Abstract
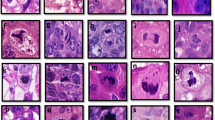
Apoptosis plays an important role in tumorigenesis. Tumour growth is determined by the rate of cell proliferation and cell death. We counted the number of apoptotic cells in haematoxylin and eosin (H&E)-stained tumour sections in series of 172 grade I and II invasive breast cancers with long-term follow-up. The number of apoptotic cells in ten high-power fields were converted to the number of apoptotic cells per mm2 to obtain the apoptotic index (AI). The AI showed a positive correlation to the mitotic activity index (MAI) (P = 0.0001), histological grade (P< 0.0001) and worse tumour differentiation. Patients with high AI showed shorter overall survival than patients with low AI in the total group as well as in the lymph node-positive group. Tumour size, MAI, lymph node status and AI were independent prognostic indicators in multivariate analysis. The AI was shown to be of additional prognostic value to the MAI in the total patients group as well as in the lymph node-positive group. The correlation between the AI and the MAI points to linked mechanisms of apoptosis and proliferation. Since apoptotic cells can be counted with good reproducibility in H&E-stained tumour sections, the AI may be used as an additional prognostic indicator in invasive breast cancer. © 2000 Cancer Research Campaign
Similar content being viewed by others
Article PDF
Change history
16 November 2011
This paper was modified 12 months after initial publication to switch to Creative Commons licence terms, as noted at publication
References
Allan DJ, Howell A, Roberts SA, Williams GT, Watson RJ, Coyde JD, Clarke RB, Laidlaw IJ and Potten CS (1991) Reduction in apoptosis relative to mitosis in histologically normal epithelium accompanies fibrocyctic change and carcinoma of the premenopausal human breast. J Pathol 163: 337–342
Baak JPA, Dop van H, Kurver PHJ and Hermans J (1985) The value of morphometry to classic prognosticators in breast cancer. Cancer 56: 374–382
Barbareschi M, Caffo O, Veronese S, Leek RD, Fina P, Fox S, Bonzanini M, Girlando S, Morelli L, Eccher C, Pezzella F, Doglioni C, Dalla Palma P and Harris A (1996) Bcl-2 and p53 expression in node-negative breast carcinoma: a study with long-term follow-up. Hum Pathol 27: 1149–1155
Bargou RC, Dainel PT, Mapara MY, Bommert K, Wagener C, Killinich B, Royer HD and Dörken B (1995) Expression of the bcl-2 gene family in normal and malignant breast tissue: low bax-alpha expression in tumor cells correlates with resistance towards apoptosis. Int J Cancer 60: 854–859
Berardo MD, Elledge RM, de Moor C, Clark GM, Osborne CK and Allred DC (1998) Bcl-2 and apoptosis in lymph node positive carcinoma. Cancer 82: 1296–1302
Berns EMJJ, Klijn JGM, Putten WLJvan, Staveren ILvan, Portengen H and Foekens JA (1992) c-myc amplification is a better prognostic factor than HER2/neu amplification in primary breast cancer. Cancer Res 52: 1107–1113
Borg Å, Baldetorp B, Fernö M, Olsson H and Sigurdsson H (1992) c-myc amplification is an independent prognostic factor in postmenopausal breast cancer. Int J Cancer 51: 687–691
Clayton F (1991) Pathologic correlates of survival in 378 lymph node-negative infiltrating ductal breast carcinomas. Mitotic count is the best single predictor. Cancer 68: 1309–1317
Davis PL, Shaiu WL, Scott GL, Iglehart JD, Hsieh TS and Marks JR (1998) Complex response of breast epithelial cell lines to topoisomerase inhibitors. Anticancer Res 18: 2918–2932
Diest PJ van and Baak JPA (1992) The morphometric multivariate prognostic index (MPI) is the strongest prognosticator in premenopausal lymph node negative and lymph node positive breast cancer patients. Hum Pathol 22: 479–489
Diest PJ van, Michalides RJAM, Jannink I, Valk P van der, Peterse HL, Jong JS de, Meijer CJLM and Baak JPA (1997) Cyclin D1 overexpression in breast cancer. Correlations and prognostic value. Am J Pathol 150: 705–711
Doglioni C, Dei Tos AP, Laurino L, Chiarelli C, Barbareschi M and Viale G (1994) The prevalence of Bcl-2 immunoreactivity in breast carcinomas and its clinicopathological correlations, with particular reference to estrogen receptor status. Virchows Arch 424: 47–51
Elston CW (1987) Grading of invasive carcinoma of the breast. Page DL, Anderson TJ, eds. Diagnostic histopathology of the breast. Edinburgh, Churchill Livingstone.300–311
Evan GI, Wyllie A and Gilbert C (1992) Induction of apoptosis in fibroblasts by c-myc protein. Cell 69: 119–128
Evan GI, Harrington E, McCarthy N, Gilbert C, Benedict MA and Nunez G (1996) Integrated control of cell proliferation and apoptosis by oncogenes. In: Apoptosis and Cell Cycle Control in Cancer, Thomas NSB (ed), pp. 109–129. BIOS Scientific: Oxford
Farrow SN and Brown R (1996) New members of the Bcl-2 family and their protein partners. Curr Opinion Genet Develop 6: 45–49
Gulli LF, Palmer KC, Chen YQ and Reddy KB (1996) Epidermal growth factor-induced apoptosis in A431 cells can be reversed by reducing the tyrosine kinase activity. Cell Grow Diff 7: 173–178
Heatley MK (1995) Association between the apoptotic index and established prognostic parameters in endometrial adenocarcinoma. Histopathology 27: 469–472
Jannink I, Diest PJ van and Baak JPA (1995) Comparison of the prognostic value of four methods to assess mitotic activity in 186 invasive breast cancer patients: classical and random mitotic activity assessments with correction for volume percentage of epithelium. Hum Pathol 26: 1086–1092
Jong JS de, Diest PJ van, Michalides RJAM, Valk P van der, Meijer CJLM and Baak JPA (1998) Correlation of cyclin D1 and Rb gene expression with apoptosis in invasive breast cancer. Mol Pathol 51: 30–34
Kapucuoglu N, Losi L and Eusebi V (1997) Immunohistochemical localization of Bcl-2 and Bax proteins in in situ and invasive breast carcinomas. Virchows Arch 430: 17–22
Krajewski S, Krajewska M, Shabaik A, Miyashita T, Wang HG and Reed JC (1994) Immunohistochemical determination of in vivo distribution of Bax, a dominant inhibitor of Bcl-2. Am J Pathol 145: 1323–1336
Krajewski S, Blomqvist C, Franssila K, Krajewska M, Wasenius VM, Niskanen E and Reed JC (1995) Reduced expression of the proapoptotic gene BAX is associated with poor response rates to combined chemotherapy and shortened survival in women with metastatic breast adenocarcinoma. Cancer Res 55: 4471–4478
Kranenburg O, Eb AJ van der and Zantema A (1996) Cyclin D1 is an essential mediator of apoptotic neuronal cell death. EMBO J 15: 46–54
Leoncini L, Del Vecchio MT and Magha T (1993) Correlations between apoptotic and proliferation indices in malignant non-Hodgkin’s lymphoma. Am J Pathol 142: 755–763
Linden JC van der, Baak JPA, Lindeman J, Hermans J and Meijer CJLM (1987) Prospective evaluation of prognostic value of morphometry in patients with primary breast cancer. J Clin Pathol 40: 302–306
Lipponen P (1999) Apoptosis in breast cancer: relationship with other pathological parameters. Endocrine-Rel Cancer 6: 13–16
Lipponen P and Aaltomaa S (1994) Apoptosis in bladder cancer as related to standard prognostic factors and prognosis. J Pathol 173: 333–339
Lipponen P, Collan Y and Eskelinen MJ (1991) Volume corrected mitotic index (M/V index), mitotic activity index (MAI), and histological grading in breast cancer. Int Surg 76: 245–249
Lipponen P, Aaltomaa S, Kosma VM and Syrjanen K (1994) Apoptosis in breast cancer as related to histopathological characteristics and prognosis. Eur J Cancer 30A: 2068–2073
Minn AJ, Boise LH and Thompson CB (1996) Bcl-xSantagonizes the protective effects of Bcl-xL . J Biol Chem 271: 6306–6312
Miyashita T, Kitada S, Krajewski S, Horne WA, Delia D and Reed JC (1995) Overexpression of Bcl-2 protein increases the half-life of p21 (Bax). J Biol Chem 270: 26049–26052
Mustonene M, Taunio H, Pääkkö P and Soini Y (1997) The extent of apoptosis is inversely associated with bcl-2 expression in premalignant and malignant breast lesions. Histopathology 31: 347–353
Perego P, Giarola M, Righetti SC, Supino R, Caserini C, Delia D, Pierotti MA, Miyashita T, Reed JC and Zunino F (1996) Association between cisplatin resistance and mutation ofp53 gene and reduced bax expression in ovarian carcinoma cell systems. Cancer Res 56: 556–562
Schepop HAM, Jong JS de, Diest PJ van and Baak JPA (1996) Counting of apoptotic cells: a methodological study in invasive breast cancer. Mol Pathol 49: M214–M217
Sierra A, Castellsagué X, Coll T, Mañas S, Escobedo A, Moreno A and Fabra A (1998) Expression of death-related genes and their relationship to loss of apoptosis in T1ductal breast carcinomas. Int J Cancer 79: 103–110
Sjöström J, Krajewski S, Franssila K, Niskanen E, Wasenius VM, Nordling S, Reed JC and Blomqvist C (1998) A multivariate analysis of tumour biological factors predicting response to cytotoxic treatment in advanced breast cancer. Br J Cancer 78: 812–815
Slooten HJ van, Vijver MJ van de, Velde CJ van de and Dierendonck JH van (1998) Loss of Bcl-2 in invasive breast cancer is associated with high rates of cell death, but also with increased proliferation activity. Br J Cancer 77: 789–796
Staunton MJ and Gaffney EF (1998) Apoptosis: basic concepts and potential significance in human cancer. Arch Pathol Lab Med 122: 310–319
Uyterlinde AM, Schipper NW, Baak JPA, Peterse H and Matze P (1988) Limited prognostic value of cellular DNA content to classical and morphological parameters in invasive ductal breast cancer. Am J Clin Pathol 89: 301–307
Veronese S, Mauri FA, Caffo O, Scaioli M, Aldovini D, Perrone G, Galligioni E, Doglioni C, Dalla Palma P and Barbareschi M (1998) Bax immunohistochemical expression in breast carcinoma: a study with long term follow-up. Int J Cancer 79: 13–18
Weisman JH, Jonker RR, Keijzer R, Velde CJH van de, Cornelisse CJ and Dierendonck JH van (1993) A new method to detect apoptosis in paraffin sections: in situ end-labelling of fragmented DNA. J Histochem Cytochem 41: 7–12
Yang E and Korsmeyer SJ (1996) Molecular thanatopsis: a discourse on the Bcl-2 family and cell death. Blood 88: 386–401
Zhang GJ, Kimijima I, Abe R, Watanabe T, Kanno W, Kiyoshi H and Tsuchiya A (1998) Apoptotic index correlates to bcl-2 and p53 protein expression, histological grade and prognosis in invasive breast cancers. Anticancer Res 18: 1989–1998
Author information
Authors and Affiliations
Rights and permissions
From twelve months after its original publication, this work is licensed under the Creative Commons Attribution-NonCommercial-Share Alike 3.0 Unported License. To view a copy of this license, visit http://creativecommons.org/licenses/by-nc-sa/3.0/
About this article
Cite this article
de Jong, J., Diest, P. & Baak, J. Number of apoptotic cells as a prognostic marker in invasive breast cancer. Br J Cancer 82, 368–373 (2000). https://doi.org/10.1054/bjoc.1999.0928
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1054/bjoc.1999.0928
Keywords
This article is cited by
-
COVID-19 and cancer: start the resolution!
Cancer and Metastasis Reviews (2022)
-
Debris-stimulated tumor growth: a Pandora’s box?
Cancer and Metastasis Reviews (2021)
-
A fate worse than death: apoptosis as an oncogenic process
Nature Reviews Cancer (2016)
-
The search for optimal cutoff points for apoptosis and proliferation rate in prognostification of early stage breast cancer patients treated with anthracyclines in adjuvant settings
Tumor Biology (2016)
-
The role and prognostic value of apoptosis in colorectal carcinoma
BMC Clinical Pathology (2013)