Abstract
Aim:
To investigate the relationship between aquaporin-5 (AQP5) expression and mucus overproduction in the airways of Chinese patients with chronic obstructive pulmonary disease (COPD) and the correlation with pulmonary function change.
Methods:
Bronchial tissues were obtained from fiberoptic bronchoscopy and bronchial biopsy in West China Hospital from April to July 2004. Twenty-five patients were diagnosed as COPD patients, and another 20 were diagnosed as the control patients. The expressions of AQP5, mucin 5 AC (MUC5AC), and mucin in bronchial tissues were detected by semiquantitative RT-PCR, in situ hybridization, and immunohistochemical and alcian blue-periodic acid-Schiff (AB-PAS) staining, respectively.
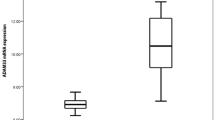
Results:
Compared with the control group, an attenuated expression of AQP5 was detected throughout the bronchial tissues from patients with COPD (P<0.01), but no difference existed in the lung tissues (P>0.05). Simultaneously, increased staining of MUC5 AC and mucus in submucosal glands were noted (P<0.01, respectively). Smoking attenuated the expressions of AQP5 and increased the staining of MUC5AC and mucus in submucosal glands in the COPD groups (P<0.01), while there were no significant differences observed in the control group (P>0.05). The decreased expression of AQP5 mRNA was correlated with forced expiratory volume in 1 s (FEV1)/forced vital capacity (FVC) (r=0.60, P<0.01), FEV1% predicted value (r= 0.60, P<0.01), maximal expiratory flow in 50% vital capacity (V50)% predicted value (r=0.55, P<0.01), and maximal expiratory flow in 25% vital capacity (V25) % predicted value (r=0.45, P<0.01). The decreased expression was negatively correlated with MUC5 AC mRNA of the epithelium airways (r=−0.45, P<0.01) and the AB-PAS-stained area of submucosal glands (r=−0.61, P<0.01). The upregulation of MUC5AC mRNA correlated with the positively AB-PAS-stained area of submucosal glands and correlated negatively with FEV1/FVC (r=−0.53; P<0.01), FEV1% predicted value (r=−0.53; P<0.01), V50% predicted value (r=−0.48; P<0.01), and V25% predicted value (r=−0.43; P<0.01).
Conclusion:
The attenuated gene expression of AQP5 existed in the airways of Chinese COPD patients, which was complicated by mucus hypersecretion. The decreased expression of AQP5 mRNA may be related to the severity of airflow obstruction.
Similar content being viewed by others
Article PDF
References
Wang F, Feng XC, Li YM, Yang H, Ma TH . Aquaporins as potential drug targets. Acta Pharmacol Sin 2006; 27: 394–401.
Verkman AS, Matthay MA, Song Y . Aquaporin water channels and lung physiology. Am J Physiol Lung Cell Mol Physiol 2000; 278: L867–79.
Verkman AS . Aquaporin water channels and endothelial cell function. J Anat 2002; 200: 617–27.
Borok Z, Verkman AS . Lung edema clearance: 20 years of progress interview: role of aquaporin water channels in fluid transport in lung and airways. J Appl Physiol 2002; 93: 2199–206.
Miller AL, Strieter RM, Gruber AD, Ho SB, Lukacs NW . CXCR2 regulates respiratory syncytial virus-induced airway hyperreactivity and mucus overproduction. J Immunol 2003; 170: 3348–56.
Kuperman DA, Huang X, Koth LL, Chang GH, Dolganov GM, Zhu Z, et al. Direct effects of interleukin-13 on epithelial cells cause airway hyperreactivity and mucus overproduction in asthma. Nat Med 2002; 8: 885–9.
Jayaraman S, Joo NS, Reitz B, Wine JJ, Verkman AS . Submucosal gland secretions in airways from cystic fibrosis patients have normal Na+ and pH but elevated viscosity. Proc Natl Acad Sci USA 2001; 98: 8119–23.
Rose MC, Piazza FM, Chen YA, Alimam MZ, Bautista MV, Letwin N, et al. Model systems for investigating mucin gene expression in airway diseases. J Aerosol Med 2000; 13: 245–61.
Zuhdi Alimam M, Piazza FM, Selby DM, Letwin N, Huang L, Rose MC, et al. Muc-5/5ac mucin messenger RNA and protein expression is a marker of goblet cell metaplasia in murine airways. Am J Respir Cell Mol Biol 2000; 22: 253–60.
Hoshino M, Nakamura Y, Sim JJ, Shimojo J, Isogai S . Bronchial subepithelial fibrosis and expression of matrix metalloproteinase-9 in asthmatic airway inflammation. J Allergy Clin Immunol 1998; 102: 783–8.
Desseyn JL, Buisine MP, Porchet N, Aubert JP, Laine A . Genomic organization of the human mucin gene MUC5B. cDNA and genomic sequences upstream of the large central exon. J Biol Chem 1998; 273: 30157–64.
Boucher RC . Molecular insights into the physiology of the ‘thin film’ of airway surface liquid. J Physiol 1999; 516: 631–8.
Kreda SM, Gynn MC, Fenstermacher DA, Boucher RC, Gabriel SE . Expression and localization of epithelial aquaporins in the adult human lung. Am J Respir Cell Mol Biol 2001; 24: 224–34.
King LS, Nielsen S, Agre P . Aquaporins in complex tissues. I. Developmental patterns in respiratory and glandular tissues of rat. Am J Physiol 1997; 273: C1541–8.
Jiao G, Li E, Yu R . Decreased expression of AQP1 and AQP5 in acute injured lungs in rats. Chin Med J 2002; 115: 963–7.
Song Y, Verkman AS . Aquaporin-5 dependent fluid secretion in airway submucosal glands. J Biol Chem 2001; 276: 41288–92.
Ma T, Song Y, Gillespie A, Carlson EJ, Epstein CJ, Verkman AS . Defective secretion of saliva in transgenic mice lacking aquaporin-5 water channels. J Biol Chem 1999; 274: 20071–4.
Krane CM, Fortner CN, Hand AR, McGraw DW, Lorenz JN, Wert SE, et al. Aquaporin-5 deficient mouse lungs are hyper-responsive to cholinergic stimulation. Proc Natl Acad Sci USA 2001; 98: 1059–63.
Bascom R . Differential susceptibility to tobacco smoke: possible mechanisms. Pharmacogenetics 1991; 1: 101–6.
Towne JE, Krane CM, Bachurski CJ, Menon AG . Tumor necrosis factor-α inhabits aquaporin 5 expression in mouse lung epithelial cells. J Biol Chem 2001; 276: 18657–64.
Lora JM, Zhang DM, Liao SM, Burwell T, King AM, Barker PA, et al. Tumor necrosis factor-alpha triggers mucus production in airway epithelium through an IkappaB kinase beta-dependent mechanism. J Biol Chem 2005; 280: 36510–7.
Tanaka M, Inase N, Fushimi K, Ishibashi K, Ichioka M, Sasaki S, et al. Induction of aquaporin 3 by corticosteroid in a human airway epithelial cell line. Am J Physiol 1997; 273: L1090–5.
Author information
Authors and Affiliations
Corresponding author
Additional information
This study was supported by grants from the National Natural Science Foundation of China (No 30425007, 30370627) and from the China Medical Board of New York to Dr Fu-qiang WEN (No 00-722).
Rights and permissions
About this article
Cite this article
Wang, K., Feng, Yl., Wen, Fq. et al. Decreased expression of human aquaporin-5 correlated with mucus overproduction in airways of chronic obstructive pulmonary disease. Acta Pharmacol Sin 28, 1166–1174 (2007). https://doi.org/10.1111/j.1745-7254.2007.00608.x
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1111/j.1745-7254.2007.00608.x
Keywords
This article is cited by
-
Differences in vulnerability to desiccating stress between corneal and conjunctival epithelium in rabbit models of short-term ocular surface exposure
Scientific Reports (2022)
-
Aquaporins in the lung
Pflügers Archiv - European Journal of Physiology (2019)
-
Aquaporin-5: from structure to function and dysfunction in cancer
Cellular and Molecular Life Sciences (2016)
-
Expression of aquaporins in bronchial tissue and lung parenchyma of patients with chronic obstructive pulmonary disease
Multidisciplinary Respiratory Medicine (2014)
-
Repair and remodeling of airway epithelium after injury in chronic obstructive pulmonary disease
Current Respiratory Care Reports (2013)