chen MH, MacGowan A, Ward S, Bavik C, Greer JJ . The activation of the retinoic acid response element is inhibited in an animal model of congenital diaphragmatic hernia. Biol Neonate 2003; 83: 157–61.
Mason RJ . Biology of alveolar type II cells. Respirology 2006; 11: S12–5.
Li J, Hu T, Liu W, Xiang B, Jiang X . Effect of epidermal growth factor on pulmonary hypoplasia in experimental diaphragmatic hernia. J Pediatr Surg 2004; 39: 37–42.
Gonzalez-Reyes S, Martinez L, Tovar JA . Vitamin C rescues in part the effects of nitrofen on cultured human pneumocytes. Pediatr Surg Int 2004; 20: 258–62.
Nardone LL, Andrews SB . Cell line A549 as a model of the type II pneumocyte. Phospholipid biosynthesis from native and or-ganometallic precursors. Biochim Biophys Acta 1979; 573: 276–95.
Mosmann T . Rapid colorimetric assay for cellular growth and survival: application to proliferation and cytotoxicity assays. J Immunol Methods 1983; 65: 55–63.
Sheffield LG . Effect of sialoadenectomy on the ability of mouse serum to induce deoxyribonucleic acid synthesis in mammary epithelial cells: possible role of epidermal growth factor. J Dairy Sci 1990; 73: 2087–92.
Lebrin F, Goumans MJ, Jonker L, Carvalho RL, Valdimarsdottir G, Thorikay M, et al. Endoglin promotes endothelial cell proliferation and TGF-beta/ALK1 signal transduction. EMBO J 2004; 23: 4018–8.
Overbeeke R, Steffens-Nakken H, Vermes I, Reutelingsperger C, Haanen C . Early features of apoptosis detected by four different flow cytometry assays. Apoptosis 1998; 3: 115–21.
Leite M, Quinta-Costa M, Leite PS, Guimaraes JE . Critical evaluation of techniques to detect and measure cell death — study in a model of UV radiation of the leukaemic cell line HL60. Anal Cell Pathol 1999; 19: 139–51.
Pietra G, Mortarini R, Parmiani G, Anichini A . Phases of apoptosis of melanoma cells, but not of normal melanocytes, differently affect maturation of myeloid dendritic cells. Cancer Res 2001; 61: 8218–26.
Cossarizza A, Baccarani-Contri M, Kalashnikova G, Franceschi C . A new method for the cytofluorimetric analysis of mitochondrial membrane potential using the J-aggregate forming lipophilic cation 5, 5′, 6, 6′-tetrachloro-1, 1′, 3, 3′-tetraethylbenzimi-dazolcarbocyanine iodide (JC-1). Biochem Biophys Res Commun 1993; 197: 40–5.
Shrivastava A, Tiwari M, Sinha RA, Kumar A, Balapure AK, Bajpai VK, et al. Molecular iodine induces caspase-independent apoptosis in human breast carcinoma cells involving the mitochondria-mediated pathway. J Biol Chem 2006; 281: 19 762–71.
Cass DL, Quinn TM, Yang EY, Liechty KW, Crombleholme TM, Flake AW, et al. Increased cell proliferation and decreased apoptosis characterize congenital cystic adenomatoid malformation of the lung. J Pediatr Surg 1998; 33: 1043–7.
Casasco A, Giordano M, Danova M, Casasco M, Icaro Cornaglia A, Calligaro A . PC 10 monoclonal antibody to proliferating cell nuclear antigen as probe for cycling cell detection in developing tissues. A combined immunocytochemical and flow cytometric study. Histochemistry 1993; 99: 191–9.
Alles AJ, Losty PD, Donahoe PK, Manganaro TF, Schnitzer JJ . Embryonic cell death patterns associated with nitrofen-induced congenital diaphragmatic hernia. J Pediatr Surg 1995; 30: 353–60.
Shao RG, Cao CX, Pommier Y . Activation of PKCalpha downstream from caspases during apoptosis induced by 7–hydro-xystaurosporine or the topoisomerase inhibitors, camptothecin and etoposide, in human myeloid leukemia HL60 cells. J Biol Chem 1997; 272: 31 321–5.
Otera H, Ohsakaya S, Nagaura Z, Ishihara N, Mihara K . Export of mitochondrial AIF in response to proapoptotic stimuli depends on processing at the intermembrane space. EMBO J 2005; 24: 1375–86.
Sarkar D, Su ZZ, Lebedeva IV, Sauane M, Gopalkrishnan RV, Valerie K, et al. mda-7 (IL-24) mediates selective apoptosis in human melanoma cells by inducing the coordinated overexpression of the GADD family of genes by means of p38 MAPK. Proc Natl Acad Sci USA 2002; 99: 10054–9.
Porras A, Zuluaga S, Black E, Valladares A, Alvarez AM, Ambrosino C, et al. p38α mitogen-activated protein kinase sensitizes cells to apoptosis induced by different stimuli. Mol Biol Cell 2004; 15: 922–33.
Maga G, Hubscher U . Proliferating cell nuclear antigen (PCNA): a dancer with many partners. J Cell Sci 2003; 116: 3051–60.
Keijzer R, Liu J, Deimling J, Tibboel D, Post M . Dual-hit hypothesis explains pulmonary hypoplasia in the nitrofen model of congenital diaphragmatic hernia. Am J Pathol 2000; 156: 1299–306.
Carvalho H, Evelson P, Sigaud S, Gonzalez-Flecha B . Mitogen-activated protein kinases modulate H(2)O(2)-induced apoptosis in primary rat alveolar epithelial cells. J Cell Biochem 2004; 92: 502–13.
Kling DE, Aidlen JT, Fisher JC, Kinane TB, Donahoe PK, Schnitzer JJ . Nitrofen induces a redox-dependent apoptosis associated with increased p38 activity in P19 teratocarcinoma cells. Toxicol In Vitro 2005; 19: 1–10.
Bahi N, Zhang J, Llovera M, Ballester M, Comella JX, Sanchis D . Switch from caspase-dependent to caspase-independent death during heart development: essential role of endonuclease G in ischemia-induced DNA processing of differentiated cardio-myocytes. J Biol Chem 2006; 281: 22 943–52.
McBurney MW, Jones-Villeneuve EM, Edwards MK, Anderson PJ . Control of muscle and neuronal differentiation in a cultured embryonal carcinoma cell line. Nature 1982; 299: 165–7.
Ravagnan L, Roumier T, Kroemer G . Mitochondria, the killer organelles and their weapons. J Cell Physiol 2002; 192: 131–7.
Griffiths GJ, Dubrez L, Morgan CP, Jones NA, Whitehouse J, Corfe BM, et al. Cell damage-induced conformational changes of the pro-apoptotic protein bak in vivo precede the onset of apoptosis. J Cell Biol 1999; 144: 903–14.
Yin XM, Oltvai ZN, Korsmeyer SJ . BH1 and BH2 domains of Bcl-2 are required for inhibition of apoptosis and heterodimerization with Bax. Nature 1994; 369: 321–3.
Daugas E, Nochy D, Ravagnan L, Loeffler M, Susin SA, Zamzami N, et al. Apoptosis-inducing factor (AIF): a ubiquitous mitochondrial oxidoreductase involved in apoptosis. FEBS Lett 2000; 476: 118–23.
Klein JA, Longo-Guess CM, Rossmann MP, Seburn KL, Hurd RE, Frankel WN, et al. The harlequin mouse mutation downregulates apoptosis-inducing factor. Nature 2002; 419: 367–74.
Cande C, Cohen I, Daugas E, Ravagnan L, Larochette N, Zamzami N, et al. Apoptosis-inducing factor (AIF): a novel caspase-independent death effector released from mitochondria. Biochimie 2000; 84: 215–22.
Susin SA, Lorenzo HK, Zamzami N, Marzo I, Snow BE, Brothers GM, et al. Molecular characterization of mitochondrial apoptosis-inducing factor. Nature 1999; 397: 441–6.
Wada T, Penninger JM . Mitogen-activated protein kinases in apoptosis regulation. Oncogene 2004; 23: 2838–49.
Seger R, Krebs EG . The MAPK signaling cascade. FASEB J 1995; 9: 726–35.
Irigoyen JP, Besser D, Nagamine Y . Cytoskeleton reorganization induces the urokinase-type plasminogen activator gene via the Ras/extracellular signal-regulated kinase (ERK) signaling pathway. J Biol Chem 1997; 272: 1904–9.
Stoica BA, Movsesyan VA, Knoblach SM, Faden AI . Ceramide induces neuronal apoptosis through mitogen-activated protein kinases and causes release of multiple mitochondrial proteins. Mol Cell Neurosci 2005; 29: 355–71.
Author information
Authors and Affiliations
Corresponding author
Additional information
Project supported by the National Natural Science Foundation of China (No 30200284) and the Science Foundation of Huazhong University of Science and Technology (No 2005-120).
Rights and permissions
About this article
Cite this article
Tong, Qs., Zheng, Ld., Tang, St. et al. Nitrofen suppresses cell proliferation and promotes mitochondria-mediated apoptosis in type II pneumocytes. Acta Pharmacol Sin 28, 672–684 (2007). https://doi.org/10.1111/j.1745-7254.2007.00552.x
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1111/j.1745-7254.2007.00552.x
Keywords
This article is cited by
-
The Regenerative Potential of Amniotic Fluid Stem Cell Extracellular Vesicles: Lessons Learned by Comparing Different Isolation Techniques
Scientific Reports (2019)
-
Endoplasmic reticulum stress response is activated in pulmonary hypoplasia secondary to congenital diaphragmatic hernia, but is decreased by administration of amniotic fluid stem cells
Pediatric Surgery International (2019)
-
The transcriptome of nitrofen-induced pulmonary hypoplasia in the rat model of congenital diaphragmatic hernia
Pediatric Research (2016)
-
Nitrofen interferes with trophoblastic expression of retinol-binding protein and transthyretin during lung morphogenesis in the nitrofen-induced congenital diaphragmatic hernia model
Pediatric Surgery International (2012)
-
Maternal administration of betamethasone inhibits proliferation induced by fetal tracheal occlusion in the nitrofen rat model for congenital diaphragmatic hernia: a placebo-controlled study
Pediatric Surgery International (2008)