Abstract
Aim:
The spinal cord is pivotal in immobility induced by volatile anesthetics because the anesthetics depress the activity of motor neurons in the spinal cord. The aim of this study was to observe the effects of sevoflurane on pain processing at the spinal level.
Methods:
The firing of the gastrocnemius muscle was evoked by electrical stimulation to the ipsilateral hindpaw in rats. The nociceptive C response of electromyography (EMG) was selected to study. The GABAA receptor antagonist bicuculline (0.1 mg/kg) and opioid receptor antagonist naloxone (0.4 mg/kg) were administered intravenously, either in the presence or in the absence of 1.0% sevoflurane.
Results:
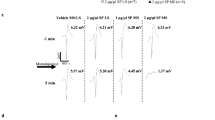
In rats with transected spinal cord, sevoflurane produced a profound reduction in the C response in a dose- and time-dependent manner. In the presence of 1.0% sevoflurane, the C responses were increased after injections of bicuculline and naloxone.
Conclusion:
Sevoflurane is a volatile anesthetic that acts directly on the spinal cord to suppress the nociceptive reflex. The sevoflurane-induced suppression of the C response is antagonized by either bicuculline or naloxone. The results suggest that spinal GABAA receptors and opioid peptide receptors are involved in the sevoflurane-induced suppression of spinal nociception.
Similar content being viewed by others
Article PDF
References
Jones MU, Brooks PA, Harrison NL . Enhancement of gamma-aminobutyric acid-activated Cl- currents in cultured rat hippocampal neurons by three volatile anaesthetics. J Physiol (Lond) 1992; 449: 279–93.
Krasowski MD, Harrison NL . General anaesthetic actions on ligand-gated ion channels. Cell Mol Life Sci 1999; 55: 1278–303.
Franks NP, Lieb WR . Which molecular targets are most relevant to general anaesthesia? Toxicol Lett 1998; 100-101: 1–8.
Bernd A . Different actions of general anaesthetics on the firing patterns of neocortical neurons mediated by the GABAA receptor. Anaesthesiology 1999; 91: 500–11.
Rampil IJ, King BS . Volatile anaesthetics depress spinal motor neurons. Anaesthesiology 1996; 85: 129–34.
Kammer T, Rehberg B, Menne D, Wartenberg HC, Wenningmann I, Urban BW . Propofol and sevoflurane in subanaesthetic concentrations act preferentially on the spinal cord. Anaesthesiology 2002; 97: 1416–25.
Mason P, Owens CA, Hammond DL . Antagonism of the antinocifensive action of halothane by intrathecal administration of GABAA receptor antagonists. Anaesthesiology 1996; 84: 1205–14.
Schwieger IM, Klopfenstein CE, Forster A . Epidural morphine reduces halothane MAC in humans. Can J Anaesth 1992; 39: 911–4.
Drasner K, Bernards CM, Ozanne GM . Intrathecal morphine reduces the minimum alveolar concentration of halothane in humans. Anaesthesiology 1988; 69: 310–2.
Criado AB, Gomez de Segura IA, Tendillo FJ, Marsico F . Reduction of isoflurane MAC with buprenorphine and morphine in rats. Lab Anim 2000; 34: 252–9.
Bai L, Zhao ZQ . Ketamine-induced peripheral analgesia in rats. Acta Pharmacol Sin 1997; 18: 377–9.
Antognini JF, Carstens E, Buzin V . Isoflurane depresses motoneuron excitability by a direct spinal action: an F-wave study. Anaesth Analg 1999; 88: 681–5.
Sivilotti L, Nistri A . GABA receptor mechanisms in the central nervous system. Prog Neurobiol 1991; 36: 35–92.
Zhang Y, Wu S, Eger I II, Sonner JM . Neither GABAA nor strychnine-sensitive glycine receptors are the sole mediators of MAC for isoflurane. Anaesth Analg 2001; 92: 123–7.
Yamauchi M, Sekiyama H, Shimada SG, Collins JG . Halothane suppression of spinal sensory neuronal responses to noxious peripheral stimuli is mediated, in part, by both GABAA and glycine receptor systems. Anaesthesiology 2002; 97: 412–7.
Hirota K, Roth SH . Sevoflurane modulates both GABAA and GABAB receptors in area CA1 of rat hippocampus. Br J Anaesth 1997; 78: 60–5.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Wang, Yw., Deng, Xm., You, Xm. et al. Involvement of GABA and opioid peptide receptors in sevoflurane-induced antinociception in rat spinal cord. Acta Pharmacol Sin 26, 1045–1048 (2005). https://doi.org/10.1111/j.1745-7254.2005.00175.x
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1111/j.1745-7254.2005.00175.x