Abstract
Aim:
To investigate whether activation and translocation of extracellular signal-regulated kinase (ERK) is involved in the induction and maintenance of neuropathic pain, and effects of activation and translocation of ERK on expression of pCREB and Fos in the chronic neuropathic pain.
Methods:
Lumbar intrathecal catheters were chronically implanted in male Sprague-Dawley rats. The left sciatic nerve was loosely ligated proximal to the sciatica's trifurcation at approximately 1.0 mm intervals with 4-0 silk sutures. The mitogen-activated protein kinase kinase (MEK) inhibitor U0126 or phosphorothioate-modified antisense oligonucleotides (ODN) were intrathecally administered every 12 h, 1 d pre-chronic constriction injury (CCI) and 3 d post-CCI. Thermal and mechanical nociceptive thresholds were assessed with the paw withdrawal latency (PWL) to radiant heat and von Frey filaments. The expression of pERK, pCREB, and Fos were assessed by both Western blotting and immunohistochemical analysis.
Results:
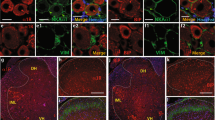
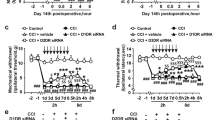
Intrathecal injection of U0126 or ERK antisense ODN significantly attenuated CCI-induced mechanical allodynia and thermal hyperalgesia. CCI significantly increased the expression of p-ERK-IR neurons in the ipsilateral spinal dorsal horn to injury, not in the contralateral spinal dorsal horn. The time courses of pERK expression showed that the levels of both cytosol and nuclear pERK, but not total ERK, were increased at all points after CCI and reached a peak level on postoperative d 5. CCI also significantly increased the expression of pCREB and Fos. Phospho-CREB-positive neurons were distributed in all laminae of the bilateral spinal cord and Fos was expressed in laminae I and II of the ipsilateral spinal dorsal horn. Intrathecal injection of U0126 or ERK antisense ODN markedly suppressed the increase of CCI-induced pERK, pCREB and c-Fos expression in the spinal cord.
Conclusion:
The activation of ERK pathways contributes to neuropathic pain in CCI rats, and the function of pERK may partly be accomplished via the cAMP response element binding protein (CREB)-dependent gene expression.
Similar content being viewed by others
Article PDF
References
Ikeda H, Heinke B, Ruscheweyh R, Sandkuhler J . Synaptic plasticity in spinal lamina. I projection neurons that mediate hyperalgesia. Science 2003; 299: 1237–40.
Melzack R, Coderre TJ, Katz J, Vaccarino AL . Central neuroplasticity and pathological pain. Ann NY Acad Sci 2001; 933: 157–74.
Wolpaw JR, Tennissen AM . Activity-dependent spinal cord plasticity in health and disease. Annu Rev Neurosci 2001; 24: 807–43.
Ji RR, Kohno T, Moore KA, Woolf CJ . Central sensitization and LTP: do pain and memory share similar mechanisms? Trends Neurosci 2003; 26: 696–705.
Rygh LJ, Tjolsen A, Hole K, Svendsen F . Cellular memory in spinal nociceptive circuitry. Scand J Psychol 2002; 43: 153–9.
Messaoudi E, Ying SW, Kanhema T, Croll SD, Bramham CR . Brain-derived neurotrophic factor triggers transcription-dependent, late phase long-term potentiation in vivo. J Neurosci 2002; 22: 7453–61.
Woolf CJ, Costigan M . Transcriptional and posttranslational plasticity and the generation of inflammatory pain. Proc Natl Acad Sci USA 1999; 96: 7723–30.
Chen A, Muzzio IA, Malleret G, Bartsch D, Verbitsky M, Pavlidis P, et al. Inducible enhancement of memory storage and synaptic plasticity in transgenic mice expressing an inhibitor of ATF4 (CREB-2) and C/EBP proteins. Neuron 2003; 39: 655–69.
Ho N, Liauw JA, Blaeser F, Wei F, Hanissian S, Muglia LM, et al. Impaired synaptic plasticity and cAMP response element-binding protein activation in Ca2+/calmodulin-dependent protein kinase type IV/Gr-deficient mice. J Neurosci 2000; 20: 6459–72.
Hoeger-Bement MK, Sluka KA . Phosphorylation of CREB and mechanical hyperalgesia is reversed by blockade of the cAMP pathway in a time-dependent manner after repeated intramuscular acid injections. J Neurosci 2003; 23: 5437–45.
Ma W, Hatzis C, Eisenach JC . Intrathecal injection of cAMP response element binding protein (CREB) antisense oligonucleotide attenuates tactile allodynia caused by partial sciatic nerve ligation. Brain Res 2003; 988: 97–104.
Ma W, Quirion R . Increased phosphorylation of cyclic AMP response element-binding protein (CREB) in the superficial dorsal horn neurons following partial sciatic nerve ligation. Pain 2001; 93: 295–301.
Kelleher RJ, Govindarajan A, Jung HY, Kang H, Tonegawa S . Translational control by MAPK signaling in long-term synaptic plasticity and memory. Cell 2004; 116: 467–79.
Morozov A, Muzzio IA, Bourtchouladze R, Van-Strien N, Lapidus K, Yin D, et al. Rap1 couples cAMP signaling to a distinct pool of p42/44MAPK regulating excitability, synaptic plasticity, learning, and memory. Neuron 2003; 39: 309–25.
Thomas GM, Huganir RL . MAPK cascade signalling and synaptic plasticity. Nature Rev Neurosci 2004; 5: 173–83.
Aley KO, Martin A, McMahon T, Mok J, Levine JD, Messing RO . Nociceptor sensitization by extracellular signal-regulated kinases. J Neurosci 2001; 21: 6933–9.
Ciruela A, Dixon AK, Bramwell S, Gonzalez MI, Pinnock RD, Lee K . Identification of MEK1 as a novel target for the treatment of neuropathic pain. Br J Pharmacol 2003; 138: 751–6.
Galan A, Cervero F, Laird JM . Extracellular signaling-regulated kinase-1/2 (ERK1/2) mediate referred hyperalgesia in a murine model of visceral pain. Mol Brain Res 2003; 116: 126–34.
Ji RR, Baba H, Brenner GJ, Woolf CJ . Nociceptive-specific activation of ERK in spinal neurons contributes to pain hypersensitivity. Nat Neurosci 1999; 2; 1114–9.
Ji RR, Befort K, Brenner GJ, Woolf CJ . ERK MAP kinase activation in superficial spinal cord neurons induces prodynorphin and NK-1 upregulation and contributes to persistent inflammatory pain hypersensitivity. J Neurosci 2002; 22; 478–85.
Wang H, Dai Y, Fukuoka T, Yamanaka H, Obata K, Tokunaga A, et al. Enhancement of stimulation-induced ERK activation in the spinal dorsal horn and gracile nucleus neurons in rats with peripheral nerve injury. Eur J Neurosci 2004; 19: 884–90.
Yaksh TL, Rudy TA . Chronic catheterization of the subarachnoid space. Physiol Behav 1976; 7: 1032–6.
Bennett GJ, Xie YK . A peripheral mononeuropathy in rat that produces disorder of pain sensation like those seen in man. Pain 1988; 33: 87–107.
Sale EM, Atkinson PG, Sale GJ . The requirement of MAP kinase for the differentiation of fibroblasts to adipocytes, for insulin activation of p90 S6 kinase and for insulin or serum stimulation of DNA synthesis. J EMBO 1995; 14: 674–84.
Chaplan SR, Bach FW, Pogrel JW, Chung JM, Yaksh TJ . Quantitative assessment of tactile allodynia in the rat paw. J Neurosci Methods 1994; 53: 55–63.
Hargreaves K, Dubner R, Brown F, Flores C, Joris J . A new and sensitive method for measuring thermal nociception in cutaneous hyperalgesia. Pain 1988; 32: 77–88.
Cao JL, Zeng YM, Zhang LC, Gu J, Liu HF, Zhou WH, et al. NO mediated the increase of Fos protein and NDMA1AR mRNA expression in the rat spinal cord during morphine withdrawal. Acta Pharmacol Sin 2001; 22: 505–11.
Cao JL, Ding HL, Zhang LC, Duan SM, Zeng YM . Pretreatment with midazolam suppresses morphine withdrawal response in mice and rats. Acta Pharmacol Sin 2002; 23: 685–90.
Aley KO, Levine JD . Role of protein kinase A in the maintenance of inflammatory pain. J Neurosci 1999; 19: 2181–6.
Fang L, Wu J, Lin Q, Willis WD . Calcium-calmodulin-dependent protein kinase II contributes to spinal cord central sensitization. J Neurosci 2002; 22: 4196–204.
Zimmermann M . Pathobiology of neuropathic pain. Eur J Pharmacol 2001; 429: 23–37.
Hu HJ, Glauner KS, Gereau RW IV . ERK integrates PKA and PKC signaling in superficial dorsal horn neurons. I. Modulation of A-type K+ currents. J Neurophysiol 2003; 90: 1671–9.
Hu HJ, Gereau RW IV . ERK integrates PKA and PKC signaling in superficial dorsal horn neurons. II. Modulation of neuronal excitability. J Neurophysiol 2003; 90: 1680–8.
Adams JP, Anderson AE, Varga AW, Deneley KT, Cook RG, Pfaffinger PJ, et al. The A-type potassium channel KV4.2 is a substrate for the mitogen-activated protein kinase ERK. J Neurochem 2000; 75: 2277–87.
Lewin MR, Walters ET . Cyclic GMP pathway is critical for inducing long-term sensitization of nociceptive sensory neurons. Nat Neurosci 1999: 2: 18–23.
Miletic G, Pankratz MT, Miletic V . Increases in the phosphorylation of cyclic AMP response element binding protein (CREB) and decreases in the content of calcineurin accompany thermal hyperalgesia following chronic constriction injury in rats. Pain 2002; 99: 493–500.
White DM, Walker S, Brenneman DE, Gozes I . CREB contributes to the increased neurite outgrowth of sensory neurons induced by vasoactive intestinal polypeptide and activity-dependent neurotrophic factor. Brain Res 2000; 868: 31–8.
Ji RR, Rupp F . Phosphorylation of transcription factor CREB in rat spinal cord after formalin-induced hyperalgesia: relationship to c-fos induction. J Neurosci 1997; 17: 1776–85.
Messersmith DJ, Kim DJ, Iadarola MJ . Transcription factor regulation of prodynorphin gene expression following rat hindpaw inflammation. Mol Brain Res 1998; 53: 260–9.
Solodkin A, Traub RJ, Gebhart GF . Unilateral hindpaw inflammation produces a bilateral increase in NADPH-diaphorase histochemical staining in the rat lumbar spinal cord. Neuroscience 1992; 51: 495–9.
Milligan ED, Twining C, Chacur M, Biedenkapp J, O'Connor K, Poole S, et al. Spinal glia and proinflammatory cytokines mediate mirror-image neuropathic pain in rats. J Neurosci 2003; 23: 1026–40.
Author information
Authors and Affiliations
Corresponding author
Additional information
Project supported by an Open Project Grant from the Jiangsu Province Key Laboratory of Anesthesiology (No KSJ03081) and in part supported by the National Natural Science Foundation of China (No 30200267).
Rights and permissions
About this article
Cite this article
Song, Xs., Cao, Jl., Xu, Yb. et al. Activation of ERK/CREB pathway in spinal cord contributes to chronic constrictive injury-induced neuropathic pain in rats. Acta Pharmacol Sin 26, 789–798 (2005). https://doi.org/10.1111/j.1745-7254.2005.00123.x
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1111/j.1745-7254.2005.00123.x
Keywords
This article is cited by
-
Sex differences in the inflammatory response of the mouse DRG and its connection to pain in experimental autoimmune encephalomyelitis
Scientific Reports (2022)
-
Identification of Biomarkers Related to Neuropathic Pain Induced by Peripheral Nerve Injury
Journal of Molecular Neuroscience (2019)
-
MicroRNA-211-5p Enhances Analgesic Effect of Dexmedetomidine on Inflammatory Visceral Pain in Rats by Suppressing ERK Signaling
Journal of Molecular Neuroscience (2019)
-
The Central Analgesic Mechanism of YM-58483 in Attenuating Neuropathic Pain in Rats
Cellular and Molecular Neurobiology (2016)
-
Endogenous CBS–H2S Pathway Contributes to the Development of CCI-Induced Neuropathic Pain
Neurochemical Research (2016)