Abstract
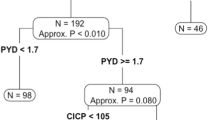
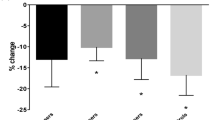
This study was designed to evaluate new bone resorption and tumour markers as possible alternatives to serial plain radiographs for the assessment of response to treatment. Thirty-seven patients with newly diagnosed bone metastases from breast cancer, randomized to receive oral pamidronate or placebo tablets in addition to anticancer treatment within the context of a multicentre EORTC trial, who were both assessable for radiographic response in bone and had serum and urine samples collected for more than 1 month were studied. The markers of bone metabolism measured included urinary calcium (uCa), hydroxyproline (hyp), the N-telopeptide cross-links of type I collagen (NTx) and total alkaline phosphatase. The tumour markers measured were CA15-3 and cancer-associated serum antigen (CASA). Before treatment, levels of Ntx, uCa and Hyp were elevated in 41%, 24% and 28% respectively, and CA15-3 and CASA increased in 69% and 50%. For assessment of response and identification of progression, Ntx was the most useful bone marker. All markers behaved similarly in no change (NC) and partial response (PR) patients. There was a significant difference (P ≤ 0.05) in Ntx levels (compared to baseline) at 1 and 4 months and in CA15-3/CASA at 4 months between patients with PR or NC and those with progressive disease (PD), and at 4 months between those with time to progression (TP) > 7 and those with TP ≤ 7 months. The diagnostic efficiency (DE) for prediction of PD following a > 50% increase in Ntx or CA15-3 was 78% and 62% respectively. An algorithm to predict response to therapy has been developed for future prospective evaluation.
Similar content being viewed by others
Article PDF
Change history
16 November 2011
This paper was modified 12 months after initial publication to switch to Creative Commons licence terms, as noted at publication
References
Blomqvist, C., Sarna, S. & Elomaa, I. et al ( (1996) Markers of type I collagen degradation and synthesis in the monitoring of treatment response in bone metastases from breast carcinoma. Br J Cancer 73: 1074–1079.
Blumsohn, A., Naylor, R. A. & Eastell, R. et al ( (1994) The effects of calcium supplementation on the circadian rhythm of bone resorption. J Clin Endoc Metab 79: 730–735.
Bonde, M., Qvist, P. & Christiansen, C. et al ( (1995) Applications of an enzyme immunoassay for a new marker of bone resorption (Crosslaps): follow-up on hormone replacement therapy and osteoporosis risk assessment. J Clin Endocrinol Metab 80: 864–868.
Campbell, F. C., Blamey, R. W., Woolfson, A. M. J., Elston, C. W. & Hosking, D. T. (1983) Calcium excretion CaE in metastatic breast cancer. Br J Surg 70: 202–204.
Coleman, R. E. (1994) Evaluation of bone disease in breast cancer. Breast 3: 73–78.
Coleman, R. E. (1999) Monitoring of bone metastases. Eur J Cancer 34: 252–259.
Coleman, R. E. & Rubens, R. D. (1985) Bone metastases and breast cancer. Cancer Treat Rev 12: 251–270.
Coleman, R. E., Whitaker, K. B., Moss, D. W., Mashiter, G., Fogelman, I. & Rubens, R. D. (1988) Biochemical prediction of response of bone metastases to treatment. Br J Cancer 58: 205–210.
Downey, S., Eastell, R. & Howell, A. et al ( (1994) Pyridinoline excretion can monitor bone metastases in breast cancer. Breast Cancer Res Treat 32: 83
Galasko, C. S. B. (1994). Diagnosis of skeletal metastases and assessment of response to treatment. In Metastatic Bone Disease, Diel IJ, Kaufmann M and Bastert G (eds), pp. 93–107. Springer Verlag: Heidelberg
Hanson, D., Weis, M. A. E. & Bollen, A. M. et al ( (1992) A specific immunoassay for monitoring human bone resorption: quantification of type 1 collagen cross-linked N-telopeptides in urine. J Bone Miner Res 7: 1251–1258.
Hayward, J. L., Carbone, P. P., Heuson, J. C., Kumaoka, S., Segaloff, A. & Rubens, R. D. (1977) Assessment of response to therapy in advanced breast cancer. Cancer 3: 1389–1394.
Howell, A., Mackintosh, J., Jones, M., Radford, J., Wagstaff, J. & Selwood, R. A. (1988) The definition of the ‘no change’ category in patients treated with endocrine therapy and chemotherapy for 6 advanced carcinoma of the breast. Eur J Cancer 24: 1567–1572.
Lipton, A., Hortobagyi, G. & Reitsma, D. et al ( (1995) Markers of bone resorption in patients treated with pamidronate. Bone 17: 615
Robertson, J. F. R., Pearson, D., Price, M. R., Selby, C., Blamey, R. & Howell, A. (1991) Objective assessment of therapeutic response in breast cancer using tumour markers. Br J Cancer 64: 757–763.
Vinholes, J. J., Guo, C-Y, Purohit, O. P., Eastell, R. & Coleman, R. E. (1996) Metabolic effects of pamidronate in patients with metastatic bone disease. Br J Cancer 73: 1089–1095.
Vinholes, J., Guo, C-Y, Purohit, O. P., Eastell, R. & Coleman, R. E. (1997a) Evaluation of new bone resorption markers in a randomized comparison of pamidronate or clodronate for hypercalcaemia of malignancy. J Clin Oncol 15: 131–138.
Vinholes, J., Purohit, O. P., Abbey, M. E., Eastell, R. & Coleman, R. (1997b) Relationships between biochemical and symptomatic response in a double-blind randomised trial of pamidronate for metastatic bone disease. Ann Oncol 8: 1243–1250.
Author information
Authors and Affiliations
Rights and permissions
From twelve months after its original publication, this work is licensed under the Creative Commons Attribution-NonCommercial-Share Alike 3.0 Unported License. To view a copy of this license, visit http://creativecommons.org/licenses/by-nc-sa/3.0/
About this article
Cite this article
Vinholes, J., Coleman, R., Lacombe, D. et al. Assessment of bone response to systemic therapy in an EORTC trial: preliminary experience with the use of collagen cross-link excretion. Br J Cancer 80, 221–228 (1999). https://doi.org/10.1038/sj.bjc.6690506
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/sj.bjc.6690506
Keywords
This article is cited by
-
Strontium-89 plus zoledronic acid versus zoledronic acid for patients with painful bone metastatic breast cancer
Journal of Bone and Mineral Metabolism (2022)
-
Phase II studies of two different schedules of dasatinib in bone metastasis predominant metastatic breast cancer: SWOG S0622
Breast Cancer Research and Treatment (2016)
-
Randomised, phase II, placebo-controlled, trial of fulvestrant plus vandetanib in postmenopausal women with bone only or bone predominant, hormone-receptor-positive metastatic breast cancer (MBC): the OCOG ZAMBONEY study
Breast Cancer Research and Treatment (2014)
-
Can bone markers guide more effective treatment of bone metastases from breast cancer?
Breast Cancer Research and Treatment (2006)
-
Clinical use of markers of bone turnover in metastatic bone disease
Nature Clinical Practice Oncology (2005)