Abstract
Aim:
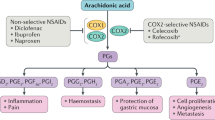
COX-2(cyclooxygenase-2) has sparked a surge in pharmaceutical interest since its discovery at the beginning of the 1990s. Several COX-2 selective inhibitors that avoid gastrointestinal side effects have been successfully launched into the market in recent years. The first selective COX-2 inhibitor, celecoxib, entered the market in December 1999. However, there are a few organs that physiologically and functionally express COX-2, particularly the glomeruli of the kidney and the cortex of the brain. Inhibition of COX-2 expression in these organs possibly causes heart attack and stroke in long-term COX-2 inhibitor users. Recently, a USA Food and Drug Agency (FDA) advisory panel re-evaluated COX-2 inhibitors and unanimously concluded that the entire class of COX-2 inhibitors increase the risk of cardiovascular problems. Thus the use of COX-2 inhibitors is still controversial, and there is a challenge for not only pharmacologists, but also the pharmaceutical industry, to develop improved painkilling and anti-inflammatory drugs. This may involve exploring a new generation of COX-2 inhibitors with different inhibitory mechanisms through computer-aided design, screening different sources of inhibitors with lower selectivity, or seeking completely new targets. Synthetic COX-2 inhibitors have high selectivity and the advantage of irreversible inhibition, whereas naturally derived COX-2 inhibitors have lower selectivity and fewer side effects, with the medical effects in general not being as striking as those achieved using synthetic inhibitors. This review discusses the mechanism of COX-2 inhibitor therapy and a possible new way of exploration in the development of anti-inflammatory, analgetic, and antipyretic drugs.
Similar content being viewed by others
Article PDF
References
Xie WL, Chipman JG, Robertson DL, Erikson RL, Simmons DL . Expression of a mitogen-responsive gene encoding prostaglandin synthase is regulated by mRNA splicing. Proc Natl Acad Sci USA 1991; 88: 692–6.
Fu JY, Masferr JL, Seibert K, Raz A, Needleman P . The induction and suppression of prostaglandin h2 synthase (cyclooxygenase) in human monocytes. J Biol Chem 1990; 265: 16737–40.
Luo C, Laine JVO, Ylinen L, Teros T, Mäkinen M, Ristimäki A, et al. Expression of cyclooxygenase-2 in intestinal goblet cells of prediabetic non-obese diabetic mice. Acta Physiol Scand 2002; 174: 265–74.
Luo C, Trauss L, Treng T, Ristimäki A, Santti R . Constant exprkession of cyclooxygenase-2 gene in the lower urinary tract and prostate of the male rats treated with estrogen. Z Naturforsch 2001; 56C: 455–63.
Gilroy DW, Colville-Nash PR, Willis D, Chivers J, Paul-Clark MJ, Willoughby DA . Inducible cyclooxygenase may have anti-inflammatory properties. Nat Med 1999; 5: 621–2.
Topol EJ . Arthritis Medicines and Cardiovascular Events: “House of Coxibs”. J Am Med Assoc 2005; 293: 366–8.
Warner TD, Mitchell JA . Cyclooxygenases: new forms, new inhibitors, and lessons from the clinic. FASEB J 2004; 18: 790–804.
Kraemer SA, Meade EA, DeWitt DL . Prostaglandin endoperox-ide synthase gene structure: identification of the transcriptional start site and 5′-flanking regulatory sequences. Arch Biochem Biophys 1992; 293, 391–400.
Colville-Nash PR, Gilroy DW . Potential adverse effects of cyclooxygenase-2 inhibition: evidence from animal models of inflammation. BioDrugs 2001; 15: 1–9.
Onoe Y, Miyaura C, Kaminakayashiki T, Nagai Y, Noguchi K, Chen QR, et al. IL-13 and IL-4 inhibit bone resorption by suppressing cyclooxygenase-2-dependent prostaglandin synthesis in osteoblasts. J Immunol 1996; 156: 758–64.
Hinz B, Brune K, Pahl A . Cyclooxygenase-2 expression in li-popolysaccharide-stimulated human monocytes is modulated by cyclic AMP, prostaglandin E2 and non-steroidal anti-inflammatory drugs. Biochem Biophys Res Commun 2000; 278: 790–6.
Dixon DA, Kaplan CD, McIntyre TM, Zimmerman GA, Prescott SM . Post-transcriptional control of cyclooxygenase-2 gene expression. The role of the 3′-untranslated region. J Biol Chem 2000; 275: 11750–7.
Ristimäki A, Honkanen N, Jankala H, Sipponen P, Harkonen M . Expression of cyclooxygenase-2 in human gastric carcinoma. Cancer Res 1997; 57: 1276–80.
Vane JR . Inhibition of prostaglandin synthesis as a mechanism of action for the aspirin-like drugs. Nature 1971; 231: 232–5.
Michiels JJ . Aspirin and platelet-lowering agents for the prevention of vascular complications in essential thrombocythemia. Clin Appl Thromb Hemost. 1999; 5: 247–51.
Lichtenstein DR, Wolfe MM . COX-2 selective NSAID, new and improved? J Am Med Assoc 2000; 284: 1297–9.
Luo C, Kallajoki M, Mäkinen M, Teros T, Ylinen L, Simell O . Distribution and contribution of cyclooxygenase (COX)-2 for diabetogenesis in NOD mouse model. Cell Tissue Res 2002; 310: 169–75.
McKanna JA, Zhang MZ, Wang JL, Cheng H, Harris RC . Constitutive expression of cyclooxygenase-2 in rat vas deferens. Am J Physiol 1998; 275: R227–33.
Harris RC . Macula densa signaling, a potential role of cyclooxygenase-2 (COX-2)? Nephrol Dial Transplant 2000; 15: 1504–6.
Luo C, Laaja P . Inhibitors of JAKs/STATs and the kinases, a possible new cluster of drugs. Drug Discov Today 2004; 9: 268–75.
Tiano HF, Loftin CD, Akunda J, Lee CA, Spalding J, Sessoms A, et al. Deficiency of either cyclooxygenase (COX)-1 or COX-2 alters epidermal differentiation and reduces mouse skin tumorigenesis. Cancer Res 2002; 62: 3395–401.
Baba H, Kohno T, Moore KA and Woolf CJ . Direct activation of rat spinal dorsal horn neurons by prostaglandin E2. J Neurosci 2001; 21: 1750–6.
Samad TA, Moore KA, Sapirstein A, Billet S, Allchorne A, Poole S, et al. Interleukin- 1b-mediated induction of Cox-2 in the CNS contributes to inflammatory pain hypersensitivity. Nature 2001; 410: 471–5.
Ma W, Eisenach JC . Morphological and pharmacological evidence for the role of peripheral prostaglandins in the pathogenesis of neuropathic pain. Eur J Neurosci 2002; 15: 1037–47.
Ellman MH, Curran J . Trial of tramadol/acetaminophen tablets for osteoarthritis pain in subjects receiving a COX-2 nonsteroidal antiinflammatory drug. J Rheumatol 2005; 32: 568–8.
Boutaud O, Aronoff DM, Richardson JH, Marnett LJ, Oates JA . Determinants of the cellular specificity of acetaminophen as an inhibitor of prostaglandin H2 synthases. Proc Natl Acad Sci USA 2002; 99: 7130–5.
Botting R, Ayoub SS . COX-3 and the mechanism of action of paracetamol/acetaminophen. Prostaglandins Leukot Essent Fatty Acids 2005; 72: 85–7.
van Ryn J, Trummlitz G, Pairet M . COX-2 selectivity and inflammatory processes. Curr Med Chem 2000; 7: 1145–61.
Kurumbail RG, Stevens AM, Gierse JK, McDonald JJ, Stegeman RA, Pak JY, et al. Structural basis for selective inhibition of cyclooxygenase-2 by anti-inflammatory agents. Nature 1997; 385: 555–62.
Yang T, Endo Y, Huang YG, Smart A, Briggs JP, Schnermann J . Renin expression in COX-2-knockout mice on normal or low-salt diets. Am J Physiol Renal Physiol 2000; 279: F819–25.
Bonner JC, Rice AB, Ingram JL, Moomaw CR, Nyska A, Bradbury A, et al. Susceptibility of cyclooxygenase-2-deficient mice to pulmonary fibrogenesis. Am J Pathol 2002; 161: 459–70.
Mukherjee D, Topol EJ . COX-2: where are we in 2003? Cardiovascular risk and COX-2 inhibitors. Arthritis Res Ther 2003; 5: 8–11.
Adegboyega PA, Ololade O . Immunohistochemical expression of cyclooxygenase-2 in normal kidneys. Appl Immunohistochem Mol Morphol 2004; 12: 71–4.
McAdam BF, Catella-Lawson F, Mardini IA, Kapoor S, Lawson JA, FitzGerald GA . Systemic biosynthesis of prostacyclin by cyclooxygenase (COX)-2: the human pharmacology of a selective inhibitor of COX-2. Proc Natl Acad Sci USA 1999; 96: 272–7.
Catella-Lawson F, McAdam B, Morrison BW, Kapoor S, Kujubu D, Antes L, et al. Effects of specific inhibition of cyclooxygenase-2 on sodium balance, hemodynamics, and vasoactive eicosanoids. J Pharmacol Exp Ther 1999; 289: 735–41.
Solomon SD, McMurray JJ, Pfeffer MA, Wittes J, Fowler R, Finn P, et al. Cardiovascular risk associated with celecoxib in a clinical trial for colorectal adenoma prevention. N Engl J Med 2005; 352: 1071–80.
Bresalier RS, Sandler RS, Quan H, Bolognese JA, Oxenius B, Horgan K, et al. Cardiovascular events associated with rofecoxib in a colorectal adenoma chemoprevention trial. N Engl J Med 2005; 352: 1092–102.
Perera P, Ringbom T, Huss U, Vasänge M, Bohlin L . Search for natural products with effect on cyclooxygenase-2. In: Tringali C, editor. Bioactive Compounds from Natural Sources. London: Taylor & Francis; 2001. p 433–72.
Subbaramaiah K, Michaluart P, Chung WJ, Tanabe T, Telang N, Dannenberg AJ . Resveratrol inhibits cyclooxygenase-2 transcription in human mammary epithelial cells. Ann NY Acad Sci 1999; 889: 214–23.
Ringbom T, Huss U, Stenholm A, Flock S, Skattebol L, Perera P, et al. COX-2 inhibitory effects of naturally occurring and modified fatty acids. J Nat Prod 2001; 64: 745–9.
Arita M, Bianchini F, Aliberti J, Sher A, Chiang N, Hong S, et al. Stereochemical assignment, antiinflammatory properties, and receptor for the omega-3 lipid mediator resolvin E1. J Exp Med 2005; 201: 713–22.
Ringbom T, Segura L, Noreen Y, Perera P, Bohlin L . Ursolic acid from Plantago major, a selective inhibitor of cyclooxygenase-2 catalyzed prostaglandin biosynthesis. J Nat Prod 1998; 61: 1212–5.
Stock JL, Shinjo K, Burkhardt J, Roach M, Taniguchi K, Ishikawa T, The prostaglandin E2 EP1 receptor mediates pain perception and regulates blood pressure. J Clin Invest 2001; 107: 325–31.
Author information
Authors and Affiliations
Corresponding author
Additional information
Disclaimer: the views expressed herein are those of the authors and are not intended to represent those of the institutions with which they are affiliated.
Rights and permissions
About this article
Cite this article
Luo, C., He, Ml. & Bohlin, L. Is COX-2 a perpetrator or a protector? Selective COX-2 inhibitors remain controversial. Acta Pharmacol Sin 26, 926–933 (2005). https://doi.org/10.1111/j.1745-7254.2005.00150.x
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1111/j.1745-7254.2005.00150.x
Keywords
This article is cited by
-
Prediction of the potency of mammalian cyclooxygenase inhibitors with ensemble proteochemometric modeling
Journal of Cheminformatics (2015)
-
Assessment of pharmacological activities of two medicinal plant of Bangladesh: Launaea sarmentosa and Aegialitis rotundifolia roxb in the management of pain, pyrexia and inflammation
Biological Research (2014)
-
In vivo analgesic, antipyretic, and anti-inflammatory potential in Swiss albino mice and in vitro thrombolytic activity of hydroalcoholic extract from Litsea glutinosa leaves
Biological Research (2014)
-
Does administration of non-steroidal anti-inflammatory drug determine morphological changes in adrenal cortex: ultrastructural studies
Protoplasma (2010)
-
Promotion of prostatic metastatic migration towards human bone marrow stoma by Omega 6 and its inhibition by Omega 3 PUFAs
British Journal of Cancer (2006)