Summary
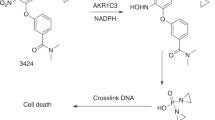
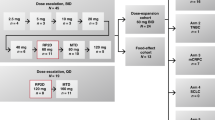
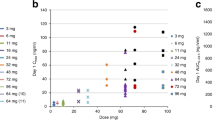
Temozolomide, an oral cytotoxic agent with approximately 100% bioavailability after one administration, has demonstrated schedule-dependent clinical activity against highly resistant cancers. Thirty patients with minimal prior chemotherapy were enrolled in this phase I trial to characterize the drug’s safety, pharmacokinetics and anti-tumour activity, as well as to assess how food affects oral bioavailability. To determine dose-limiting toxicities (DLT) and the maximum tolerated dose (MTD), temozolomide 100–250 mg m–2 was administered once daily for 5 days every 28 days. The DLT was thrombocytopenia, and the MTD was 200 mg m–2 day–1. Subsequently, patients received the MTD to study how food affects the oral bioavailability of temozolomide. When given orally once daily for 5 days, temozolomide was well tolerated and produced a non-cumulative, transient myelosuppression. The most common non-haematological toxicities were mild to moderate nausea and vomiting. Clinical activity was observed against several advanced cancers, including malignant glioma and metastatic melanoma. Temozolomide demonstrated linear and reproducible pharmacokinetics and was rapidly absorbed (mean Tmax ~1 h) and eliminated (mean t1/2 = 1.8 h). Food produced a slight reduction (9%) in absorption of temozolomide. Temozolomide 200 mg m–2 day–1 for 5 days, every 28 days, is recommended for phase II studies.
Similar content being viewed by others
Article PDF
Change history
16 November 2011
This paper was modified 12 months after initial publication to switch to Creative Commons licence terms, as noted at publication
References
Bleehen, NM, Newlands, ES, Lee, SM, Thatcher, LN, Selby, P, Calvert, AH, Rustin, GJS, Brampton, M & Stevens, MFG (1995). Cancer Research Campaign phase II trial of temozolomide in metastatic melanoma. J Clin Oncol 13: 910–913.
Bower, M, Newlands, ES, Bleehen, NM, Brada, M, Begent, RJH, Calvert, H, Colquhoun, I, Lewis, P & Brampton, MH (1997). Multicentre CRC phase II trial of temozolomide in recurrent or progressive high-grade glioma. Cancer Chemother Pharmacol 40: 484–488.
Brock, CS, Matthews, JC, Brown, G, Newlands, ES & Price, P (1997). In vivo demonstration of 11C-temozolomide uptake by human recurrent high-grade astrocytomas [abstract]. Br J Cancer 75: 1241.
Catapano, CV, Broggini, M, Erba, E, Ponti, M, Marianti, L, Citti, L & D’Incalci, M (1987). In vitro and in vivo methazolastone-induced DNA damage and repair in L1210 leukemia sensitive and resistant to chloroethylnitrosoureas. Cancer Res 47: 4884.
Carter, CA, Waud, WR & Plowman, J (1994). Responses of human melanoma, ovarian, and colon tumor xenografts in nude mice to oral temozolomide. Proc Am Assoc Cancer Res 35: 297 (abstract 1769).
D’Atri, S, Piccioni, D, Castellano, A, Tuorto, V, Franchi, A, Lu, K, Christiansen, N, Frankel, S, Rustum, YM, Papa, G, Mandelli, F & Bonmassar, E (1995). Chemosensitivity to triazene compounds and O6-alkylguanine-DNA alkyltransferase levels: studies with blasts of leukaemic patients. Ann Oncol 6: 389–393.
Denny, BJ, Wheelhouse, RT, Stevens, MFG, Tsang, LLH & Slack, JA (1994). NMR and molecular modeling investigation of the mechanism of activation of the antitumor drug temozolomide and its interaction with DNA. Biochemistry 33: 9045–9051.
Dhodapkar, M, Rubin, J, Reid, JM, Burch, PA, Pitot, HC, Buckner, JC, Ames, MM & Suman, VJ (1997). Phase I clinical trial of temozolomide (NSC 362856) in patients with advanced cancer. Clin Cancer Res 3: 1093–1100.
Friedman, HS, Dolan, ME, Pegg, AE, Marcelli, S, Keir, S, Catino, JJ, Bigner, DD & Schold, SC Jr (1995). Activity of temozolomide, the treatment of central nervous system tumor xenografts. Cancer Res 55: 2853–2857.
Kim, KH, Lin, CC, Parker, D, Veals, J, Lim, J, Likhari, P, Statkevich, P, Marco, A & Nomeir, AA (1997). High-performance liquid chromatographic determination and stability of 5-(3-methyltriazen-1-yl)-imidazo-4-carboximide, the biologically active product of the antitumor agent temozolomide, in human plasma. J Chromatogr B 703: 225–233.
Levin, V, Yung, A, Prados, M, Poisson, M, Rosenfeld, S, Brada, M, Friedman, H, Albright, R, Olson, J, Bruner, J, Yue, N & Dugan, M, Temodal Brain Group (1997). Phase II study of Temodal® (temozolomide) at first relapse in anaplastic astrocytoma (AA) patients [abstract 1370]. Proc Am Soc Clin Oncol 16: 384a.
Liu, L, Chatterjee, S & Gerson, SL (1997). Blockade of base excision repair appears to mediate cytotoxicity to temozolomide in mismatch repair deficient tumor cells [abstract 1536]. Proc Am Assoc Cancer Res 38: 288.
Newlands, ES, Blackledge, GRP, Slack, JA, Rustin, GJS, Smith, DB, Stuart, NSA, Quarterman, CP, Hoffman, R, Stevens, MFG, Brampton, MH & Gibson, AC (1992). Phase I trial of temozolomide (CCRG 81045: M&B 39831: NSC 362856). Br J Cancer 65: 287–291.
Newlands, ES, O’Reilly, SM, Glaser, MG, Bower, M, Evans, H, Brock, C, Brampton, MH, Colquhoun, I, Lewis, P, Rice-Edwards, JM, Illingworth, RD & Richards, PG (1996). The Charing Cross Hospital experience with temozolomide in patients with gliomas. Eur J Cancer 32A: 2236–2241.
O’Reilly, SM, Newlands, ES, Glaser, MG, Brampton, M, Rice-Edwards, JM, Illingworth, RD, Richards, PG, Kennard, C, Colquohoun, IR, Lewis, P & Stevens, MFG (1993). Temozolomide: a new oral cytotoxic chemotherapeutic agent with promising activity against primary brain tumours. Eur J Cancer 29A: 940–942.
Patel, M, McCully, C, Godwin, K & Balis, F (1995). Plasma and cerebrospinal fluid pharmacokinetics of temozolomide [abstract 1485]. Proc Am Soc Clin Oncol 14: 461.
Plowman, J, Waud, WR, Koutsoukos, AD, Rubinstein, LV, Moore, TD & Grever, MR (1994). Preclinical antitumor activity of temozolomide in mice: efficacy against human brain tumor xenografts and synergism with 1,3-bis(2-chloroethyl)-1 nitrosourea. Cancer Res 54: 3793–3799.
Reidenberg, P & Villalona, M (1996). Phase I clinical and pharmacokinetic study of temozolomide in advanced cancer patients stratified by extent of prior therapy [abstract 344]. Ann Oncol 7: 99.
Shen, F, Decosterd, LA, Gander, M, Leyvraz, S, Biollaz, J & Lejeune, FJ (1995). Determination of temozolmide in human plasma and urine by high-performance liquid chromatography after solid-phase extraction. Chromatogr B 667: 291–300.
Stevens, MFG, Hickman, JA, Langdon, SP, Chubb, D, Vickers, L, Stone, R, Baig, G, Goddard, C, Bigson, NW, Slack, JA, Newton, C, Lunt, E, Fizames, C & Lavelle, F (1987). Anti-tumor activity and pharmacokinetics in mice of 8-carbamoyl-3-methylimidazo (5,1-d)-1,2,3,5-tetrazin-4(3H)-one; a novel drug with potential as an alternative to dacarbazine. Cancer Res 47: 5846–5852.
Tsang, LLH, Farmer, PB, Gescher, A & Slack, JA (1990). Characterization of urinary metabolites of temozolomide in humans and mice and evaluation of their cytotoxicity. Cancer Chemother Pharmacol 26: 429–436.
Waud, WR, Rubinstein, LV, Kaldandrug, S, Plowman, J & Alley, MC (1996). In vivo combination chemotherapy evaluations of topotecan with cisplatin and temozolomide [abstract 1988]. Proc Am Assoc Cancer Res 37: 292.
Wedge, SR, Porteous, JK & Newlands, ES (1996). 3-Aminobenzamide and/or O6 benzylguanine evaluated as an adjunct to temozolomide or BCNU treatment in cell lines of variable mismatch repair status and O6-alkylguanine–DNA alkyltransferase activity. Br J Cancer 74: 1030–1036.
Wedge, SR, Porteous, JK & Newlands, ES (1997a). Effect of single and multiple administration of an O6-benzylguanine/temozolomide combination: an evaluation in a human melanoma xenograft model. Cancer Chemother Pharmacol 40b: 266–272.
Wedge, SR, Porteous, JK, Glaser, MG, Marcus, K & Newlands, ES (1997b). In vitro evaluation of temozolomide combined with X-irradiation. Anticancer Drugs 8a: 92–97.
Author information
Authors and Affiliations
Rights and permissions
From twelve months after its original publication, this work is licensed under the Creative Commons Attribution-NonCommercial-Share Alike 3.0 Unported License. To view a copy of this license, visit http://creativecommons.org/licenses/by-nc-sa/3.0/
About this article
Cite this article
Brada, M., Judson, I., Beale, P. et al. Phase I dose-escalation and pharmacokinetic study of temozolomide (SCH 52365) for refractory or relapsing malignancies. Br J Cancer 81, 1022–1030 (1999). https://doi.org/10.1038/sj.bjc.6690802
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/sj.bjc.6690802
Keywords
This article is cited by
-
Temozolomide and flavonoids against glioma: from absorption and metabolism to exosomal delivery
Naunyn-Schmiedeberg's Archives of Pharmacology (2024)
-
P2X7 receptor antagonism by AZ10606120 significantly reduced in vitro tumour growth in human glioblastoma
Scientific Reports (2023)
-
A facile and scalable in production non-viral gene engineered mesenchymal stem cells for effective suppression of temozolomide-resistant (TMZR) glioblastoma growth
Stem Cell Research & Therapy (2020)
-
Bioequivalence study of 20-mg and 100-mg temozolomide capsules (TOZ309 and Temodal®) in glioma patients in China
Cancer Chemotherapy and Pharmacology (2020)
-
Interleukin-8/CXCR2 signaling regulates therapy-induced plasticity and enhances tumorigenicity in glioblastoma
Cell Death & Disease (2019)