Abstract
Aim:
To evaluate the in vivo antitumor effects of antisense oligonucleotides targeting midkine (MK-AS).
Methods:
An in situ human hepatocellular carcinoma (HCC) model was established in mice livers orthotopically. The MK-AS and 5-fluorouracil (5-Fu) were administered intravenously. The tumor sizes and plasma alpha-fetoprotein (AFP) were measured by calipers and radiation immunoassay respectively. The morphology of tumors was evaluated by hematoxylin-eosin staining of histological sections. Human MK, p53, Bax, Bcl-2, and caspase-3 protein content were detected by Western blotting.
Results:
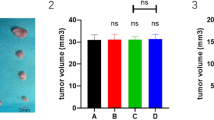
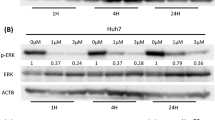
MK-AS significantly inhibited in situ human HCC growth in mice compared with the saline group in a dose-dependent manner. After the treatment with MK-AS or with 5-Fu, the plasma AFP concentration decreased in a dose-dependent manner. Interestingly, MK-AS also clearly downregulated the protein level of Bcl-2, and upregulated p53, Bax, and caspase-3 in the hepatocellular carcinoma tissue.
Conclusion:
These results demonstrated that MK-AS was an effective antitumor antisense oligonucleotide in vivo in mice; its antitumor effect is associated with the increase of pro-apoptotic proteins, such as p53, Bax, and caspase-3, and the decrease of the anti-apoptotic protein, Bcl-2.
Similar content being viewed by others
Article PDF
References
Okuda K . Hepatocellular carcinoma. J Hepatol 2000; 32: 225–37.
Befeler AS, Di Bisceglie, AM . Hepatocellular carcinoma: diagnosis and treatment. Gastroenterology 2002; 122: 1609–161.
Llovet JM, Burroughs A, Bruix J . Hepatocellular carcinoma. Lancet 2003; 362: 1907–17.
Marrero JA . Hepatocellular carcinoma. Curr Opin Gastroenterol 2006; 22: 248–53.
Mineura M, Tanimura H, Tabor E . Overexpression of multidrug resistance genes MDR1 and cMOAT in human hepatocellular carcinoma and hepatoblastoma cell lines. Int J Oncol 1999; 15: 559–63.
Huang M, Liu G . The study of innate drug resistance of human hepatocellular carcinoma Bel7402 cell line. Cancer Lett 1999; 135: 97–105.
Nies AT, Konig J, Pfannschimdt M, Klar E, Hofmann WJ, Keppler D . Expression of the multidrug resistance proteins MRP2 and MRP3 in human hepatocellular carcinoma. Int J Cancer 2001; 94: 492–9.
Heideman DA, Overmeer RM, van Beusechem VW, Lamers WH, Hakvoort TB, Snijders PJ, et al. Inhibition of angiogenesis and HGF-cMET-elicited malignant processes in human hepatocellular carcinoma cells using adenoviral vector-mediated NK4 gene therapy. Cancer Gene Ther 2005; 12: 954–62.
Lee HC, Kim M, Wands JR . Wnt/frizzled signaling in hepatocellular carcinoma. Front Biosci 2006; 11: 1901–15.
Han C, Michalopoulos GK, Wu T . Prostaglandin E2 receptor EP1 transactivates EGFR/MET receptor tyrosine kinases and enhances invasiveness in human hepatocellular carcinoma cells. J Cell Physiol 2006; 207: 261–70.
Ikeguchi M, Iwamoto A, Taniguchi K, Katano K, Hirooka Y . The gene expression level of transforming growth factor-beta (TGF-beta) as a biological prognostic marker of hepatocellular carcinoma. J Exp Clin Cancer Res 2005; 24: 415–21.
Musch A, Rabe C, Paik MD, Berna MJ, Schmitz V, Hoffmann P, et al. Altered expression of TGF-beta receptors in hepatocellular carcinoma—effects of a constitutively active TGF-beta type I receptor mutant. Digestion 2005; 71: 78–91.
Kadomatsu K, Tomomura M, Muramatsu T . cDNA cloning and sequencing of a new gene intensely expressed in early differentiation stages of embryonal carcinoma cells and in mid-gestation period of mouse embryogenesis. Biochem Biophys Res Commun 1988; 151: 1312–8.
Tomizawa M, Yu L, Wada A, Tamaoki T, Kadomatsu K, Muramatsu T, et al. A promoter region of the midkine gene that is frequently expressed in human hepatocellular carcinoma can activate a suicide gene as effectively as the alpha-fetoprotein promoter. Br J Cancer 2003; 89: 1086–90.
Yin Z, Luo X, Kang X, Wu Z, Qian H, Wu M . Correlation between midkine protein overexpression and intrahepatic metastasis in hepatocellular carcinoma. Zhonghua Zhong Liu Za Zhi 2002; 24: 27–9. Chinese.
Kato M, Shinozawa T, Kato S, Awaya A, Terada T . Increased midkine expression in hepatocellular carcinoma. Arch Pathol Lab Med 2000; 124: 848–52.
Li YS, Milner PG, Chauhan AK, Watson MA, Hoffman RM, Kodner CM, et al. Cloning and expression of a developmentally regulated protein that induces mitogenic and neurite outgrowth activity. Science 1990; 250: 1690–4.
Tsutsui J, Kadomatsu K, Matsubara S, Nakagawa A, Hamanoue M, Takao S, et al. A new family of heparin-binding growth differentiation factors: increased midkine expression in Wilms' tumor and other human carcinomas. Cancer Res 1993; 53: 1281–5.
Owada K, Sanjo N, Kobayashi T, Mizusawa H, Muramatsu H, Muramatsu T, et al. Midkine inhibits caspase-dependent apoptosis via the activation of mitogen-activated protein kinase and phosphatidylinositol 3–kinase in cultured neurons. J Neurochem 1999; 73: 2084–92.
Qi M, Ikematsu S, Ichihara-Tanaka K, Sakuma S, Muramatsu T, Kadomatsu K . Midkine rescues Wilms' tumor cells from cisplatin-induced apoptosis: regulation of Bcl-2 expression by midkine. J Biochem 2000; 127: 269–77.
Muramatsu H, Muramatsu T . Purification of recombinant midkine and examination of its biological activities: functional comparison of new heparin binding factors. Biochem Biophys Res Commun 1991; 177: 652–8.
Muramatsu H, Shirahama H, Yonezawa S, Maruta H, Muramatsu, T . Midkine (MK), a retinoic acid-inducible growth/differentiation factor: immunochemical evidence for the function and distribution. Dev Biol 1993; 159: 392–402.
Takada T, Toriyama K, Muramatsu H, Song XJ, Torii S, Muramatsu T . Midkine, a retinoic acid-inducible heparin-binding cytokine in inflammatory responses: chemotactic activity to neutrophils and association with inflammatory synovitis. J Biochem 1997; 122: 453–8.
Maeda N, Ichihara-Tanaka K, Kimura T, Kadomatsu K, Muramatsu T, Noda M . A receptor-like protein-tyrosine phosphatase PTPzeta/ RPTPbeta binds a heparin-binding growth factor midkine. Involvement of arginine 78 of midkine in the high affinity binding to PTPzeta. J Biol Chem 1999; 274: 12 474–9.
Horiba M, Kadomatsu K, Nakamura E, Muramatsu, H, Ikematsu, S, Sakuma S, et al. Neointima formation in a restenosis model is suppressed in midkine-deficient mice. J Clin Invest 2000; 105: 489–95.
Takei Y, Kadomatsu K, Yuasa K, Sato W, Muramatsu T . Morpholino antisense oligomer targeting human midkine: its application for cancer therapy. Int J Cancer 2005; 114: 490–7.
Takei Y, Kadomatsu K, Matsuo S, Itoh H, Nakazawa K, Kubota S, et al. Antisense oligodeoxynucleotide targeted to midkine, a heparin-binding growth factor, suppresses tumorigenicity of mouse rectal carcinoma cells. Cancer Res 2001; 61: 8486–91.
Dai LC, Wang X, Yao X, Lu YL, Ping JL, He JF . Antisense oligonucleotides targeting midkine induced apoptosis and increased chemosensitivity in hepatocellular carcinoma cells. Acta Pharmacol Sin 2006; 27: 1630–6.
Lin RX, Tuo CW, Lv QJ, Zhang W, Wang SQ . Inhibition of tumor growth and metastasis with antisense oligonucleotides (Cantide) targeting hTERT in an in situ human hepatocellular carcinoma model. Acta Pharmacol Sin 2005; 26: 762–8.
Bates PJ, Kahlon JB, Thomas SD, Trent JO, Miller DM . Antiproliferative activity of G-rich oligonucleotides correlates with protein binding. J Biol Chem 1999; 274: 26 369–77.
Nita ME, Nagawa H, Tominaga O, Tsuno N, Fujii S, Sasaki S, et al. 5-fluorouracil induces apoptosis in human colon cancer cell lines with modulation of Bcl-2 family proteins. Br J Cancer 1998; 78: 986–92.
Kanda T, Yokosuka O, Imazeki F, Arai M, Saisho H . Enhanced sensitivity of human hepatoma cells to 5-fluorouracil by small interfering RNA targeting Bcl-2. DNA Cell Biol 2005; 24: 805–9.
Sasaki M, Kumazaki T, Tanimoto K, Nishivama M . Bcl-2 in cancer and normal tissue cells as a prediction marker of response to 5-fluorouracil. Int J Oncol 2003; 22: 181–6.
Mirjolet JF, Barberi-Heyob M, Didelot C, Peyrat JP, Abecassis J, Millon R, et al. Bcl-2/Bax protein ratio predicts 5-fluorouracil sensitivity independently of p53 status. Br J Cancer 2000; 83: 1380–6.
Ohuchida T, Okamoto K, Akahane K, Higure A, Todoroki H, Abe Y, et al. Midkine protects hepatocellular carcinoma cells against TRAIL-mediated apoptosis through down-regulation of caspase-3 activity. Cancer 2004; 100: 2430–6.
Owada K, Sanjo N, Kobayashi T, Mizusawa H, Muramatsu H, Muramatsu T, et al. Midkine inhibits caspase-dependent apoptosis via the activation of mitogen-activated protein kinase and phosphatidylinositol 3–kinase in cultured neurons. J Neurochem 1999; 73: 2084–92.
Author information
Authors and Affiliations
Corresponding author
Additional information
Project supported by a grant from the Zhejiang Province Medicine and Sanitation Research Foundation (No 2003A077) and Huzhou Natural Science Foundation(No 2004SZX07-11).
The Editor has retracted this article because some of the figures have errors which were introduced during the process of integrating and editing the original images (Figures 1 and 3). Due to the fact that the research was conducted over ten years ago, the original data can no longer be found, and the article cannot be corrected. All authors agree to this retraction.
About this article
Cite this article
Dai, Lc., Wang, X., Yao, X. et al. RETRACTED ARTICLE: Antisense oligonucleotides targeting midkine inhibit tumor growth in an in situ human hepatocellular carcinoma model. Acta Pharmacol Sin 28, 453–458 (2007). https://doi.org/10.1111/j.1745-7254.2007.00532.x
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1111/j.1745-7254.2007.00532.x
Keywords
This article is cited by
-
Midkine confers Adriamycin resistance in human gastric cancer cells
Tumor Biology (2012)
-
Promotion of self-renewal of embryonic stem cells by midkine
Acta Pharmacologica Sinica (2010)
-
Effects of human midkine on spontaneous resorption of herniated intervertebral discs
International Orthopaedics (2010)