Abstract
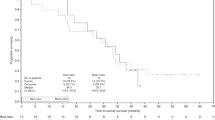
Mycobacterial preparations have been used with limited success against cancer apart from superficial bladder cancer. Recently, a therapeutic vaccine derived from Mycobacterium vaccae has been given to patients with prostate cancer and melanoma indicating a possible beneficial effect on disease activity in such patients. We have recently initiated a series of randomized studies to test the feasibility and toxicity of combining a preparation of heat-killed Mycobacterium vaccae (designated SRL172) with a multidrug chemotherapy regimen to treat patients with inoperable non-small cell lung cancer (NSCLC) and mesothelioma. 28 evaluable patients with previously untreated symptomatic NSCLC and mesothelioma were randomized to receive either 3 weekly intravenous combination chemotherapy alone, or chemotherapy given with monthly intra-dermal injections of SRL172. Safety and tolerability were scored by common toxicity criteria and efficacy was evaluated by survival of patients and by tumour response assessed by CT scanning. The toxicity of chemotherapy was similar in the two groups. SRL172 caused mild inflammation at the injection site. In the group of patients randomized to receive chemotherapy combined with SRL172, there was a trend towards improved response rate (54% vs. 33%) with more patients in the combined arm receiving radical surgery and radiotherapy, improved median survival (9.7 months vs. 7.5 months) and improved 1 year survival (42% vs. 18%). SRL172 appeared to improve sleep (P = 0.08) and improved appetite (P = 0.01). There was no detectable change in serum cytokine levels for gamma-interferon and TNF-α before and after treatment. In patients with NSCLC and mesothelioma, there may be a beneficial interaction when chemotherapy is administered in combination with SRL172. Confirmation of this effect and further investigation is underway in a randomized phase III trial and in laboratory models. © 2000 Cancer Research Campaign
Similar content being viewed by others
Article PDF
Change history
16 November 2011
This paper was modified 12 months after initial publication to switch to Creative Commons licence terms, as noted at publication
References
Al-Moundhri M, O'Brien M and Souberbielle BE (1998) Immunotherapy and lung cancer. Br J Cancer 78: 282–288
Ellis PA, Smith IE and Hardy JR (1995) Symptom relief with MVP (mitomycin C, vinblastine and cisplatin) chemotherapy in advanced non-small-cell lung cancer. Br J Cancer 71: 366–370
Ellis PA, Smith IE, McCarthy K, Detre S, Salter J and Dowsett M (1997) Preoperative chemotherapy induces apoptosis in realy breast cancer. Lancet 349: 849
Ferrarini M, Heltai S, Zocchi MR and Rugarli C (1992) Unusual expression of and localisation heat-shock proteins in human tumour cells. Int J Cancer 51: 613–619
Grange JM, Stanford JL and Rook GAW (1995) Tuberculosis and cancer: parallels in host responses and therapeutic approaches?. Lancet 345: 1350–1352
Hanna MG and Key ME (1982) Immunotherapy of metastases enhances subsequent chemotherapy. Science 217: 367–369
Hrouda D, Baban B, Dunsmuir WD, Kirby RS and Dalgleish AG (1998) Immunotherapy of advanced prostate cancer: a phase I/II trial using Mycobacterium vaccae (SRL172). Br J Urol 82: 568–573
Maraveyas A, Baban B, Kennard D, Rook GAW, Westby M, Grange JM, Lydiard P, Stanford JL, Jones M, Selby P and Dalgleish AGD (1999) Possible improved survival of patients with stage IV AJCC melanoma receiving SRL172 immunotherapy: correlation with induction of increased levels of intracellular interleukin-2 in peripheral blood lymphocytes. Ann Oncol 10: 817–824
Mitchell MS (1992) Chemotherapy in combination with biomodulation: a 5 year experience with cyclophosphamide and IL-2. Semin Oncol 19: 80–87
Pellegrine P, Berhelia AM and Del Beato T (1996) Dysregulation of Th1 and Th2 subsets of CD4+ cells in peripheral blood of colorectal patients and involvement in cancer establishment and progression. Cancer Immunol Immunother 42: 1–8
Webb A and O'Brien MER (1998) Where do we go with new expensive treatments in NSCLC?. Br J Cancer 78: 159–162
Author information
Authors and Affiliations
Rights and permissions
From twelve months after its original publication, this work is licensed under the Creative Commons Attribution-NonCommercial-Share Alike 3.0 Unported License. To view a copy of this license, visit http://creativecommons.org/licenses/by-nc-sa/3.0/
About this article
Cite this article
O'Brien, M., Saini, A., Smith, I. et al. A randomized phase II study of SRL172 (Mycobacterium vaccae) combined with chemotherapy in patients with advanced inoperable non-small-cell lung cancer and mesothelioma. Br J Cancer 83, 853–857 (2000). https://doi.org/10.1054/bjoc.2000.1401
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1054/bjoc.2000.1401
Keywords
This article is cited by
-
Old Friends, immunoregulation, and stress resilience
Pflügers Archiv - European Journal of Physiology (2019)
-
Mycobacteria activate γδ T-cell anti-tumour responses via cytokines from type 1 myeloid dendritic cells: a mechanism of action for cancer immunotherapy
Cancer Immunology, Immunotherapy (2012)
-
Factors Affecting Efficacy and Safety of Add-On Combination Chemotherapy for Non-Small-Cell Lung Cancer: A Literature-Based Pooled Analysis of Randomized Controlled Trials
Lung (2012)
-
The production of a new extracellular putative long-chain saturated polyester by smooth variants of Mycobacterium vaccae interferes with Th1-cytokine production
Antonie van Leeuwenhoek (2006)