Abstract
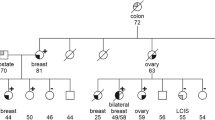
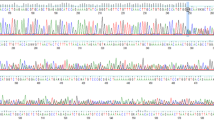
BRCA1 mutations have been identified in breast and ovarian cancer families from diverse ethnic backgrounds. We studied 17 different families with the BRCA1 2800delAA mutation; seven were ascertained in Scotland (Dundee, Edinburgh, Glasgow, St Andrews), five in Canada (Toronto, Victoria) and five in the United States (Chicago, Philadelphia, Seattle). Overall there was a clear preponderance of Scottish ancestry. Genotype analysis performed on key members from 17 families was consistent with a common haplotype, strongly suggesting a single ancestral origin. A possible link was established between two families by tracing their genealogies through the records of the Registrar General for Scotland. This is the first example of a BRCA1 mutation likely to be derived from a common founder in Scotland. Further studies will be necessary to estimate more accurately the population frequency of the BRCA1 2800delAA mutation among unselected cases of breast and ovarian cancer in Scotland and the UK. © 2000 Cancer Research Campaign
Similar content being viewed by others
Article PDF
Change history
16 November 2011
This paper was modified 12 months after initial publication to switch to Creative Commons licence terms, as noted at publication
References
BIC (1998). Breast Cancer Information Core Database: http://www.nhgri.nih.gov/Intramural_research/Lab_transfer/Bic/
Bodmer JG, Kennedy LJ, Lindsay J and Wasik AM (1987) Applications of serology and the ethnic distribution of three locus HLA haplotypes. Br Med Bull 43: 94–121
Boyd M, Harris F, McFarlane R, Davidson HR and Black DM (1995) A human BRCA1 gene knockout. Nature 375: 541–542
Claus EB, Schildkraut JM, Thompson WD and Risch NJ (1996) The genetic attributable risk of breast and ovarian cancer. Cancer 77: 2318–2324
Collyer S and De Mey R (1987) Public records and recognition of genetic disease in Scotland. Clin Genet 31: 125–131
Easton DF, Ford D and Peto J (1993) Inherited susceptibility to breast cancer. Cancer Surv 18: 1–17
Easton DF, Ford D and Bishop DT (1995) Breast and ovarian cancer incidence in BRCA1 -mutation carriers. Breast Cancer Linkage Consortium. Am J Hum Genet 56: 265–271
Ford D, Easton DF, Bishop DT, Narod SA and Goldgar DE (1994) Risks of cancer in BRCA1 mutation carriers. Lancet 343: 692–695
Ford D, Easton DF and Peto J (1995) Estimates of the gene frequency of BRCA1 and its contribution to breast and ovarian cancer incidence. Am J Hum Genet 57: 1457–1462
Friedman LS, Ostermeyer EA, Szabo CI, Dowd P, Lynch ED, Rowell SE and King M-C (1994) Confirmation of BRCA1 by analysis of germline mutations linked to breast and ovarian cancer in ten families. Nat Genet 8: 399–404
Friedman LS, Szabo CI, Ostermeyer EA, Dowd P, Butler L, Park T, Lee MK, Goode EL, Rowell SE and King M-C (1995) Novel inherited mutations and variable expressivity of BRCA1 alleles, including the founder mutation 185delAG in Ashkenazi Jewish families. Am J Hum Genet 57: 1284–1297
Gayther SA, Harrington P, Russell P, Kharkevich G, Garkavtseva RF and Ponder BAJ (1997) Frequently occurring germ-line mutations of the BRCA1 gene in ovarian cancer families from Russia. Am J Hum Genet 60: 1239–1242
IARC (1997). Cancer Incidence in Five Continents Vol. VII. International Agency for Research on Cancer: Lyon
Johannesdottir G, Gudmundsson J, Bergthorsson JT, Arason A, Agnarsson BA, Eiriksdottir G, Johannsson OT, Borg A, Ingvarsson S, Easton DF, Egilsson V and Barkardottir RB (1996) High prevalence of the 999del5 mutation in Icelandic breast and ovarian cancer patients. Cancer Res 56: 3663–3665
Lancaster JM, Carney ME, Gray J, Myring J, Gumbs C, Sampson J, Wheeler D, France E, Wiseman R, Harper P and Futreal PA (1998) BRCA1 and BRCA2 in breast cancer families from Wales: moderate mutation frequency and two recurrent mutations in BRCA1. Br J Cancer 78: 1417–1420
Liede A, Rehal P, Vesprini D, Jack E, Abrahamson J and Narod SA (1998) A breast cancer patient of Scottish descent with germ-line mutations in BRCA1 and BRCA2. Am J Hum Genet 60: 1543–1544
Liu CY, Flesken-Nikitin A, Li S, Zeng Y and Lee WH (1996) Inactivation of the mouse BRCA1 gene leads to failure in the morphogenesis of the egg cylinder in early postimplantation development. Genes Dev 10: 1835–1843
Lyon RLL (1962) Huntington's chorea in the Moray Firth area. Br Med J i: 1301–1306
Mullen P, Miller WR, Mackay J, Fitzpatrick DR, Langdon SP and Warner JP (1997) BRCA1 5382insC in sporadic and familial breast and ovarian carcinoma in Scotland. Br J Cancer 75: 1377–1380
Neuhausen SL, Mazoyer S, Friedman L, Stratton M, Offit K, Caligo A, Tomlinson G, Cannon-Albright L, Bishop T, Kelsell D, Solomon E, Weber B, Couch F, Struewing J, Tonin P, Durocher F, Narod S, Skolnick MH, Lenoir G, Serova O, Ponder B, Stoppa-Lyonnet D, Easton D, King M-C and Goldgar DE (1996) Haplotype and phenotype analysis of six recurrent BRCA1 mutations in 61 families: results of an international study. Am J Hum Genet 58: 271–280
Newman B, Mu H, Butler LM, Millikan RC, Moorman PG and King M-C (1998) Frequency of breast cancer attributable to BRCA1 in a population-based series of American women. J Am Med Assoc 279: 915–921
Peto J, Collins N, Barfoot R, Seal S, Warren W, Rahman N, Easton DF, Evans C, Deacon J and Stratton MR (1999) Prevalence of BRCA1 and BRCA2 gene mutations in patients with early-onset breast cancer. Journal of the National Cancer Institute 91: 943–949
Peelen T, van Vliet M, Petruj-Bosch A, Mieremet C, van den Ouweland AMW, Hoervorst F, Brohet R, Ligtenberg MJL, Teugels E, van der Luijt R, van der Hout AH, Gille JJP, Pals G, Jedema I, Olmer R, van Leeuwen I, Newman B, Plandsoen M, van der Est M, Brink G, Hageman S, Arts PJW, Bakker MM, Willems HW, van der Looij E, Neyns B, Bonduelle M, Jansen R, Oosterwijk JC, Sijmons R, Smeets HJM, van Asperen CJ, Meijers-Heijboer H, Klijn JGM, de Greve J, King M-C, Menko FH, Brunner HG, Halley D, van Ommen G-JB, Vasen HFA, Cornelisse CJ, van't Veer LJ, de Knijff P, Bakker E and Devilee P (1997) A high proportion of novel mutations in BRCA1 with strong founder effects among Dutch and Belgian hereditary breast and ovarian cancer families. Am J Hum Genet 60: 1041–1049
Petrij-Bosch A, Peelen T, van Vliet M, van Eijk R, Olmer R, Drusedau M, Hogervorst FBL, Hageman S, Arts PJW, Ligtenberg MJL, Meijers-Heijboer H, Klijn JGM, HFA V, Cornelisse CJ, van't Veer LJ, Bakker E, van Ommen G-JB and Devilee P (1997) BRCA1 genomic deletions are major founder mutations in Dutch breast cancer patients. Nat Genet 17: 341–345
Ramus SJ, Kote-Jarai Z, Friedman LS, van der Looij M, Gayther SA, Csokay B, Ponder BAJ and Olah E (1997) Analysis of BRCA1 and BRCA2 mutations in Hungarian families with breast or breast-ovarian cancer. Am J Hum Genet 60: 1242–1246
Roa BB, Boyd AA, Vocik K and Richards CS (1996) Ashkenazi Jewish population frequencies for common mutations in BRCA1 and BRCA2. Nat Genet 14: 185–187
Sinclair S (1990). Tracing your Scottish Ancestors: a Guide to Ancestry Research in the Scottish Record Office. HMSO: Edinburgh
Struewing JP, Abeliovich D, Peretz T, Avishai N, Kaback MM, Collins FS and Brody LC (1995) The carrier frequency of the BRCA1 185delAG mutation is approximately 1 percent in Ashkenazi Jewish individuals. Nat Genet 11: 198–200
Struewing JP, Hartge P, Wacholder S, Baker SM, Berlin M, McAdams M, Timmerman MM, Brody LC and Tucker MA (1997) The risk of cancer associated with specific mutations of BRCA1 and BRCA2 among Ashkenazi Jews. New Engl J Med 336: 1401–1408
Szabo CI and King M-C (1997) Population genetics of BRCA1 and BRCA2. Am J Hum Genet 60: 1013–1020
Thorlacius S, Sigurdsson S, Bjarnadottir H, Olafsdottir G, Jonasson JG, Tryggvadottir L, Tulinius H and Eyfjord JE (1997) Study of a single BRCA2 mutation with high carrier frequency in a small population. Am J Hum Genet 60: 1079–1084
Tonin P, Weber B, Offit K, Couch F, Rebbeck TR, Neuhausen S, Godwin AK, Daly M, Wagner-Costalos J, Berman D, Grana G, Fox E, Kane MF, Kolodner RD, Krainer M, Haber DA, Struewing JP, Warner E, Rosen B, Lerman C, Peshkin B, Norton L, Serova O, Foulkes WD, Lynch HT, Lenoir GM, Narod SA and Garber JE (1996) Frequency of recurrent BRCA1 and BRCA2 mutations in Ashkenazi Jewish breast cancer families. Nat Med 2: 1179–1183
Tonin PN, Mes-Masson A-M, Futreal A, Morgan K, Mahon M, Foulkes WD, Cole DEC, Provencher D, Ghadirian P and Narod SA (1998) Founder BRCA1 and BRCA2 mutations in French Canadian breast and ovarian cancer families. Am J Hum Genet 63: 1341–1351
Xu CF, Chambers JA, Nicolai H, Brown MA, Hujeirat Y, Mohammed S, Hodgson S, Kelsell DP, Spurr NK, Bishop DT and Solomon E (1997) Mutations and alternative splicing of the BRCA1 gene in UK breast/ovarian cancer families. Genes Chromosomes Cancer 18: 102–110
Author information
Authors and Affiliations
Rights and permissions
From twelve months after its original publication, this work is licensed under the Creative Commons Attribution-NonCommercial-Share Alike 3.0 Unported License. To view a copy of this license, visit http://creativecommons.org/licenses/by-nc-sa/3.0/
About this article
Cite this article
Liede, A., Cohen, B., Black, D. et al. Evidence of a founder BRCA1 mutation in Scotland. Br J Cancer 82, 705–711 (2000). https://doi.org/10.1054/bjoc.1999.0984
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1054/bjoc.1999.0984
Keywords
This article is cited by
-
Founder BRCA1/2 mutations in the Europe: implications for hereditary breast-ovarian cancer prevention and control
EPMA Journal (2010)
-
A new germline mutation in BRCA1 gene in a sicilian family with ovarian cancer
Breast Cancer Research and Treatment (2006)
-
Molecular and genealogical characterization of the R1443X BRCA1 mutation in high-risk French-Canadian breast/ovarian cancer families
Human Genetics (2005)
-
Highly penetrant hereditary cancer syndromes
Oncogene (2004)
-
BRCA1 and BRCA2 mutations in Scotland and Northern Ireland
British Journal of Cancer (2003)