Abstract
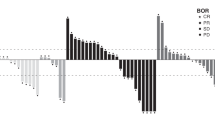
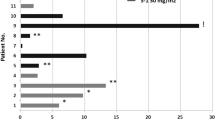
This study set out to evaluate, in patients with metastatic colorectal carcinoma, the efficacy and toxicity of S-1, which contains tegafur, 5-chloro-2,4-dihydroxypyridine (CDHP) and potassium oxonate, based on a biochemical modulation of 5-fluorouracil (5-FU) targeted at inhibition of dihydropyrimidine dehydrogenase (DPD). Sixty-three patients with measurable metastatic colorectal carcinoma were enrolled into the study. None of the patients had received prior chemotherapy except for adjuvant setting. S-1 was administered orally twice daily at a standard dose of 80 mg m–2day–1for 28 days followed by a 14-day rest. This agent is continued until disease progression, unaccepted toxicity, or patient refusal. Twenty-two (35%) of the 62 eligible patients achieved PR with a 95% confidence interval of 25–48%. Five of the 10 patients with a history of adjuvant chemotherapy achieved partial remission. The median survival time was 12 months. Major adverse reactions included myelosuppressive and gastrointestinal toxicities, though their incidence of grade 3 or 4 being 13% in neutropenia and less than 10% in the others. None of the 53 patients treated as outpatients required hospitalization due to adverse reactions: These results suggest that S-1 achieves similar responses to those of infusional 5-FU plus leucovorin and shows the potential of another biochemical modulation with easily manageable toxicity. © 2000 Cancer Research Campaign
Similar content being viewed by others
Article PDF
Change history
16 November 2011
This paper was modified 12 months after initial publication to switch to Creative Commons licence terms, as noted at publication
References
Advanced Colorectal Cancer Meta-Analysis Project (1992) Modulation of fluorouracil by leucovorin in patients with advanced colorectal cancer. J Clin Oncol 10: 896–903
Baccanari DP, Davis ST, Knick VC and Spector T (1993) 776C85: Effects on the pharmacokinetics and antitumor activity of 5-fluorouracil. Proc Natl Acad Sci USA 90: 11064–11068
DeMario MD and Ratain MJ (1998) Oral chemotherapy: rationale and future directions. J Clin Oncol 16: 2557–2567
Diasio RB, Beavers TL and Carpenter JT (1988) Familial deficiency of dihydropyrimidine dehydrogenase. J Clin Invest 81: 47–51
Etienne MC, Cheradame S, Fischel JL, Formento P, Dassonville O, Renee N, Schneider M, Thyss A, Demard F and Milano G (1995) Response to fluorouracil therapy in cancer patients: The role of tumoral dihydropyrimidine dehydrogenase activity. J Clin Oncol 13: 1663–1670
Harris BE, Song R, Soong SJ and Diasio RB (1990) Relationship between dihydropyrimidine dehydrogenase activity and plasma 5-fluorouracil levels with evidence for circadian variation of enzyme activity and plasma drug levels in cancer patients receiving 5-fluorouracil by protracted continuous infusion. Cancer Res 50: 197–201
Heggie GD, Sommadossi JP, Cross DS, Huster WJ and Diasio RB (1987) Clinical pharmacokinetics of 5-fluorouracil and its metabolites in plasma, urine, and bile. Cancer Res 47: 2203–2206
Houghton JA, Houghton PJ and Wooten RS (1979) Mechanism of induction of gastrointestinal toxicity in the mouse by 5-fluorouracil, 5-fluorouridine, and 5-fluoro-2’-deoxyuridine. Cancer Res 39: 2406–2413
Ikenaka K, Shirasaka T and Kitano S (1979) Effect of uracil on metabolism o 5-fluorouracil in vitro. Jpn J Cancer Res 70: 353–359
Japanese Research Society for Cancer of Colon and Rectum (1994). General rules for clinical and pathological studies on cancer of the colon, rectum and anus, Kanehara Syuppan: Tokyo
Japan Society for Cancer Therapy (1993) Criteria for the evaluation of the clinical effects of solid cancer chemotherapy. J Jpn Soc Cancer Ther 28: 101–130
Levi F, Misset JL, Brienza S, Adam R, Metzger G, Itzakhi M, Caussanel JP, Kunstlinger F, Lecouturier S, Descorps-Declere A, Jasmin C, Bismuth H and Reinberg A (1992) A chronopharmacologic phase II clinical trial with 5-fluorouracil, folinic acid, and oxaliplatin using an ambulatory multichannel programmable pump. Cancer 69: 893–900
Leichman CG, Fleming TR, Muggia FM, Tangen CM, Ardalan B, Doroshow JH, Meyers FJ, Holcombe RF, Weiss GR, Mangalik A and Macdonald JS (1995) Phase II study of fluorouracil and its modulation in advanced colorectal cancer: A Southwest Oncology Group Study. J Clin Oncol 13: 1303–1311
Liu G, Franssen E, Fitch MI and Warner E (1997) Patient preference for oral versus intravenous palliative chemotherapy. J Clin Oncol 15: 110–115
Malik ST, Talbot D, Clarke PI, Osborne R, Reznek R, Wrigley PF and Slevin ML (1990) Phase II trial of UFT in advanced colorectal and gastric cancer. Br J Cancer 62: 1023–1025
Pazdur R, Lassere Y, Rhodes V, Ajani JA, Sugarman SM, Patt YZ, Jones DV Jr, Markowitz AB, Abbruzzese JL, Bready B and Levin B (1994) Phase II trial of uracil and tegafur plus oral leucovorin: An effective oral regimen in the treatment of metastatic colorectal carcinoma. J Clin Oncol 12: 2296–2300
Petrelli N, Douglass HO Jr, Herrera L, Russell D, Stablein DM, Bruckner HW, Mayer RJ, Schinella R, Green MD, Muggia FM, Megibow A, Greenwald ES, Bukowski RM, Harris J, Levin B, Gaynor E, Loutfi A, Kalser MH, Barkin JS, Benedetto P, Woolley PV, Nauta R, Weaver DW and Leichman LP for the gastrointestinal tumor study group (1989) The modulation of fuorouracil with leucovorin in metastatic colorectal carcinoma: A prospective randomized phase III trial. J Clin Oncol 7: 1419–1426
Poon MA, O’Connel MJ, Moertel CG, Wieand HS, Cullina SA, Everson LK, Krook JE, Mailliard JA, Laurie JA, Tschetter LK and Wiesenfeld M (1989) Biochemical modulation of fluorouracil: Evidence of significant improvement of survival and quality of life in patients with advanced colorectal carcinoma. J Clin Oncol 7: 1407–1418
Sakata Y, Ohtsu A, Horikoshi N, Sugimachi K, Mitachi Y and Taguchi T (1998) Late phase II study of novel oral fluoropyrimidine anticancer drugs S-1 (1M tegafur–0.4M gimestat–1M otastat potassium) in advanced gastric cancer patients. Eur J Cancer 34: 1715–1720
Schilsky R, Bukowski R, Burris H, Crawford J, Hochster H, O’Rourke M, Steinfeldt H, Doucette M, Levin J and Hohneker J (1997) A phase II study of a five day regimen of 5-fluorouracil (5-FU) plus GW776 (776C85) with or without leucovorin in patients with metastatic colorectal cancer. Proc Am Soc Clin Oncl 16: 271a (abst 961)
Shirasaka T, Nakano K, Takechi T, Satake H, Uchida J, Fujioka A, Saito H, Okabe H Oyama K, Takeda S, Unemi N and Fukushima M (1996) Antitumor activity of 1M tegafur-0.4M 5-chloro-2,4-dihydroxypyridine-1M potassium oxonate (S-1) against human colon carcinoma orthotopically implanted into nude rats. Cancer Res 56: 2602–2606
Shirasaka T, Shimamoto Y and Fukushima M (1993) Inhibition by oxonic acid of gastrointestinal toxicity of 5-fluorouracil without loss of its antitumor activity in rats. Cancer Res 53: 4004–4009
Sugimachi K, Maehara Y, Horikoshi N, Shimada Y, Sakata Y, Mitachi Y and Taguchi T The S-1 Gastrointestinal Cancer Study Group (2000) An early phase II study of oral S-1, a newly developed 5-fluorouracil derivative for advanced and recurrent gastrointestinal cancers. Oncology 57: 202–210
Takiuchi H and Ajani JA (1998) Uracil-tegafur in gastric carcinoma: a comprehensive review. J Clin Oncol 16: 2877–2885
Tatsumi K, Fukushima M, Shirasaka T and Fujii S (1987) Inhibitory effects of pyrimidine, barbituric acid and pyridine derivatives on 5-fluorouracil degradation in rat liver extracts. Jpn J Cancer Res 78: 748–755
Takechi T, Nakano K, Uchida J, Mita A, Toko K, Takeda S, Unemi N and Shirasaka T (1997) Antitumor activity and low intestinal toxicity of S-1, a new formulation of oral tegafur, in experimental tumor models in rats. Cancer Chemother Pharmacol 39: 205–211
Taguchi T, Inuyama Y, Kanamaru R, Hasegawa K, Akazawa S, Niitani H, Furue H, Kurihara M, Ota K, Suga S, Ariyoshi Y, Takai S, Shimoyama T, Toge T, Takashima S, Sugimachi K, Hara Y, Fujita H, Kimura K, Saito T, Tsukagoshi S and Nakao I (1997) Phase I study of S-1. Jpn J Cancer Chemother 24(15): 2253–2264
Tuchman M, Stoeckeler JS, Kiang DT, O’Dea RF, Ramnaraine ML and Mirkin BL (1985) Familial pyrimidinemia and pyrimidinuria associated with severe fluorouracil toxicity. N Engl J Med 313: 245–249
Author information
Authors and Affiliations
Consortia
Rights and permissions
From twelve months after its original publication, this work is licensed under the Creative Commons Attribution-NonCommercial-Share Alike 3.0 Unported License. To view a copy of this license, visit http://creativecommons.org/licenses/by-nc-sa/3.0/
About this article
Cite this article
Ohtsu, A., Baba, H., Sakata, Y. et al. Phase II study of S-1, a novel oral fluoropyrimidine derivative, in patients with metastatic colorectal carcinoma. Br J Cancer 83, 141–145 (2000). https://doi.org/10.1054/bjoc.2000.1236
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1054/bjoc.2000.1236
Keywords
This article is cited by
-
A case of primary ovarian signet-ring cell carcinoma treated with S-1/CDDP therapy
Journal of Ovarian Research (2020)
-
Phase I study of DFP-11207, a novel oral fluoropyrimidine with reasonable AUC and low Cmax and improved tolerability, in patients with solid tumors
Investigational New Drugs (2020)
-
Phase II study of S-1 on alternate days plus bevacizumab in patients aged ≥ 75 years with metastatic colorectal cancer (J-SAVER)
International Journal of Clinical Oncology (2019)
-
Determination of 5-fluorouracil and tegafur in tear fluid of patients treated with oral fluoropyrimidine anticancer agent, S-1
Japanese Journal of Ophthalmology (2018)
-
Phase I/II study of bi-weekly XELIRI plus bevacizumab treatment in patients with metastatic colorectal cancer resistant to oxaliplatin-based first-line chemotherapy
Cancer Chemotherapy and Pharmacology (2017)