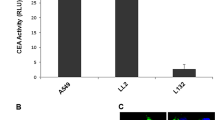
Abstract
Treatment of advanced lung cancer is one of the major challenges in current medicine because of the high morbidity and mortality of the disease. Advanced stage lung cancer is refractory to conventional therapies and has an extremely poor prognosis. Thus, new therapeutic approaches are needed. Lung tumor formation depends on angiogenesis in which the vascular endothelial growth factor (VEGF) produced by cancer cells plays a pivotal role. Neutralizing VEGF with a soluble VEGF receptor suppresses tumor growth; however, the anticancer effect with this therapy is weakened after the intratumoral vascular network is completed. In this study, we turned the expression of VEGF by tumors to therapeutic advantage using a conditionally replication-competent adenovirus (CRAd) in which the expression of E1 is controlled by the human VEGF promoter. This virus achieved good levels of viral replication in lung cancer cells and induced a substantial anticancer effect in vitro and in vivo. As a further enhancement, the cancer cell killing effect was improved with tropism modification of the virus to express the knob domain of Ad3, which improved infectivity for cancer cells. These VEGF promoter-based CRAds also showed a significant cell killing effect for various types of cancer lines other than lung cancer. Conversely, the VEGF promoter has low activity in normal tissues, and the CRAd caused no damage to normal bronchial epithelial cells. Since tumor-associated angiogenesis via VEGF signalling is common in many types of cancers, these CRAds may be applicable to a wide range of tumors. We concluded that VEGF promoter-based CRAds have the potential to be an effective strategy for cancer treatment.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout









Similar content being viewed by others
Abbreviations
- NSCLC:
-
non-small cell lung cancer
- IL-8:
-
interleukin-8
- VEGF:
-
vascular endothelial growth factor
- CAR:
-
coxsackie and adenovirus receptor
- Ad5:
-
serotype 5 adenovirus
- Ad3:
-
serotype 3 adenovirus
- Ad5/3:
-
Ad5 containing a chimeric fiber protein possessing the Ad3 knob
- CRAd:
-
conditionally replicative adenovirus
- E1:
-
early region 1
- E4:
-
early region 4
- RT-PCR:
-
revers transcription-PCR
- CMV:
-
cytomegalovirus
- CsCl:
-
cesium chloride
- VP:
-
viral particle
- PFU:
-
plaque forming unit
References
Levi F, Lucchini F, Negri E, La Vecchia C . Worldwide patterns of cancer mortality, 1990–1994. Eur J Cancer Prev 1999; 8: 381–400.
Folkman J . What is the evidence that tumors are angiogenesis dependent? J Natl Cancer Inst 1990; 82: 4–6.
Liotta LA, Steeg PS, Stetler-Stevenson WG . Cancer metastasis and angiogenesis: an imbalance of positive and negative regulation. Cell 1991; 64: 327–336.
Ohta Y, Endo Y, Tanaka M, Shimizu J, Oda M, Hayashi Y et al. Significance of vascular endothelial growth factor messenger RNA expression in primary lung cancer. Clin Cancer Res 1996; 2: 1411–1416.
Fontanini G, Vignati S, Boldrini L, Chine S, Silvestri V, Lucchi M et al. Vascular endothelial growth factor is associated with neovascularization and influences progression of non-small cell lung carcinoma. Clin Cancer Res 1997; 3: 861–865.
Inoshima N, Nakanishi Y, Minami T, Izumi M, Takayama K, Yoshino I et al. The influence of dendritic cell infiltration and vascular endothelial growth factor expression on the prognosis of non-small cell lung cancer. Clin Cancer Res 8: 3480–3486.
Goldman CK, Kendall RL, Cabrera G, Soroceanu L, Heike Y, Gillespie GY et al. Paracrine expression of a native soluble vascular endothelial growth factor receptor inhibits tumor growth, metastasis, and mortality rate. Proc Natl Acad Sci USA 1998; 95: 8795–8800.
Takayama K, Ueno H, Nakanishi Y, Sakamoto T, Inoue K, Shimizu K et al. Suppression of tumor angiogenesis and growth by gene transfer of a soluble form of vascular endothelial growth factor receptor into a remote organ. Cancer Res 2000; 60: 2169–2177.
Curiel DT . The development of conditionally replicative adenoviruses for cancer therapy. Clin Cancer Res 2000; 6: 3395–3399.
Bischoff JR, Kirn DH, Williams A, Heise C, Horn S, Muna M et al. An adenovirus mutant that replicates selectively in p53-deficient human tumor cells. Science 1996; 274: 373–376.
Fueyo J, Comez-Manzano C, Alemany R, Lee PSY, McDonell TJ, Mitlianga P et al. A mutant oncolytic adenovirus targeting the Rb pathway produces anti-glioma effect in vivo. Oncogene 1999; 19: 1–11.
Hallenbeck PL, Chang Y-N, Hay C, Golightly D, Stewart D, Lin J et al. A novel tumor-specific replication-restricted adenoviral vector for gene therapy of hepatocellular carcinoma. Hum Gene Ther 1999; 10: 1721–1733.
Rodriguez R, Schuur ER, Lim HY, Henderson GA, Simons JW, Henderson DR . Prostate attenuated replacation competent adenovirus (ARCA) CV706: a selective cytotoxic for prostate-specific antigen-positive prostate cancer cells. Cancer Res 1997; 57: 2559–2563.
Matsubara S, Wada Y, Gardner TA, Egawa M, Park M-S, Hsieh C-L et al. A conditional replication-competent adenoviral vector, Ad-OC-E1a, to cotarget prostate cancer and bone stroma in an experimental model of androgen-independent prostate cancer bone metastasis. Cancer Res 2001; 61: 6012–6019.
Kurihara T, Brough DE, Kovesdi I, Kufe DW . Selectivity of a replication-competent adenovirus for human breast carcinoma cells expressing the MUC1 antigen. J Clin Invest 2000; 106: 763–771.
Adachi Y, Reynolds PN, Yamamoto M, Wang M, Takayama K, Matsubara S et al. A midkine promoter-based conditionally replicative adenovirus for treatment of pediatric solid tumors and bone marrow tumor purging. Cancer Res 2001; 61: 7882–7888.
Nettelbeck DM, Rivera AA, Balague C, Alemany R, Curiel DT . Novel oncolytic adenoviruses targeted to melanoma: specific viral replication and cytolysis by expression of E1A mutants from the tyrosinase enhancer/promoter. Cancer Res 2002; 62: 4663–4670.
DeWeese TL, Van der Poel H, Li S, Mikhak B, Drew R, Goemann M et al. A phase I trial of CV706, a replication-competent, PSA selective oncolytic adenovirus, for the treatment of locally recurrent prostate cancer following radiation therapy. Cancer Res 2001; 61: 7464–7472.
Nemunaitis J, Ganly I, Khuri F, Arseneau J, Kuhn J, McCarty T et al. Selective replication and oncolysis in p53 mutant tumors with ONYX-015, an E1B-55KD gene-deleted adenovirus, in patients with advanced head and neck cancer: a Phase II trial. Cancer Res 2000; 60: 6359–6366.
He T-C, Zhou S, da Costa LT, Yu J, Kinzler KW, Vogelstein B . A simplified system for generating reombinant adenoviruses. Proc Natl Acad Sci USA 1998; 95: 2509–2514.
Forsythe JA, Jiang B-H, Iyer NV, Agani E, Leung SW, Koos RD et al. Activation of vascular endothelial growth factor gene transcription by hypoxia-inducible factor I. Mol Cell Biol 1996; 16: 4604–4613.
Krasnykh VN, Mikheeva GV, Douglas JT, Curiel DT . Generation of recombinant adenovirus vectors with modified fibers for altering viral tropism. J Virol 1996; 70: 6839–6846.
Takayama K, Ueno H, Pei X-H, Nakanishi Y, Yatsunami J, Hara N . The levels of integrin αvβ5 may predict the susceptibility to adenovirus-mediated gene transfer in human lung cancers. Gene Therapy 1998; 5: 361–368.
Maizel JVJ, White O, Scharff MD . The polypeptides of adenovirus. I. Evidence for multiple protein components in the virion and a comparison of types 2, 7A, and 12. Virology 1968; 36: 115–125.
Mittereder N, March KL, Trapnell BC . Evaluation of the concentration and bioactivity of adenovirus vectors for gene therapy. J Virol 1996; 70: 7498–7509.
Chartier C, Degryse E, Gantzer M, Dieterle A, Pavirani A, Mehtali M . Efficient generation of recombinant adenovirus vectors by homologous recombination in Escherichia coli. J Virol 1996; 70: 4805–4810.
Kaliberov S, Buchsbaum D, Gillespie G, Curiel DT, Arafat W, Carpenter M et al. Adenovirus-mediated transfer of BAX driven by the vascular endothelial growth factor promoter induces apoptosis in lung cancer cells. Mol Ther 2002; 6: 190–198.
Suzuki K, Fueyo J, Krasnykh V, Reynolds PN, Curiel DT, Alemany R . A conditionally replicative adenovirus with enhanced infectivity shows improved oncolytic potency. Clin Cancer Res 2001; 7: 120–126.
Kanerva A, Mikheeva GV, Krasnykh V, Coolidge CJ, Lam JT, Mahasreshti PJ et al. Targeting adenovirus to the serotype 3 receptor increases gene transfer efficiency to ovarian cancer cells. Clin Cancer Res 2002; 8: 275–280.
Haviv YS, Blackwell JL, Kanerva A, Nagi P, Krasnykh V, Dmitriev I et al. Adenoviral gene therapy for renal cancer requires retargeting to alternative cellular receptors. Cancer Res 2002; 62: 4273–4281.
Hermiston T . Gene delivery from replication-selective viruses: arming guided missiles in the war against cancer. J Clin Invest 2000; 105: 1169–1172.
Heise CC, Williams A, Olesch J, Kirn DH . Efficacy of a replication-competent adenovirus (ONYX-015) following intratumoral injection: intratumoral spread and distribution effects. Cancer Gene Ther 1999; 6: 499–504.
van der Eb MM, Cramer SJ, Vergouwe Y, Schagen FHE, van Krieken JHM, van der Eb AJ et al. Severe hepatic dysfunction after adenovirus-mediated transfer of the herpes simplex virus thymidine kinase gene and ganciclovir administration. Gene Therapy 1998; 5: 451–458.
Rosen LS . Clinical experience with angiogenesis signaling inhibitors: focus on vascular endothelial growth factor (VEGF) blockers. Cancer Control 2002; 9: 36–44.
Ferrara N, Chen H, Davis-Smyth T, Gerber HP, Nguyen TN, Peers D et al. Vascular endothelial growth factor is essential for corpus luteum angiogenesis. Nat Med 1998; 4: 336–340.
Yuan A, Yu C-J, Chen W-J, Lin F-Y, Kuo S-H, Luh K-T et al. Correlation of total VEGF mRNA and protein expression with histologic type, tumor angiogenesis, patient survival and timing of relapse in non-small-cell lung cancer. Int J Cancer 2000; 89: 475–483.
Mall JW, Schwenk W, Philipp AW, Meyer-Kipker C, Mall W, Muller J et al. Serum vascular endothelial growth factor levels correlate better with tumor stage in small cell lung cancer than albumin, neuron-specific enolase or lactate dehydrogenase. Respirology 2002; 7: 99–102.
Shweiki D, Itin A, Soffer D, Keshet E . Vascular endothelial growth factor induced by hypoxia may mediate hypoxia-initiated angiogenesis. Nature 1992; 359: 843–845.
Wiesener MS, Munchenhagen PM, Berger I, Morgan NV, Roigas J, Schwiertz A et al. Constitutive activation of hypoxia-inducible genes related to overexpression of hypoxia-inducible factor-1 alpha in clear cell renal carcinoma. Cancer Res 2001; 61: 5215–5222.
Haviv YS, Takayama K, Glasgow JN, Blackwell JL, Wang M, Lei X et al. A model system for the design of armed replicating adenoviruses using p53 as a candidate transgene. Mol Cancer Ther 2002; 1: 321–328.
Miller CR, Buchsbaum DJ, Reynolds PN, Douglas JT, Gillespie GY, Mayo MS et al. Differential susceptibility of primary and established human glioma cells to adenovirus infection: targeting via the epidermal growth factor receptor achieves fiber receptor-independent gene transfer. Cancer Res 1998; 58: 5738–5748.
Hemmi S, Geertsen R, Mezzacasa A, Peter I, Dummer R . The presence of human coxsackievirus and adenovirus receptor is associated with efficient adenovirus-mediated transgene expression in human melanoma cell cultures. Hum Gene Ther 1998; 9: 2363–2373.
Cripe TP, Dunphy EJ, Holub A, Vasi NH, Mahller YY, Collins MH et al. Fiber knob modifications overcome low, heterogeneous expression of the coxsackievirus-adenovirus receptor that limits adenovirus gene transfer and oncolysis for human rhabdomyosarcoma cells. Cancer Res 2001; 61: 2951–2960.
Okegawa T, Li Y, Pong RC, Bergelson JM, Zhou J, Hsieh JT . The dual impact of coxsackie and adenovirus receptor expression on human prostate cancer gene therapy. Cancer Res 2000; 60: 5031–5036.
Lam JT, Kanerva A, Bauershcmitz GJ, Takayama K, Suzuki K, Yamamoto M et al. Inter-patient variation in efficacy of five oncolytic adenovirus candidates for ovarian cancer therapy. J Gene Med 2004; 6: 1333–1342.
Acknowledgements
We thank Professor Semenza (John's Hopkins University) for providing us with the human VEGF promoter. We also thank Dirk M Nettelbeck, Joel N Glasgow, Akiko Harada and Nobuyuki Hara (Kyushu university, Fukuoka, Japan) for their excellent technical support and expert advice. This work was supported by National Cancer Institute Grant CA83821, The CapCURE Foundation, The Lustgarten Foundation, United States Department of Defense Grant 991018 and the American Cancer Society.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Takayama, K., Reynolds, P., Adachi, Y. et al. Vascular endothelial growth factor promoter-based conditionally replicative adenoviruses for pan-carcinoma application. Cancer Gene Ther 14, 105–116 (2007). https://doi.org/10.1038/sj.cgt.7700991
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/sj.cgt.7700991
Keywords
This article is cited by
-
Survivin a radiogenetic promoter for glioblastoma viral gene therapy independently from CArG motifs
Clinical and Translational Medicine (2017)
-
Addition of bevacizumab enhances antitumor activity of erlotinib against non-small cell lung cancer xenografts depending on VEGF expression
Cancer Chemotherapy and Pharmacology (2014)