Abstract
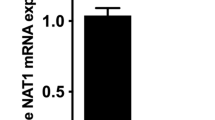
Therapeutic and environmental aromatic amines and hydrazines are substrates for the arylamine N-acetyltransferases (NAT). In all, 10 transgenic lines containing either the human NAT1 or NAT2 transgene were developed using multiple promoters. The presence of the transgene was confirmed by determining copy number, mRNA and enzyme activity. Despite some lines having high copy numbers of the transgene, only modest or no increases in enzymatic activity could be found in a variety of tissues. The NAT1 transgene could not be bred to homozygosity. The cytomegalovirus (CMV)-promoted NAT1 transgene increased endogenous Nat1 mRNA levels in liver and had little effect on endogenous Nat2 mRNA levels. The presence of the CMV-promoted NAT2 transgene appeared to suppress endogenous hepatic Nat2 mRNA, but did not alter Nat1 mRNA levels. The failure to achieve high expression of any of the transgenes suggests that overexpression of NAT genes may have harmful effects during development.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 6 print issues and online access
$259.00 per year
only $43.17 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout
Similar content being viewed by others
References
Hein DW . Molecular genetics and function of NAT1 and NAT2: role in aromatic amine metabolism and carcinogenesis. Mutat Res 2002; 506/507: 65–77.
Glowinski IB, Weber WW, Fysh JM, Vaught JB, King CM . Evidence that arylhydroxamic acid N, O-acetyltransferase and the genetic polymorphic N-acetyltransferase are properties of the same enzyme in rabbit liver. J Biol Chem 1980; 255: 7883–7890.
Flammang TJ, Kadlubar FF . Acetyl coenzyme A-dependent metabolic activation of N-hydroxy-3,2′-dimethyl-4-aminobiphenyl and several carcinogenic N-hydroxy arylamines in relation to tissues and species differences, other acyl donors and arylhydroxamic acid-dependent acetyltransferases. Carcinogenesis 1986; 7: 926–929.
Fretland AJ, Doll MA, Gray K, Feng Y, Hein DW . Cloning, sequencing and recombinant expression of NAT1, NAT2 and NAT3 derived from the C3H/HeJ (rapid) and A/HeJ (slow) acetylator inbred mouse: functional characterization of the activation and detoxification of aromatic amine carcinogens. Toxicol Appl Pharmacol 1997; 1432: 360–366.
Mattano SS, Land S, King CM, Weber WW . Purification and biochemical characterization of hepatic N-acetyltransferase from rapid and slow acetylator mice: identity with arylhydroxamic acid N,O-acetyltransferase and N-hydroxylamine O-acetyltransferase. Mol Pharmacol 1989; 35: 599–609.
Ward A, Summers M, Sim E . Purification of recombinant human NAT1 in E. coli. Biochem. Pharmacol 1995; 49: 1759–1767.
Smelt VA, Upton A, Adjaye J, Payton MA, Boulouvala S, Johnson N et al. Expression of arylamine N-acetyltransferases in pre-term placenta and in human pre-implantation embryos. Hum Mol Genet 2000; 9: 1101–1107.
Glowinski IB, Weber WW . Genetic regulation of aromatic amine N-acetylation in inbred mice. J Biol Chem 1982; 257: 1424–1430.
Mattano SS, Erickson RP, Nesbitt MN, Weber WW . Linkage of Nat and Es-1 in the mouse and development of strains congenic for N-acetyltransferase. J Heredity 1988; 79: 430–433.
Ohsako S, Ohtomi M, Sakamoto Y, Uyemara K, Deguchi T . Arylamine N-acetyltransferase from chicken liver II. Cloning of cDNA and expression in Chinese hamster ovary cells. J Biol Chem 1988; 263: 7534–7538.
Martell KJ, Vatsis KP, Weber WW . Molecular genetic basis of rapid and slow acetylation in mice. Mol Pharmacol 1991; 40: 218–227.
Martell KJ, Levy GN, Weber WW . Cloned mouse N-acetyltransferases: enzymatic properties of expressed Nat-1 and Nat-2 gene products. Mol Pharmacol 1992; 42: 265–272.
Hein DW, Trinidad A, Yerokun T, Ferguson RJ, Kirlin WG, Weber WW . Genetic control of acetyl coenzyme A-dependent arylamine N-acetyltransferase, hydrazine N-acetyltransferase, and N-hydroxy-arylamine O-acetyltransferase enzymes in C57Bl/6, A/J. AC57F1 and the rapid and slow acetylator A.B6 and B6. A congenic inbred mouse. Drug Metab Dispos 1988; 16: 341–347.
Hein DW, Doll MA, Fretland AJ, Leff MA, Webb SJ, Xio GH et al. Molecular genetics and epidemiology of the NAT1 and NAT2 acetylation polymorphisms. Cancer Epidemiol Biomarkers Prev 2000; 9: 29–42.
Estrada-Rodgers L, Levy GN, Weber WW . Substrate selectivity of mouse N-acetyltransferases 1, 2 and 3 expressed in COS-1 cell. Drug Metab Dispos 1998; 26: 502–505.
Payton M, Smelt V, Upton A, Sim E . A method for genotyping murine arylamine N-acetyltransferase type2 (NAT2): a gene expressed in preimplantation embryonic stem cells encoding an enzyme acetylating the folate catabolite p-aminobenzoylglutamate. Biochem Pharmacol 1999; 58: 779–785.
Fakis G, Boukouvala S, Buckle V, Payton M, Denning C, Sim E . Chromosome mapping of the genes for murine arylamine N-acetyltransferases (NATs), enzymes involved in the metabolism of carcinogens: identification of a novel upstream noncoding exon for murine Nat2. Cytogenet Cell Genet 2000; 90: 134–138.
Hein DW, Doll MA, Fretland AJ, Leff MA, Webb SJ, Xio GH et al. Molecular genetics and epidemiology of the NAT1 and NAT2 acetylation polymorphisms. Cancer Epidemiol Biomarkers Prev 2000; 9: 29–42.
Cornish VA, Pinter K, Boukouvala S, Johnson N, Labrousse C, Payton M et al. Generation and analysis of mice with a targeted disruption of the arylamine N-acetyltransferase type 2 gene. Pharmacogenomics 2003; 3: 169–177.
Sugamori KS, Wong S, Gaedigk A, Yu V, Abramovici H, Rozmahel R et al. Generation and functional characterization of arylamine N-acetyltransferase Nat1/Nat2 double-knockout mice. Mol Pharmacol 2003; 64: 170–179.
Leff MA, Epstein PN, Doll MA, Fretland A-J, Udaya-Sankar D, Rustan TD et al. Prostate-specific human N-acetyltransferase 2 (NAT2) expression in the mouse. J Pharmacol Exp Ther 1999; 290: 182–187.
Sim E, Pinter K, Mushtaq A, Upton A, Sandy J, Bhakta S et al. Arylamine N-acetyltransferases: a pharmacogenomics approach to drug metabolism and endogenous function. Biochem Soc Trans 2003; 31: 615–619.
Tannen RH, Weber WW . Rodent models of the human isoniazid acetylator polymorphism. Drug Metab Dispos 1979; 7: 274–279.
Palmiter RD, Brinster RL . Germ-line transformation of mice. Annu Rev Genet 1986; 20: 465–499.
Minchin RF . Acetylation of p-aminobenzoylglutamate, a folic acid catabolite, by recombinant human arylamine N-acetyltransferase and U937 cells. Biochemistry 1995; 307: 1–3.
Sim E, Payton M, Noble M, Minchin R . An update on genetic, structural and functional studies of arylamine N-acetyltransferases in eucaryotes and procaryotes. Hum Mol Genet 2000; 9: 2435–2441.
Gregory III JF, Williamson J, Liao JF, Bailey LB, Toth JP . Kinetic model of folate metabolism in nonpregnant women consuming 2H2 folic acid: isotopic labeling of urinary folate and the catabolite para-acetamidobenzoylglutamate indicates slow, intake-dependent, turnover of folate pools. J Nutr 1998; 128: 1896–1906.
McPartlin J, Courtney G, McNulty H, Weir D, Scott J . The quantitative analysis of endogenous folate catabolites in human urine. Anal Biochem 1992; 206: 256–261.
Schirch V, Strong W . Interaction of folypolyglutamates with enzymes in one-carbon metabolism. Arch Biochem Biophys 1989; 269: 371–380.
Strong WB, Schirch V . In vitro conversion of formate to serine. Effect of tetrahydropteroylpolyglutamates and serine hydroxymethyltransferase on the rate of 10-fomyltetrahydrofolate synthetase. Biochemistry 1989; 28: 9430–9439.
Chu E, Allegra CJ . The role of thymidylate synthase as an RNA binding protein. BioEssays 1996; 18: 191–198.
Kitchens ME, Forsthoefel AM, Rafique Z, Spencer HT, Berger FG . Ligand-mediated induction of thymidylate synthase occurs by enzyme stabilization. J Biol Chem 1999; 274: 12544–12547.
McGuire JJ, Coward JK . Pteroyl-polyglutamates: biosynthesis, degradation and function. In: Blakley RL, Benkovic SJ (eds). Folates and Pterins. Wiley: New York, 1984, pp 135–190.
Blakley RL, Benkovic SJ . Folates and Pterins. Wiley: New York, 1984.
Ebisawa T, Deguchi T . Structure and restriction fragment length polymorphism of genes for human liver N-acetyltransferases. Biochem Biophys Res Commun 1991; 279: 1252–1257.
Butcher N, Arulpragasam A, Pope C, Minchin RF . Identification of a minimal promoter sequence for the human n-acetyltransferase type 1 gene that binds to AP-1 (activator protein 1) and YY-1 (Yin and Yang). Biochem J 2003; 376: 441–448.
Husain A, Barker DF, States JC, Doll MA, Hein DJ . Identification of the major promoter and non-coding exons of the human arylamine N-acetyltransferase 1 gene (NAT1). Pharmacogenetics 2004; 14: 397–406.
Kent KR, Warshawsky D . Xenobiotic inducible regions of the human arylamine N-acetyltransferase 1 and 2 genes. Toxicol Lett 2003; 139: 11–23.
Boukoulava S, Price N, Plant KE, Sim E . Structure and transcriptional regulation of the Nat2 gene encoding for the drug metabolizing enzyme arylamine N-acetyltransferase type 2 in mice. Biochem J 2003; 375: 593–602.
Liu Y, Levy GN . Activation of heterocyclic amines by combinations of prostaglandin H synthase-1 and -2 with N-acetyltransferase 1 and 2. Cancer Lett 1998; 133: 115–123.
Schmidt EV, Christoph G, Zeller R, Leder P . The cytomegalovirus enhancer: a pan-active control element in transgenic mice. Mol Cell Biol 1990; 10: 4406–4411.
Furth PA, Hennighausen L, Baker C, Beatty B, Woychick R . The variability in activity of the universally expressed human cytomegalovirus immediate early gene 1 enhancer/promoter in transgenic mice. Nucleic Acids Res 1991; 19: 6205–6208.
Yull FE, Wallace RM, Clark AJ . Restricted tissue-specific but correct developmental expression mediated by a short human α1AT promoter fragment in transgenic mice. Transgenic Res 1995; 4: 70–74.
Mattano SS, Weber WW . Kinetics of arylamine N-acetyltransferase in tissues from rapid and slow acetylator mice. Carcinogenesis 1987; 8: 133–137.
McQueen CA . Arylamine N-acetyltransferases. In: Costa LG, Maines MD, Reed DJ, Sasa S, Sipes IG (eds). Current Protocols in Toxicology. John Wiley and Sons: New York, 2001, pp 4.6.1–4.6.1.13.
McQueen CA, Chau B, Erickson RP, Tjalkens RB, Philbert MA . The effects of genetic variation in N-acetyltransferases on 4-aminobiphenyl genotoxicity in mouse liver. Chem Biol Interact 2003; 146: 51–60.
Hein DW, Hirata M, Glowinski IB, Weber WW . Biochemical evidence for the coexistence of monomorphic and polymorphic N-acetyltransferase activities on a common protein in rabbit liver. J Pharmacol Exp Ther 1982; 220: 1–7.
Karsai A, Miller S, Platz S, Hauser MT . Evaluation of a home-made SYBR Green I reaction mixture for real-time PCR quantification of gene expression. Biotechniques 2002; 32: 790–796.
Acknowledgements
We thank Drs Gerald Levy and Wendell Weber of the Department of Pharmacology and Dr Kotoku Kurachi, Department of Human Genetics, of the University of Michigan, for constructs and Ms Donelle Myers for secretarial help. This work was supported by ES10047 to CAM and the Genetically Modified Mouse Service, funded by CA23074 and ES06694.
Author information
Authors and Affiliations
Corresponding author
Additional information
DUALITY OF INTEREST
None declared.
Rights and permissions
About this article
Cite this article
Cao, W., Chau, B., Hunter, R. et al. Only low levels of exogenous N-acetyltransferase can be achieved in transgenic mice. Pharmacogenomics J 5, 255–261 (2005). https://doi.org/10.1038/sj.tpj.6500319
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/sj.tpj.6500319