Summary
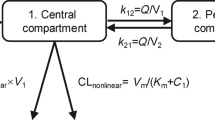
Doxorubicin pharmacokinetics were determined in 33 patients with solid tumours who received intravenous doses of 20–320 mg m–2 HPMA copolymer bound doxorubicin (PK1) in a phase I study. Since assay constraints limited the data at lower doses, conventional analysis was not feasible and a ‘population approach’ was used. Bound concentrations were best described by a biexponential model and further analyses revealed a small influence of dose or weight on V1 but no identifiable effects of age, body surface area, renal or hepatic function. The final model was: clearance (Q) 0.194 l h–1; central compartment volume (V1) 4.48 × (1+0.00074 × dose (mg)) l; peripheral compartment volume (V2) 7.94 l; intercompartmental clearance 0.685 l h–1. Distribution and elimination half-lives had median estimates of 2.7 h and 49 h respectively. Free doxorubicin was present at most sampling times with concentrations around 1000 times lower than bound doxorubicin values. Data were best described using a biexponential model and the following parameters were estimated: apparent clearance 180 l h–1; apparent V1 (l) 1450 × (1+0.0013 × dose (mg)), apparent V2 (l) 21 300 × (1–0.0013 × dose (mg)) × (1+2.95 × height (m)) and apparent Q 6950 l h–1. Distribution and elimination half-lives were 0.13 h and 85 h respectively.
Similar content being viewed by others
Article PDF
Change history
16 November 2011
This paper was modified 12 months after initial publication to switch to Creative Commons licence terms, as noted at publication
References
Amantea, MA, Forrest, A, Northfelt, DW & Mamelok, R (1997). Population pharmacokinetics and pharmacodynamics of pegylated-liposomal doxorubicin in patients with AIDS-related Kaposi’s sarcoma. Clin Pharmacol Ther 61: 301–311.
Beal, SL, Sheiner, LB & Boeckmann, AJ (1994). NONMEM IV User’s Guide, Parts I–VI, NONMEM Project Group, University of California, San Francisco
Bronchud, MH, Margison, JM, Howell, A, Lind, M, Lucas, SB & Wilkinson, PM (1990). Comparative pharmacokinetics of escalating doses of doxorubicin in patients with metastatic breast cancer. Cancer Chemother Pharmacol 25: 435–439.
Bruno, R, Vivier, N, Vergniol, JC, DePhillips, SL, Montay, G & Sheiner, LB (1996). A population pharmacokinetic model for taxotere: model building and validation. J Pharmacokinet Biopharm 24: 153–172.
Chabot, GG, Abigerges, D, Catimel, G, Culine, S, Deforni, R, Extra, JM, Mahjoubi, H, Herait, P, Armand, JP, Bugat, R, Clavel, M & Marty, ME (1995). Population pharmacokinetics and pharmacodynamics of irinotecan (CPT-11) and active metabolite SN-38 during Phase I trials. Ann Oncol 6: 141–151.
Chatelut, E, Canal, P, Brunner, V, Chevreau, C, Pujol, A, Boneu, A, Roche, H, Houin, G & Bugat, R (1995). Prediction of carboplatin clearance from standard morphological and biological patient characteristics. J Natl Cancer Inst 87: 573–580.
Cockcroft, DW & Gault, MH (1976). Prediction of creatinine clearance from serum creatinine. Nephron 16: 31–41.
Dobbs, NA, Twelves, CJ, Gillies, H, James, CA, Harper, PG & Rubens, RD (1995). Gender affects doxorubicin pharmacokinetics in patients with normal liver biochemistry. Cancer Chemother Pharmacol 36: 473–476.
Duncan, R, Cable, HC, Lloyd, JH, Rejmanova, P & Kopecek, J (1982). Degradation of side chains of N-(2-hydroxypropyl)methacrylamide) copolymers by lysosomal thioproteinases. Bioscience Rep 2: 1041–1046.
Fraier, D, Frigerio, E, Pianezzola, E, Strolin Benedetti, M, Cassidy, J & Vasey, P (1995). A sensitive procedure for the quantitation of free and N-(2-hydroxypropyl) methacrylamide polymer-bound doxorubicin (PK1) and some of its metabolites, 13-dihydrodoxorubicin, 13-dihydrodoxorubicine and doxorubicinone, in human plasma and urine by reversed-phase HPLC with fluorimetric detection. J Pharm Biomed Anal 13: 625–633.
Jacquet, J-M, Bressolle, F, Galtier, M, Bourrier, M, Donadio, D, Jourdan, J & Rossi, J-F (1996). Doxorubicin and doxorubicinol: intra- and inter-individual variations of pharmacokinetic parameters. Cancer Chemother Pharmacol 27: 219–225.
Jodrell, DI, Reyno, LM, Sridhara, R, Eisenberger, MA, Tkaczuk, KH, Zuhowski, EG, Sinibaldi, VJ, Novak, MJ & Egorin, MJ (1994). Suramin: development of a population pharmacokinetic model and its use with intermittent short infusions to control plasma drug concentration in patients with prostate cancer. J Clin Oncol 12: 166–175.
Launay Iliadis, MC, Bruno, R, Cosson, V, Vergniol, JC, Oulidaissa, D, Marty, M, Clavel, M, Aapro, M, Lebail, N & Iliadis, A (1995). Population pharmacokinetics of docetaxel during Phase I studies using nonlinear mixed effect modelling and nonparametric maximum likelihood estimation. Cancer Chemother Pharmacol 37: 47–54.
Maeda, H & Matsumura, Y (1989). Tumoritropic and lymphotropic principles of macromolecular drugs. CRC Crit Rev Ther Drug Carrier Sys 6: 193–210.
Matsumura, Y & Maeda, H (1986). A new concept for macromolecular therapeutics in cancer chemotherapy; mechanism of tumoritropic accumulation of proteins and the antitumor agent SMANCS. Cancer Res 46: 6387–6392.
McLeod, HL, Graham, MA, Aamdal, S, Setanoians, A, Groot, Y & Lund, B EORTC Early Clinical Trials Group (1996). Phase I pharmacokinetics and limited sampling strategies for the bioreductive alkylating drug EO9. Eur J Cancer 32A: 1518–1522.
Nguyen, L, Chatelut, E, Chevreau, C, Tranchand, B, Lochon, I, Bachaud, JM, Pujol, A, Houin, G, Bugat, R & Canal, P (1998). Population pharmacokinetics of total and unbound etoposide. Cancer Chemother Pharmacol 41: 125–132.
Piscitelli, SC, Rodvold, KA, Rushing, DA & Tewksbury, DA (1993). Pharmacokinetics and pharmacodynamics of doxorubicin in patients with small cell lung cancer. Clin Pharmacol Ther 53: 555–561.
Reyno, LM, Egorin, MJ, Eisenberger, MA, Sinibaldi, VJ, Zuhowski, EG & Sridhara, R (1995). J Clin Oncol 13: 2187–2195.
Samara, E & Granneman, R (1997). Role of population pharmacokinetics in drug development. A pharmaceutical industry perspective. Clin Pharmacokinet 32: 294–312.
Schoemaker, RC & Cohen, AF (1996). Estimating impossible curves using NONMEM. Br J Clin Pharmacol 42: 283–289.
Tett, SE, Holford, NHG & McLachlan, AJ (1998). Population pharmacokinetics and pharmacodynamics: an underutilised resource. Drug Inf J 32: 693–710.
Sheiner, LB, Rosenberg, B & Marathe, VV (1977). Estimation of population characteristics of pharmacokinetic parameters from routine clinical data. J Pharmacokinet Biopharm 5: 445–479.
Yuh, L, Beal, S, Davidian, M, Harrison, F, Hester, A, Kowalski, K, Vonesh, E & Wolfinger, R (1994). Population pharmacokinetic/pharmacodynamic methodology and applications: a bibliography. Biometrics 50: 566–575.
Vasey, PA, Kaye, SB, Morrison, R, Twelves, C, Wilson, P, Duncan, R, Thomson, AH, Murray, L, Hilditch, TE, Murray, T, Burtles, S, Fraier, D, Frigero, E & Cassidy, J (1999). Phase I clinical and pharmacokinetic study of PK1 (HPMA CO-Polymer doxorubicin); first member of a new class of chemotherapeutic agents – drug-polymer conjugates. Clin Cancer Res 5: 83–94.
Author information
Authors and Affiliations
Rights and permissions
From twelve months after its original publication, this work is licensed under the Creative Commons Attribution-NonCommercial-Share Alike 3.0 Unported License. To view a copy of this license, visit http://creativecommons.org/licenses/by-nc-sa/3.0/
About this article
Cite this article
Thomson, A., Vasey, P., Murray, L. et al. Population pharmacokinetics in phase I drug development: a phase I study of PK1 in patients with solid tumours. Br J Cancer 81, 99–107 (1999). https://doi.org/10.1038/sj.bjc.6690657
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/sj.bjc.6690657
Keywords
This article is cited by
-
The expanding role of prodrugs in contemporary drug design and development
Nature Reviews Drug Discovery (2018)
-
Application of population pharmacokinetic modeling in early clinical development of the anticancer agent E7820
Investigational New Drugs (2009)
-
Virotherapy of Ovarian Cancer With Polymer-cloaked Adenovirus Retargeted to the Epidermal Growth Factor Receptor
Molecular Therapy (2008)
-
Nanoparticle therapeutics: an emerging treatment modality for cancer
Nature Reviews Drug Discovery (2008)
-
Polymer conjugates as anticancer nanomedicines
Nature Reviews Cancer (2006)