Abstract
Aim:
To investigate the apoptosis-inducing effect of oridonin, a diterpenoid isolated from Rabdosia rubescens, in the human cervical carcinoma HeLa cell line.
Methods:
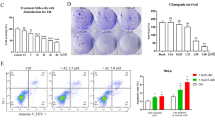
A morphological analysis, nuclear condensation, and fragmentation of chromatin were monitored using Hoechst 33342 staining. Cell viability was assessed using the 3-(4, 5-dimethylthiazol-(2)-yl)-2, 5-diphenyl tetrazolium bromide (MTT) assay. Cell apoptosis and the apoptosis-related activation in the HeLa cell line were evaluated by flow cytometry and Western blotting.
Results:
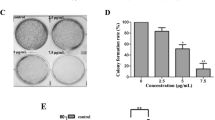
Oridonin suppressed the proliferation of the HeLa cell line in a dose- and time-dependent fashion. Oridonin treatment downregulated the activation of protein kinase B (Akt), the expression of forkhead box class O (FOXO) transcription factor, and glycogen synthase kinase 3 (GSK3). Oridonin also induced the release of cytochrome c accompanied by the activation of caspase-3 and poly-adenosine diphosphate-ribose polymerase cleavage. In addition, Z-D(OMe)-E(OMe)-V-D(OMe)-FMK (z-DEVD-fmk), an inhibitor of caspases, prevented caspase-3 activation and abrogated oridonin-induced cell death. Finally, oridonin treatment of the HeLa cell line downregulated the expression of the inhibitor of the apoptosis protein.
Conclusion:
Our results showed that oridonin-induced apoptosis involved several molecular pathways. Oridonin may suppress constitutively activated targets of phosphatidylinositol 3-kinase (Akt, FOXO, and GSK3) in the HeLa cell line, inhibiting the proliferation and induction of caspase-dependent apoptosis.
Similar content being viewed by others
Article PDF
References
Parkin DM, Bray F, Ferlay J, Pisani P . Estimating the world cancer burden:Globoscan 2000. Int J Cancer 2001; 94: 153–6.
de la Taille, A, Hayek OR, Burchardt M, Burchardt T, Katz AE . Role of herbal compounds (PC-SPES) in hormone-refractory prostate cancer: two case reports. J Altern Complement Med 2000; 6: 449–51.
Darzynkiewicz Z, Traganos F, Wu JM, Chen S . Chinese herbal mixture PC SPES in treatment of prostate cancer (review). Int J Oncol 2000; 17: 729–36.
Leung CH, Grill SP, Lam W, Han QB, Sun HD, Cheng YC . Novel mechanism of inhibition of nuclear factor-kappa B DNA-bindng activity by diterpenoids isolated from Isodon rubescens. Mol Pharmacol 2005; 68: 286–97.
Liu HM, Yan X, Kiuchi F, Liu Z . A new diterpene glycoside from Rabdosia rubescens. Chem Pharm Bull (Tokyo) 2000; 48: 148–9.
Ikezoe T, Yang Y, Bandobashi K, Saito T, Takemoto S, Machida H, et al. Oridonin, a diterpenoid purified from Rabdosia rubescens, inhibits the proliferation of cells from lymphoid malignancies in association with blockade of the NF-kappa B signal pathways. Mol Cancer Ther 2005; 4: 578–86.
Hsieh TC, Wijeratne EK, Liang JY, Gunatilaka AL, Wu JM . Differential control of growth, cell cycle progression, and expression of NF-kappaB in human breast cancer cells MCF-7, MCF-10A, and MDA-MB-231 by ponicidin and oridonin, diterpenoids from the Chinese herb Rabdosia rubescens. Biochem Biophys Res Commun 2005; 337: 224–31.
Zhang JF, Liu JJ, Liu PQ, Lin DJ, Li XD, Chen GH . Oridonin inhibits cell growth by induction of apoptosis on human hepa-tocellular carcinoma BEL-7402 cells. Hepatol Res 2006; 35: 104–10.
Liu JJ, Huang RW, Lin DJ, Peng J, Wu XY, Pan XL, et al. Anti-proliferative effects of oridonin on SPC-A-1 cells and its mechanism of action. J Int Med Res 2004; 32: 617–25.
Jin S, Shen JN, Wang J, Huang G, Zhou JG . Oridonin induced apoptosis through Akt and MAPKs signaling pathways in human osteosarcoma cells. Cancer Biol Ther 2007; 6: 261–8.
Liu YQ, You S, Tashiro S, Onodera S, Ikejima T . Activation of phosphoinositide 3-kinase, protein kinase C, and extracellular signal-regulated kinase is required for oridonin-enhanced phagocytosis of apoptotic bodies in human macrophage-like U937 cells. J Pharmacol Sci 2005; 98: 361–71.
Huang J, Wu L, Tashiro S, Onodera S, Ikejima T . Bcl-2 up-regulation and P-p53 down-regulation account for the low sensitivity of murine L929 fibrosarcoma cells to oridonin-induced apoptosis. Biol Pharm Bull 2005; 28: 2068–74.
Vivanco I, Sawyers CL . The phosphatidylinositol 3-kinase AKT pathway in human cancer. Nat Rev Cancer 2002; 2: 489–501.
Roy HK, Olusola BF, Clemens DL, Karolski WJ, Ratashak A, Lynch HT, et al. AKT proto-oncogene overexpression is an early event during sporadic colon carcinogenesis. Carcinogenesis 2002; 23: 201–5.
Wu X, Senechal K, Neshat MS, Whang YE, Sawyers CL . The PTEN/MMAC1 tumor suppressor phosphatase functions as a negative regulator of the phosphoinositide 3-kinase/Akt pathway. Proc Natl Acad Sci USA 1998; 95: 15 587–91.
Leslie NR, Gray A, Pass I, Orchiston EA, Downes CP . Analysis of the cellular functions of PTEN using catalytic domain and C-terminal mutations: differential effects of C-terminal deletion on signalling pathways downstream of phosphoinositide 3-kinase. Biochem J 2000; 346: 827–33.
Li BS, Ma W, Zhang L, Barker JL, Stenger DA, Pant HC . Activation of phosphatidylinositol-3-kinase (PI-3K) and extracellular regulated kinases (ERK1/2) is involved in muscarinic receptor mediated DNA synthesis in neural progenitor cells. J Neurosci 2001; 21: 1569–79.
Datta SR, Dudek H, Tao X, Masters S, Fu H, Gotoh Y, et al. Akt phosphorylation of BAD couples survival signals to the cell-intrinsic death machinery. Cell 1997; 91: 231–41.
Cross DA, Alessi DR, Cohen P, Andjelkovich M, Hemmings BA . Inhibition of glycogen synthase kinase-3 by insulin mediated by protein kinase B. Nature 1995; 378: 785–9.
Cardone MH, Roy N, Stennicke HR, Salvesen GS, Franke TF, Stanbridge E, et al. Regulation of cell death protease caspase-9 by phosphorylation. Science 1998; 282: 1318–21.
Brunet A, Bonni A, Zigmond MJ, Lin MZ, Juo P, Hu LS, et al. Akt promotes cell survival by phosphorylating and inhibiting a Forkhead transcription factor. Cell 1999; 96: 857–68.
Romashkova JA, Makarov SS . NF-kappaB is a target of AKT in anti-apoptotic PDGF signaling. Nature 1999; 401: 86–90.
Osawa K, Yasuda H, Maruyama T, Morita H, Takeya K, Itokawa H . Antibacterial trichorabdal diterpenes from Rabdosia trichocarpa. Phytochemistry 1994; 36: 1287–91.
Qi YL, Liao F, Zhao CQ, Lin YD, Zuo MX . Cytotoxicity, apoptosis induction, and mitotic arrest by a novel podophyllo-toxin glucoside, 4DPG, in tumor cells. Acta Pharmacol Sin 2005; 26: 1000–8.
Kennedy SG, Kandel ES, Cross TK, Hay N . Akt/Protein kinase B inhibits cell death by preventing the release of cytochrome c from mitochondria. Mol Cell Biol 1999; 19: 5800–10.
Zhou H, Li XM, Meinkoth J, Pittman RN . Akt regulates cell survival and apoptosis at a postmitochondrial level. J Cell Biol 2000; 151: 483–94.
Cui Q, Tashiro S, Onodera S, Ikejima T . Augmentation of oridonin-induced apoptosis observed with reduced autophagy. J Pharmacol Sci 2006; 101: 230–9.
Alvarez B, Martinez-A C, Burgering BM, Carrera AC . Forkhead transcription factors contribute to execution of the mitotic programme in mammals. Nature 2001; 413: 744–7.
Ciechomska I, Pyrzynska B, Kazmierczak P, Kaminska B . Inhibition of Akt kinase signalling and activation of forkhead are indispensable for upregulation of FasL expression in apoptosis of glioma cells. Oncogene 2003; 23: 7617–27.
Sawyers CL . Rational therapeutic intervention in cancer: kinases as drug targets. Curr Opin Genet Dev 2002; 12: 111–5.
Dan HC, Sun M, Kaneko S, Feldman RI, Nicosia SV, Wang HG, et al. Akt phosphorylation and stabilization of X-linked inhibitor of apoptosis protein (XIAP). J Biol Chem 2004; 279: 5405–12.
Alessi DR, Andjelkovic M, Caudwell B, Cron P, Morrice N, Cohen P, et al. Mechanism of activation of protein kinase B by insulin and IGF-1. EMBO J 1996; 15: 6541–51.
Puthalakath H, Strasser A . Keeping killers on a tight leash: transcriptional and post-translational control of the pro-apoptotic activity of BH3-only proteins. Cell Death Differ 2002; 9: 505–12.
Green DR, Kroemer, G . The pathophysiology of mitochondrial cell death. Science 2004; 305: 626–9.
Yang J, Liu X, Bhalla K, Kim CN, Ibrado AM, Cai J, et al. Prevention of apoptosis by Bcl-2 release of cytochrome c from mitochondria blocked. Science 1997; 275: 1129–32.
Zou H, Li Y, Liu X, Wang X . An APAF-1.cytochrome c multi-meric complex is a functional apoptosome that activates procaspase-9. J Biol Chem 1999; 274: 11 549–56.
Adida C, Crotty PL, McGrath J, Berrebi D, Diebold J, Altieri DC . Developmentally regulated expression of the novel cancer anti-apoptosis gene survivin in human and mouse differentiation. Am J Pathol 1998; 152: 43–9.
LaCasse EC, Baird S, Korneluk RG, MacKenzie AE . The inhibitors of apoptosis (IAPs) and their emerging role in cancer. Oncogene 1998; 17: 3247–59.
Kornacker M, Verneris MR, Kornacker B, Scheffold C, Negrin RS . Survivin expression correlates with apoptosis resistance after lymphocyte activation and is found preferentially in memory T cells. Immunol Lett 2001; 76: 169–73.
Author information
Authors and Affiliations
Corresponding author
Additional information
Project supported by grants from the National Natural Science Foundation of China (No 30571956) and the Medical Science and Technology Research Fund of Guangdong Province (No A2006188).
Rights and permissions
About this article
Cite this article
Hu, Hz., Yang, Yb., Xu, Xd. et al. Oridonin induces apoptosis via PI3K/Akt pathway in cervical carcinoma HeLa cell line. Acta Pharmacol Sin 28, 1819–1826 (2007). https://doi.org/10.1111/j.1745-7254.2007.00667.x
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1111/j.1745-7254.2007.00667.x
Keywords
This article is cited by
-
Inhibition of autophagy by chloroquine enhances the antitumor activity of gemcitabine for gallbladder cancer
Cancer Chemotherapy and Pharmacology (2020)
-
OP16 induces deadly autophagy and apoptosis of cells by inhibiting Akt in esophageal squamous cell carcinoma
Molecular and Cellular Biochemistry (2020)
-
Apatinib-induced protective autophagy and apoptosis through the AKT–mTOR pathway in anaplastic thyroid cancer
Cell Death & Disease (2018)
-
Anticancer activities and mechanisms of heat-clearing and detoxicating traditional Chinese herbal medicine
Chinese Medicine (2017)
-
Molecular Insight in the Multifunctional Effects of Oridonin
Drugs in R&D (2015)