Abstract
Aim:
To define the role of enzymes involved in glutathione metabolism in 6-hydroxydopamine (6-OHDA)-induced glutathione alteration in primary cultured astrocytes.
Methods:
Total glutathione (GSx) levels were determined using the modified enzymatic microtiter plate assay. The mRNA levels of γ-glutamylcysteine synthetase (γGCS), γ-glutamyltransferase (γGT), glutathione peroxidase (GPx), GR (glutathione reductase), and glutathione transferases (GST) were determined using RT-PCR. γGT activity was determined using γGT assay kits.
Results:
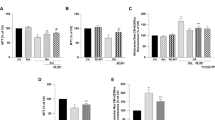
In primary cultured astrocytes, 6-OHDA induced a significant elevation of cellular GSx levels after treatment for 24 h. However, the GSx levels decreased after 24 h and the values were even lower than the value in the control group without 6-OHDA at 48 h. RT-PCR data showed that the mRNA levels of γGCS, the rate-limiting enzyme of γ-L-glutamyl-L-cysteinylglycine (GSH) synthesis, were increased by 6-OHDA after treatment for 24 h and 48 h; the mRNA levels of GPx, GR, and GST did not alter in 6-OHDA-treated astrocytes after treatment for 24 h and 48 h; and 6-OHDA increased the mRNA levels and the activity of γGT after treatment for 48 h, which induced a decrease in GSx levels, despite the up-regulation of γGCS after exposure to 6-OHDA for 48 h.
Conclusion:
The change in γGCS correlated with the increase in GSH levels induced by 6-OHDA after treatment for 24 h. GSx levels decreased because of increased γGT mRNA levels and γGT activity induced by 6-OHDA after treatment for 48 h.
Similar content being viewed by others
Article PDF
References
Jenner P . Oxidative stress in Parkinson's disease. Ann Neurol 2003; 53: S26–S38.
Blum D, Torch S, Lambeng N, Nissou M, Benabid AL, Sadoul R, et al. Molecular pathways involved in the neurotoxicity of 6-OHDA, dopamine and MPTP: contribution to the apoptotic theory in Parkinson's disease. Prog Neurobiol 2001; 65: 135–72.
Cooper AJL . Role of astrocytes in maintaining cerebral glutathione homeostasis and in protecting the brain against xenobiotics and oxidative stress. In: Shaw CA, editor The role of glutathione in the nervous system. Washington: Taylor and Francis; 1998. p 91–115.
Halliwell B, Gutteridge JMC . Free radicals in biology and medicine. New York: Oxford University Press; 1999.
Bains JS, Shaw CA . Neurodegenerative disorders in humans: the role of glutathione in oxidative stress-mediated neuronal death. Brain Res Rev 1997; 25: 335–58.
Sian J, Dexter DT, Lees AJ, Daniel S, Agid Y, Javoy-Agid F, et al. Alteration in glutathione levels in Parkinson's disease and other neurodegenerative disorders affecting basal ganglia. Ann Neurol 1994; 36: 348–55.
Aksenov MY, Markesbery WR . Changes in thiol content and expression of glutathione redox system genes in the hippocampus and cerebellum in Alzheimer's disease. Neurosci Lett 2001; 302: 141–5.
Anderson ME, Meister A . Transport and direct utilization of γ-glutamylcyst(e)ine for glutathione synthesis. Proc Natl Acad Sci USA 1983; 80: 707–11.
Dringen R, Kranich O, Hamprecht B . The 7-glutamyl transpeptidase inhibitor acivicin preserves glutathione released by astroglial cells in culture neurochemical research. Neurochem Res 1997; 22: 727–33.
Hamprecht B, Loffler F . Primary glial cultures as a model for studying hormone action. Methods Enzymol 1985; 109: 341–5.
Tietze F . Enzymic method for quantitative determination of nanogram amounts of total and oxidized glutathione: applications to mammalian blood and other tissues. Anal Biochem 1969; 27: 502–22.
Kruger NJ . The Bradford method for protein quantitation. Methods Mol Biol 1994; 32: 9–15.
Uitti RJ, Calne DB . Pathogenesis of idiopathic Parkinsonism. Eur Neurol 1993; 33 ( Suppl 1): 6–23.
Dringen R . Metabolism and functions of glutathione in brain. Prog Neurobiol 2000; 62: 649–71.
Iwata-Ichikawa E, Kondo Y, Miyazaki I, Asanuma M, Ogawa N . Glial cells protect neurons against oxidative stress via transcriptional up-regulation of the glutathione synthesis. J Neurochem 1999; 72: 2334–44.
Shimizu E, Hashimoto K, Komatsu N, Iyo M . Roles of endogenous glutathione levels on 6-hydroxydopamine induced apoptotic neuronal cell death in human neuroblastoma SK-N-SH cells. Neuropharmacology 2002; 43: 434–43.
Sagara J, Miura K, Bannai S . Cysteine uptake and glutathione level in fetal brain cells in primary culture and in suspension. J Neurochem 1993; 61: 1667–71.
Nguyen T, Sherratt PJ, Pickett CB . Regulatory mechanisms controlling gene expression mediated by the antioxidant response element. Annu Rev Phamacol Toxicol 2003; 43: 233–60.
Mulcahy RT, Gipp JJ . Identification of a putative antioxidant response element in the 5′-flanking region of the human gammaglutamylcysteine synthetase heavy subunit gene. Biochem Biophys Res Commun 1995; 209: 227–33.
Schmuck G, Rohrdanz E, Tran-Thi QH, Kahl R, Schluter G . Oxidative stress in rat cortical neurons and astrocytes induced by paraquat in vitro. Neurotox Res 2002; 4: 1–13.
Owen AD, Schapira AH, Jenner P, Marsden CD . Oxidative stress and Parkinson's disease. Ann NY Acad Sci 1996; 786: 217–23.
Dringen R, Pfeiffer B, Hamprecht B . Synthesis of the antioxidant glutathione in neurons: supply by astrocytes of CysGly as precursor for neuronal glutathione. J Neurosci 1999; 19: 562–69.
Gegg ME, Beltran B, Salas-Pino S, Bolanos JP, Clark JB, Moncada S, et al. Differential effect of nitric oxide on glutathione metabolism and mitochondrial function in astrocytes and neurones: implications for neuroprotection/neurodegeneration. J Neurochem 2003; 86: 228–37.
Ruedig C, Dringen R . TNFα increases the activity of γ-glutamyl transpeptidase in cultured rat astroglial cells. J Neurosci Res 2004; 75: 536–43.
Boka G, Anglade P, Wallach D, Javoy-Agid F, Agid Y, Hirsch EC . Immunocytochemical analysis of tumor necrosis factor and its receptors in Parkinson's disease. Neurosci Lett 1994; 172: 151–54.
Nobre HV Jr, Cunha GM, Maia FD, Oliveira RA, Moraes MO, Rao VS . Catechin attenuates 6-hydroxydopamine (6-OHDA)-induced cell death in primary cultures of mesencephalic cells. Comp Biochem Physiol C Toxicol Pharmacol 2003; 136: 175–80.
Mogi M, Togari A, Tanaka K, Ogawa N, Ichinose H, Nagatsu T . Increase in level of tumor necrosis factor (TNF)-alpha in 6-hydroxydopamine-lesioned striatum in rats without influence of systemic L-DOPA on the TNF-alpha induction. Neurosci Lett 1999; 268: 101–4.
Puka-Sundvall M, Eriksson P, Nilsson M, Sandberg M, Lehmann A . Neurotoxicity of cysteine: interaction with glutamate. Brain Res 1995; 705: 65–70.
Author information
Authors and Affiliations
Corresponding author
Additional information
Project supported by the National Natural Science Foundation of China (No 39970846).
Rights and permissions
About this article
Cite this article
Zhang, J., Hu, J., Ding, Jh. et al. 6-Hydroxydopamine-induced glutathione alteration occurs via glutathione enzyme system in primary cultured astrocytes. Acta Pharmacol Sin 26, 799–805 (2005). https://doi.org/10.1111/j.1745-7254.2005.00124.x
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1111/j.1745-7254.2005.00124.x
Keywords
This article is cited by
-
6-Hydroxydopamine: a far from simple neurotoxin
Journal of Neural Transmission (2020)
-
Monocrotophos Induces the Expression of Xenobiotic Metabolizing Cytochrome P450s (CYP2C8 and CYP3A4) and Neurotoxicity in Human Brain Cells
Molecular Neurobiology (2017)
-
Dopamine as a Potent Inducer of Cellular Glutathione and NAD(P)H:Quinone Oxidoreductase 1 in PC12 Neuronal Cells: A Potential Adaptive Mechanism for Dopaminergic Neuroprotection
Neurochemical Research (2008)