Abstract
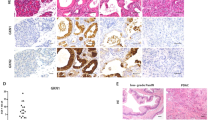
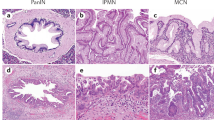
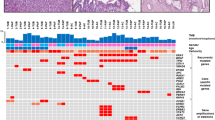
Alterations of K-ras, p53, p16 and DPC4/Smad4 characterize pancreatic ductal cancer (PDC). Reports of inactivation of these latter two genes in pancreatic endocrine tumours (PET) suggest that common molecular pathways are involved in the tumorigenesis of pancreatic exocrine and endocrine epithelia. We characterized 112 primary pancreatic tumours for alterations in p16 and DPC4 and immunohistochemical expression of DPC4. The cases included 34 PDC, 10 intraductal papillary-mucinous tumours (IPMT), 6 acinar carcinomas (PAC), 5 solid-pseudopapillary tumours (SPT), 16 ampulla of Vater cancers (AVC) and 41 PET. All tumours were also presently or previously analysed for K-ras and p53 mutations and allelic loss at 9p, 17p and 18q. Alterations in K-ras, p53, p16 and DPC4 were found in 82%, 53%, 38% and 9% of PDC, respectively and in 47%, 60%, 25% and 6% of AVC. Alterations in these genes were virtually absent in PET, PAC or SPT, while in IPMT only K-ras mutations were present (30%). Positive immunostaining confirmed the absence of DPC4 alterations in all IPMT, SPT, PAC and PET, while 47% of PDC and 38% of AVC were immunonegative. These data suggest that pancreatic exocrine and endocrine tumourigenesis involves different genetic targets and that among exocrine pancreatic neoplasms, only ductal and ampullary cancers share common molecular events. © 2001 Cancer Research Campaign http://www.bjcancer.com
Similar content being viewed by others
Article PDF
Change history
16 November 2011
This paper was modified 12 months after initial publication to switch to Creative Commons licence terms, as noted at publication
References
Achille A, Biasi MO, Zamboni G, Bogina G, Magalini AR, Pederzoli P, Perucho M and Scarpa A (1996a) Chromosome 7q allelic losses in pancreatic carcinoma. Cancer Res 56: 3808–3813
Achille A, Scupoli MT, Magalini AR, Zamboni G, Romanelli MG, Orlandini S, Biasi MO, Lemoine NR, Accolla RS and Scarpa A (1996b) APC gene mutations and allelic losses in sporadic ampullary tumours: evidence of genetic difference from tumours associated with familial adenomatous polyposis. Int J Cancer 68: 305–312
Achille A, Biasi MO, Zamboni G, Bogina G, Iacono C, Talamini G, Capella G and Scarpa A (1997) Cancers of the papilla of Vater: mutator phenotype is associated with good prognosis. Clin Cancer Res 3: 1841–1847
Achille A, Baron A, Zamboni G, Di Pace C, Orlandini S and Scarpa A (1998) Chromosome 5 allelic losses are early events in tumours of the papilla of Vater and occur at sites similar to those of gastric cancer. Br J Cancer 78: 1653–1660
Aitken J, Welch J, Duffy D, Milligan A, Green A, Martin N and Hayward N (1999) CDKN2A variants in a population-based sample of Queensland families with melanoma. J Natl Cancer Inst 91: 446–452
Almoguera C, Shibata D, Forrester K, Martin J, Arnheim N and Perucho M (1988) Most human carcinomas of the exocrine pancreas contain mutant c-K-ras genes. Cell 53: 549–554
Barton CM, Staddon SL, Hughes CM, Hall PA, O'Sullivan C, Klöppel G, Theis B, Russell C, Neoptolemos J, Williamson RCN, Lane D and Lemoine NR (1991) Abnormalities of the p53 tumour suppressor gene in human pancreatic cancer. Br J Cancer 64: 1076–1082
Bartsch D, Bastian D, Barth P, Schudy A, Nies C, Kisker O, Wagner HJ and Rothmund M (1998) K-ras oncogene mutations indicate malignancy in cystic tumors of the pancreas. Ann Surg 228: 79–86
Bartsch D, Barth P, Bastian D, Ramaswamy A, Gerdes B, Chaloupka B, Deiss Y, Simon B and Schudy A (1999a) Higher frequency of DPC4/Smad4 alterations in pancreatic cancer cell lines than in primary pancreatic adenocarcinomas. Cancer Lett 139: 43–49
Bartsch D, Hahn SA, Danichevski KD, Ramaswamy A, Bastian D, Galehdari H, Barth P, Schmiegel W, Simon B and Rothmund M (1996b) Mutations of the DPC4/Smad4 gene in neuroendocrine pancreatic tumors. Oncogene 18: 2367–2371
Beghelli S, Pelosi G, Zamboni G, Falconi M, Iacono C, Bordi C and Scarpa A (1998) Pancreatic endocrine tumours: evidence for a tumour suppressor pathogenesis and for a tumour suppressor gene on chromosome 17p. J Pathol 186: 41–50
Caldas C, Hahn SA, Costa LTd, Redston MS, Schutte M, Seymour AB, Weinstein CL, Hruban RH, Yeo CJ and Kern SE (1994) Frequent somatic mutations and homozygous deletions of the p16 (MTS1) gene in pancreatic adenocarcinoma. Nature Genet 8: 27–32
Ebert MP, Hoffmann J, Schneider-Stock R, Kasper HU, Schulz HU, Lippert H, Roessner A and Malfertheiner P (1998) Analysis of K-ras gene mutations in rare pancreatic and ampullary tumours. Eur J Gastroenterol Hepatol 10: 1025–1029
Geradts J, Hruban RH, Schutte M, Kern SE and Maynard R (2000) Immunohistochemical p161NK4a analysis of archival tumors with deletion, hypermethylation, or mutation of the CDKN2/MTS1 gene. A comparison of four commercial antibodies. Appl Immunohistochem Molecul Morphol 8: 71–79
Hahn SA, Schutte M, Hoque AT, Moskaluk CA, da Costa LT, Rozenblum E, Weinstein CL, Fischer A, Yeo CJ, Hruban RH and Kern SE (1996) DPC4, a candidate tumor suppressor gene at human chromosome 18q21.1. Science 271: 350–353
Hahn SA, Bartsch D, Schroers A, Galehdari H, Becker M, Ramaswamy A, Schwarte-Waldhoff I, Maschek H and Schmiegel W (1998) Mutations of the DPC4/Smad4 gene in biliary tract carcinoma. Cancer Res 58: 1124–1126
Herman JG, Graff JR, Myohanen S, Nelkin BD and Baylin SB (1996) Methylation-specific PCR: a novel PCR assay for methylation status of CpG islands. Proc Natl Acad Sci USA 93: 9821–9826
Hoorens A, Lemoine NR, McLellan E, Morohoshi T, Kamisawa T, Heitz PU, Stamm B, Ruschoff J, Wiedenmann B and Kloppel G (1993) Pancreatic acinar cell carcinoma. An analysis of cell lineage markers, p53 expression, and Ki-ras mutation. Am J Pathol 143: 685–698
Hruban RH, van Mansfeld AD, Offerhaus GJ, van Weering DH, Allison DC, Goodman SN, Kensler TW, Bose KK, Cameron JL and Bos JL (1993) K-ras oncogene activation in adenocarcinoma of the human pancreas. A study of 82 carcinomas using a combination of mutant-enriched polymerase chain reaction analysis and allele-specific oligonucleotide hybridization. Am J Pathol 143: 545–554
Huang L, Goodrow TL, Zhang SY, Klein-Szanto AJ, Chang H and Ruggeri BA (1996) Deletion and mutation analyses of the P16/MTS-1 tumor suppressor gene in human ductal pancreatic cancer reveals a higher frequency of abnormalities in tumor-derived cell lines than in primary ductal adenocarcinomas. Cancer Res 56: 1137–1141
Jonson T, Gorunova L, Dawiskiba S, Andren-Sandberg A, Stenman G, ten Dijke P, Johansson B and Hoglund M (1999) Molecular analyses of the 15q and 18q SMAD genes in pancreatic cancer. Genes Chromosomes Cancer 24: 62–71
Kalthoff H, Schmiegel W, Roeder C, Kasche D, Schmidt A, Lauer G, Thiele HG, Honold G, Pantel K and Riethmuller G (1993) p53 and K-RAS alterations in pancreatic epithelial cell lesions. Oncogene 8: 289–298
Klimstra DS, Heffess CS, Oertel JE and Rosai J (1992) Acinar cell carcinoma of the pancreas. A clinicopathologic study of 28 cases. Am J Surg Pathol 16: 815–837
Klöppel G, Solcia E, Longnecker D, Capella C and Sobin L (1996) Histological typing of tumours of the exocrine pancreas. International Histological Classification of Tumours. Springer-Verlag: Berlin
Kondo H, Sugano K, Fukayama N, Hosokawa K, Ohkura H, Ohtsu A, Mukai K and Yoshida S (1997) Detection of K-ras gene mutations at codon 12 in the pancreatic juice of patients with intraductal papillary mucinous tumors of the pancreas. Cancer 79: 900–905
Lam KY and Lo CY (1998) Role of p53 tumor suppressor gene in pancreatic endocrine tumors of Chinese patients. Am J Gastroenterol 93: 1232–1235
Lee WY, Tzeng CC, Chen RM, Tsao CJ, Tseng JY and Jin YT (1997) Papillary cystic tumors of the pancreas: assessment of malignant potential by analysis of progesterone receptor, flow cytometry, and ras oncogene mutation. Anticancer Res 17: 2587–2591
Lemoine NR, Jain S, Hughes CM, Staddon SL, Maillet B, Hall PA and Kloppel G (1992) Ki-ras oncogene activation in preinvasive pancreatic cancer. Gastroenterology 102: 230–236
Moore PS, Rigaud G, Baron A and Scarpa A (2000a) Two novel polymorphisms, c1086T>C and c1798C>T, in the MADH4/DPC4 gene. Hum Mutat (Online) 15: 485–486
Moore PS, Zamboni G, Falconi M, Bassi C and Scarpa A (2000b) A novel germline mutation, P48T, in the CDKN2A/p16 gene in a patient with pancreatic carcinoma. Hum Mutat (Online), in press
Muscarella P, Melvin WS, Fisher WE, Foor J, Ellison EC, Herman JG, Schirmer WJ, Hitchcock CL, DeYoung BR and Weghorst CM (1998) Genetic alterations in gastrinomas and nonfunctioning pancreatic neuroendocrine tumors: an analysis of p16/MTS1 tumor suppressor gene inactivation. Cancer Res 58: 237–240
Naumann M, Savitskaia N, Eilert C, Schramm A, Kalthoff H and Schmiegel W (1996) Frequent codeletion of p16/MTS1 and p15/MTS2 and genetic alterations in p16/MTS1 in pancreatic tumors. Gastroenterology 110: 1215–1224
Nielsen GP, Stemmer-Rachamimov AO, Shaw J, Roy JE, Koh J and Louis DN (1999) Immunohistochemical survey of p16INK4A expression in normal human adult and infant tissues. Lab Invest 79: 1137–1143
Pellegata NS, Sessa F, Renault B, Bonato M, Leone BE, Solcia E and Ranzani GN (1994) K-ras and p53 gene mutations in pancreatic cancer: ductal and nonductal tumors progress through different genetic lesions. Cancer Res 54: 1556–1560
Redston MS, Caldas C, Seymour AB, Hruban RH, da Costa L, Yeo CJ and Kern SE (1994) p53 mutations in pancreatic carcinoma and evidence of common involvement of homocopolymer tracts in DNA microdeletions. Cancer Res 54: 3025–3033
Reymond A and Brent R (1995) p16 proteins from melanoma-prone families are deficient in binding to Cdk4. Oncogene 11: 1173–1178
Rozenblum E, Schutte M, Goggins M, Hahn SA, Panzer S, Zahurak M, Goodman SN, Sohn TA, Hruban RH, Yeo CJ and Kern SE (1997) Tumor-suppressive pathways in pancreatic carcinoma. Cancer Res 57: 1731–1734
Satoh K, Shimosegawa T, Moriizumi S, Koizumi M and Toyota T (1996) K-ras mutation and p53 protein accumulation in intraductal mucin-hypersecreting neoplasms of the pancreas. Pancreas 12: 362–368
Scarpa A, Capelli P, Mukai K, Zamboni G, Oda T, Iacono C and Hirohashi S (1993a) Pancreatic adenocarcinomas frequently show p53 gene mutations. Am J Pathol 142: 1534–1543
Scarpa A, Capelli P, Zamboni G, Oda T, Mukai K, Bonetti F, Martignoni G, Iacono C, Serio G and Hirohashi S (1993b) Neoplasia of the ampulla of Vater, Ki-ras and p53 mutations. Am J Pathol 142: 1163–1172
Scarpa A, Capelli P, Villaneuva A, Zamboni G, Lluis F, Accolla R, Mariuzzi G and Capella G (1994a) Pancreatic cancer in Europe: Ki-ras gene mutation pattern shows geographical differences. Int J Cancer 57: 167–171
Scarpa A, Zamboni G, Achille A, Capelli P, Bogina G, Iacono C, Serio G and Accolla RS (1994b) ras-family gene mutations in neoplasia of the ampulla of Vater. Int J Cancer 59: 39–42
Scarpa A, Di Pace C, Talamini G, Iacono C, Falconi M, Achille A, Baron A and Zamboni G (2000) Chromosome 17p loss and prognosis of cancer of papilla of Vater. Gut 46: 842–848
Schutte M, Hruban RH, Geradts J, Maynard R, Hilgers W, Rabindran SK, Moskaluk CA, Hahn SA, Schwarte-Waldhoff I, Schmiegel W, Baylin SB, Kern SE and Herman JG (1997) Abrogation of the Rb/p16 tumor-suppressive pathway in virtually all pancreatic carcinomas. Cancer Res 57: 3126–3130
Sessa F, Solcia E, Capella C, Bonato M, Scarpa A, Zamboni G, Pellegata N, Ranzani G, Rickaert F and Klöppel G (1994) Intraductal papillary-mucinous pancreatic tumors are phenotypically, genetically and behaviorally distinct growths. An analysis of tumor cell phenotype, K-ras and p53 gene mutations. Virchows Archiv 425: 357–367
Solcia E, Capella C and Klöppel G (1997) Tumors of the pancreas. Vol. 20. Atlas of tumor pathology. Armed Forces Institute of Pathology: Washington, DC
Sorio C, Baron A, Orlandini S, Zamboni G, Pederzoli P, Huebner K and Scarpa A (1999) The FHIT gene is expressed in pancreatic ductular cells and is altered in pancreatic cancers. Cancer Res 59: 1308–1314
Terhune PG, Heffess CS and Longnecker DS (1994) Only wild-type c-Ki-ras codons 12, 13, and 61 in human pancreatic acinar cell carcinomas. Mol Carcinog 10: 110–114
Ueki T, Toyota M, Sohn T, Yeo CJ, Issa JP, Hruban RH and Goggins M (2000) Hypermethylation of multiple genes in pancreatic adenocarcinoma. Cancer Res 60: 1835–1839
Villanueva A, Garcia C, Paules AB, Vicente M, Megias M, Reyes G, de Villalonga P, Agell N, Lluis F, Bachs O and Capella G (1998) Disruption of the antiproliferative TGF-beta signaling pathways in human pancreatic cancer cells. Oncogene 17: 1969–1978
Wilentz RE, Geradts J, Maynard R, Offerhaus GJ, Kang M, Goggins M, Yeo CJ, Kern SE and Hruban RH (1998) Inactivation of the p16 (INK4A) tumor-suppressor gene in pancreatic duct lesions: loss of intranuclear expression. Cancer Res 58: 4740–4744
Wilentz RE, Su GH, Dai JL, Sparks AB, Argani P, Sohn TA, Yeo CJ, Kern SE and Hruban RH (2000) Immunohistochemical labeling for dpc4 mirrors genetic status in pancreatic adenocarcinomas: a new marker of DPC4 inactivation. Am J Pathol 156: 37–43
Zamboni G, Bonetti F, Scarpa A, Pelosi G, Doglioni C, Iannucci A, Castelli P, Balercia G, Aldovini D, Bellomi A, Iacono C, Serio G and Mariuzzi G (1993) Expression of progesterone receptors in solid-cystic tumour of the pancreas: a clinicopathological and immunohistochemical study of ten cases. Virchows Archiv A 423: 425–431
Z'Graggen K, Rivera JA, Compton CC, Pins M, Werner J, Fernandez-del Castillo C, Rattner DW, Lewandrowski KB, Rustgi AK and Warshaw AL (1997) Prevalence of activating K-ras mutations in the evolutionary stages of neoplasia in intraductal papillary mucinous tumors of the pancreas. Ann Surg 226: 491–498
Zhang SY, Klein-Szanto AJ, Sauter ER, Shafarenko M, Mitsunaga S, Nobori T, Carson DA, Ridge JA and Goodrow TL (1994) Higher frequency of alterations in the p16/CDKN2 gene in squamous cell carcinoma cell lines than in primary tumors of the head and neck. Cancer Res 54: 5050–5053
Author information
Authors and Affiliations
Rights and permissions
From twelve months after its original publication, this work is licensed under the Creative Commons Attribution-NonCommercial-Share Alike 3.0 Unported License. To view a copy of this license, visit http://creativecommons.org/licenses/by-nc-sa/3.0/
About this article
Cite this article
Moore, P., Orlandini, S., Zamboni, G. et al. Pancreatic tumours: molecular pathways implicated in ductal cancer are involved in ampullary but not in exocrine nonductal or endocrine tumorigenesis. Br J Cancer 84, 253–262 (2001). https://doi.org/10.1054/bjoc.2000.1567
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1054/bjoc.2000.1567
Keywords
This article is cited by
-
Pancreatic acinar cell carcinoma with a ductal adenocarcinoma component: a case report and analysis of the histogenesis of the tumor
World Journal of Surgical Oncology (2020)
-
SMAD4 Y353C promotes the progression of PDAC
BMC Cancer (2019)
-
Genome-wide genetic and epigenetic analyses of pancreatic acinar cell carcinomas reveal aberrations in genome stability
Nature Communications (2017)
-
E-cadherin-negative acinar cell carcinoma of the pancreas: report of a case showing a solid pseudopapillary growth pattern
Medical Molecular Morphology (2016)