Abstract
Extensive studies in vertebrate cells have assigned a central role to Rel/NF-κB and AP-1 family members in the control of apoptosis. We ask here whether parallel pathways might function in Drosophila by determining if Rel/NF-κB or AP-1 family members contribute to the steroid-triggered death of larval salivary glands during Drosophila metamorphosis. We show that two of the three Drosophila Rel/NF-κB genes are expressed in doomed salivary glands and that one family member, Dif, is induced in a stage-specific manner immediately before the onset of programmed cell death. Similarly, Djun is expressed for many hours before salivary gland cell death while Dfos is induced in a stage-specific manner, immediately before this tissue is destroyed. We show that null mutations in the three Drosophila Rel/NF-κB family members, either alone or in combination, have no apparent effect on this death response. In contrast, Dfos is required for the proper timing of larval salivary gland cell death as well as the proper induction of key death genes. This study demonstrates a role for AP-1 in the stage-specific steroid-triggered programmed cell death of larval tissues during Drosophila metamorphosis.
Similar content being viewed by others
Introduction
Programmed cell death is a critical aspect of metazoan development, providing a means of removing obsolete or damaged cells and sculpting organs to assume their appropriate function. The regulation of programmed cell death is under precise spatial and temporal control in order to ensure that only obsolete or unnecessary cells are destroyed. Accordingly, multiple signal transduction systems act together to direct a programmed cell death response. Rel/NF-κB transcription factors function as critical downstream effectors of these death pathways.1,2 Rel/NF-κB factors form homo- and heterodimers and are defined by the Rel homology domain, a region important for DNA binding, protein dimerization, interaction with cytoplasmic inhibitors, and nuclear translocation.2,3 Prior to their activation, Rel/NF-κB factors are restricted to the cytoplasm via interactions with IκB inhibitor proteins. A wide range of agents activate an IκB kinase complex that phosphorylates IκB, leading to its degradation via a proteasome-mediated pathway.4 This, in turn, frees the Rel/NF-κB factor, allowing it to translocate into the nucleus where it can activate target gene transcription. Rel/NF-κB proteins can either inhibit or promote programmed cell death in a cell-type and signal-dependent manner.1,5,6 Interestingly, these responses have been linked directly to the transcription of downstream effector genes. Thus, NF-κB directly regulates a pro-survival Bcl family member in immune tissues where NF-κB prevents cell death, and NF-κB directly regulates Fas ligand expression in T lymphocytes where this transcription factor drives activation-induced cell death.7,8,9,10,11,12,13
A number of studies have also implicated the AP-1 transcription factor as either a death inhibitor or death trigger depending on cellular context.9,14,15,16 AP-1 is a heterodimer of c-Jun and c-Fos.17 AP-1 activity is regulated by Jun N-terminal kinases (JNKs) that phosphorylate specific serine residues in c-Jun.18 JNKs, in turn, integrate a wide range of upstream signaling events through MAP kinase (MAPK) signaling cascades. Growth factors can activate these pathways through interaction with G protein-coupled receptors, and proinflammatory cytokines can activate MAPK through cell surface receptors. Extracellular stress such as irradiation, toxic drugs, or oxidative stress can also trigger the activation of AP-1 through JNK.16,19,20 The mechanisms by which divergent signals coordinate cellular responses through appropriate regulation of AP-1 activity remains a focus of current research.
Hormones also function as critical regulators of programmed cell death.21,22 For example, glucocorticoids regulate cell death in the central nervous system, sex steroids control cell death in the ovary, and progesterone can inhibit cell death in breast cancer cells.23,24,25 Steroid hormones have also been used for treatment of prostate and breast cancers26,27 and extensive efforts are underway to alter apoptotic signaling pathways in order to control other diseases, including viral infections, autoimmune diseases, and neurodegenerative disorders.28,29,30
We are studying the fruit fly, Drosophila melanogaster, as a model system for defining the hormonal regulation of programmed cell death. The Drosophila genome encodes homologs of all the major players defined in both C. elegans and mammalian cell death pathways, including TRAF, Bcl-2 family members, an Apaf/CED-4 homolog, seven caspase genes, and Inhibitor of Apoptosis (IAP) homologs.31,32,33 In addition, three key death inducer genes were defined in Drosophila, grim, reaper (rpr), and head involution defective (hid).34,35,36 These genes are necessary and sufficient for programmed cell death and appear to function in pairs to trigger the appropriate patterns of cell death during development.34,35,36,37,38 The expression of rpr foreshadows cell death throughout Drosophila development, while hid and grim display close, but not exact correlation. HID activity can be regulated through the Ras pathway.39,40 Recent studies have indicated that the mammalian Smac/DIABLO proteins exert a similar function to rpr, hid, and grim, acting as key triggers of apoptosis by suppressing the ability of IAP proteins to inhibit caspase activation.41,42 Thus, the basic components of the cell death machinery have been faithfully conserved through evolution, from worms and flies to humans.
Drosophila metamorphosis entails the simultaneous death of obsolete larval organs and the growth and differentiation of the adult fly, in response to sequential pulses of the steroid hormone 20-hydroxyecdysone (henceforth called ecdysone).43 A high titer ecdysone pulse at the end of larval development signals the destruction of the larval midgut and anterior muscles as the animal undergoes puparium formation, initiating metamorphosis and the prepupal stage of development.44 This is followed ∼10 h later by another ecdysone pulse that triggers the prepupal-pupal transition and the massive destruction of the larval salivary glands. Most salivary glands are destroyed rapidly in wild-type animals, at ∼15 h after puparium formation.45 Destruction of the larval midgut and salivary glands occurs by steroid-triggered autophagy with hallmark features of cell death, including permeability to the vital dye acridine orange, DNA fragmentation, and caspase activation.45,46,47 In addition, rpr and hid are coordinately induced by ecdysone in a stage- and tissue-specific manner immediately before the onset of larval midgut and salivary gland cell death.45 Genetic studies have identified ecdysone-inducible transcription factors that direct rpr and hid induction in the doomed salivary glands. The zinc finger transcription factors encoded by the Broad–Complex and the E74A ETS domain transcription factor are required for rpr and hid induction.48 Similarly, the stage-specific ecdysone-inducible E93 gene is required for rpr and hid expression, along with the induction of several other key death regulators.49 Moreover, mutations in the BR-C, E74A, or E93 result in defects in larval salivary gland cell death.48,49 Finally, the ecdysone receptor directly induces rpr transcription through a hormone response element in its promoter.48
In this paper, we integrate Drosophila Rel/NF-κB and AP-1 transcription factors into the ecdysone-triggered genetic hierarchy that directs larval salivary gland cell death. The Drosophila genome encodes three Rel/NF-κB transcription factors, Dorsal, Dif, and Relish. Most homodimeric and heterodimeric combinations of these factors can regulate gene expression.50,51,52,53 Like their vertebrate homologs, these proteins can be inhibited by Drosophila IκBs, Cactus and the C-terminus of Relish.52,54,55 Dorsal is required for patterning the dorsal–ventral axis of the early embryo.52 As in vertebrates, the Cactus–Dorsal complex is restricted to the cytoplasm until signaling through the Toll receptor (related to the vertebrate interleukin-1 and Toll-like receptors) results in Cactus phosphorylation and degradation.52 This allows Dorsal to translocate to the nucleus where it can directly activate target gene transcription. In a further parallel with vertebrate Rel/NF-κB function, all three Drosophila family members are key regulators of Drosophila immunity to microbial infection.56,57,58 The striking conservation in the regulation and function of Drosophila Rel/NF-κB family members raises the possibility that these proteins may also play a role in the control of programmed cell death.
Like its mammalian counterpart, Drosophila AP-1 is a heterodimer of Djun and Dfos transcription factors, and its activity is regulated by the Drosophila JNK.59,60,61,62,63,64 Unlike mammals, however, Dfos can function as a homodimer.59 Djun and Dfos are critical regulators of epithelial morphogenetic responses during both mid-embryogenesis and early metamorphosis.60,61,62,63,65,66,67,68
We show here that members of both of the Rel/NF-κB and AP-1 families are induced in larval salivary glands in a stage-specific manner, immediately before the onset of cell death and in synchrony with rpr and hid induction. Although expression of a truncated Dif, with only the Rel homology domain, led to a significant delay in salivary gland cell death, extensive loss-of-function genetic studies did not uncover any direct role for the three Rel/NF-κB proteins in this death pathway. In contrast, we found that Dfos is required for efficient destruction of larval salivary glands as well as maximal levels of rpr and hid transcription. This study demonstrates a role for Dfos in the steroid-triggered death of the Drosophila larval salivary glands during metamorphosis.
Results
Rel/NF-κB and AP-1 transcripts are induced immediately before the onset of larval salivary gland cell death
As a first step toward identifying a possible function for Drosophila Rel/NF-κB and AP-1 family members in programmed cell death, we set out to determine if these genes are expressed in larval salivary glands during the onset of metamorphosis. RNA was isolated from salivary glands dissected from staged late third instar larvae and prepupae and analyzed by Northern blot hybridization (Figure 1). Transcription of the E74A early ecdysone-inducible gene marks the two high titer pulses of ecdysone, in late third instar larvae and late prepupae.69 The second ecdysone-induced peak in E74A expression coincides with the coordinate induction of rpr and hid, consistent with the essential role of E74A in hid induction and salivary gland cell death.48 Dorsal mRNA is not detectable at these stages in larval salivary glands (data not shown). In contrast, Relish mRNA is induced in parallel with the high titer late larval ecdysone pulse, peaks in 2 h prepupal salivary glands, and is then repressed in synchrony with the prepupal ecdysone pulse (Figure 1). Interestingly, Dif is induced as Relish is repressed, and expressed for a brief 2–4 h interval in late prepupal salivary glands, immediately before the onset of cell death (Figure 1). The Drosophila IκB homolog cactus is expressed in parallel with Dif, indicating that other components of the Rel/NF-κB signaling pathway are active at this time. Djun is expressed much like Relish. It is induced in late third instar larval salivary glands and then repressed in late prepupae (Figure 1). In contrast, Dfos is expressed in a stage-specific manner, in parallel with Dif, immediately before the onset of cell death (Figure 1). Thus, Dif and Dfos expression correlates well with the induction of rpr and hid, suggesting that these transcription factors might contribute to the ecdysone-triggered regulatory hierarchy that directs larval salivary gland cell death. Below we describe a series of experiments that test this hypothesis.
Temporal profiles of Rel/NF-κB and AP-1 gene expression before the onset of salivary gland cell death. RNA was isolated from the salivary glands of staged late third instar larvae, at either ∼18, 8, or 4 h before puparium formation, or from prepupae and pupae staged at 2 h intervals after puparium formation. Equal amounts of total RNA were fractionated by formaldehyde agarose gel electrophoresis and transferred to nylon. This blot was sequentially probed and stripped to detect E74A, rpr, hid, Relish, Dif, cactus, Dfos and Djun mRNA. The boxes at the top represent the peaks of the late larval and prepupal ecdysone pulses. Developmental stages are shown at the bottom
Expression of a truncated Dif protein inhibits salivary gland cell death and impairs hid induction
As an initial test of the role of Rel/NF-κB family members in salivary gland cell death, we used a transformant line that expresses a truncated form of Dif under the control of a heat-inducible promoter. This construct, referred to here as P[hs-Dif(RD)], directs expression of the Dif Rel homology domain but lacks the transactivation domain of this protein. P[hs-Dif(RD)] prepupae were selected at 10.5 h after puparium formation, maintained at either 25°C or given a brief heat treatment, and then allowed to recover at room temperature for 3 or 5 h. Salivary glands were dissected from these pupae and the levels of rpr and hid transcription were determined by Northern blot hybridization (Figure 2). Transcription of rpr is not significantly affected in salivary glands expressing Dif(RD), but hid transcription is reduced to approximately one-fifth of the control level (Figure 2). Consistent with this effect, 75% of the animals that express Dif(RD) had persistent salivary glands at 18 h after puparium formation (n=12), 3 h after the destruction of salivary glands in control animals. These results suggest that Rel/NF-κB family members may play a role in salivary gland cell death through the regulation of hid transcription.
Ectopic expression of Dif(RD) results in reduced levels of hid transcription. P[hs-Dif(RD)] 10.5 h prepupae were maintained at either 25 or 37°C for 30 min and allowed to recover at 25°C for 3 or 5 h. Salivary glands were dissected from these 14 and 16 h pupae and the levels of rpr and hid mRNA were determined by Northern blot hybridization
Rel/NF-κB family members are known to function as dimers, and the Rel domain is sufficient to mediate this interaction.50,51,52,53 Thus, the Dif(RD) construct could recruit an active Rel/NF-κB family member to target promoters and thereby act in a manner similar to wild-type Dif, in spite of the absence of its activation domain. To test this possibility, we asked if ectopic overexpression of wild-type Dif would have a similar inhibitory effect on salivary gland cell death. Prepupae carrying both hs-GAL4 and UAS-Dif constructs were subjected to a single 30 min heat treatment at 38°C either 9 or 10 h after puparium formation, and persistent salivary glands were scored 9 h later. Of 49 animals examined, however, only one showed a detectable delay in salivary gland cell death. We thus conclude that Dif(RD) has a novel function that is distinct from that of wild-type Dif protein.
An alternate possibility is that the Dif(RD) construct is functioning as a dominant negative by preventing Rel/NF-κB family members from activating their normal targets in the genome. To test this possibility, we overexpressed a known dominant-negative regulator of Rel/NF-κB signaling. Deletion of the N-terminus of the Cactus protein results in a stable protein that holds Rel/NF-κB family members in the cytoplasm, rendering them unable to bind their genomic targets.55 We thus used either the hsp70 or a GAL4-dependent promoter to express a truncated form of Cactus that is missing the coding region for the N-terminal 125 amino acids.70 Constitutive expression of this protein in larval salivary glands using an appropriate GAL4 driver resulted in normal larval salivary gland cell death, with 6% of the 18 h pupae examined having persistent salivary glands (n=34). This is similar to the 7% persistent salivary glands detected in 18 h pupae of the control genotype (n=14). Heat-induced overexpression of the dominant-negative Cactus protein using a 30 min 38°C heat treatment at 10 h after puparium formation resulted in 11% of the 18 h pupae having persistent salivary glands (n=28). Although this per cent is slightly higher than that seen with a constitutive GAL4 driver, it is within the range that is seen in heat-treated w1118 control animals. Moreover, no reproducible effects were detected on either rpr or hid transcription by Northern blot analysis of salivary gland RNA (data not shown). We thus conclude that the Dif(RD) construct is not functioning in the same pathway as that regulated by the Cactus IκB inhibitor.
Neither Dif nor Relish is required for hid induction in salivary glands
Definitive proof of a role for Rel/NF-κB family members in larval salivary gland cell death requires an examination of null mutants in this pathway. Dorsal has no essential functions during zygotic development. It maps directly adjacent to Dif and, under certain conditions, can replace Dif function in the immune response.57,58 We thus analyzed the phenotypes associated with a deletion for both dorsal and Dif, Df(2L)J4.57 As a control for this study we examined the dlP01313 mutant, which specifically inactivates dorsal function.57 Salivary glands were dissected from staged homozygous dlP01313 and Df(2L)J4 prepupae and expression of rpr and hid was determined by Northern blot hybridization (Figure 3A). Both death genes, however, are induced normally in Df(2L)J4 mutant pupae, indicating that Dif is not required for their expression. In addition, the timing of salivary gland cell death is normal in Df(2L)J4 mutant pupae (data not shown).
Figure 3 Dif and Relish mutations have no effect on rpr and hid transcription in doomed larval salivary glands. (A) Total RNA was isolated from salivary glands dissected from either control dlP01313 or Df(2L)J4 12 and 14 h pupae, and the levels of rpr and hid mRNA were determined by Northern blot hybridization. Df(2L)J4 is a deletion mutation for both dorsal and Dif.57 Hybridization to detect rp49 mRNA was used as a control for loading and transfer. (B) Total RNA was isolated from salivary glands dissected from either control E23 or Df(3R)E20 8, 10 and 12 h pupae, and the levels of rpr and hid mRNA were determined by Northern blot hybridization. Df(3R)E20 is a null mutation for Relish.56 Hybridization to detect rp49 mRNA was used as a control for loading and transfer. The 2 h difference in rpr and hid induction in panels A and B is consistent with the known differences in the timing of the prepupal ecdysone pulse in different genetic backgrounds87
We also examined a possible contribution of Relish to programmed cell death because this Rel/NF-κB family member is expressed in the larval salivary glands before they are destroyed (Figure 1). Salivary glands were dissected from staged Df(3R)E20 Relish mutant and control prepupae,56 and the expression of rpr and hid was determined by Northern blot hybridization (Figure 3B). Again, this study revealed normal patterns of rpr and hid induction, indicating that Relish is not required for death gene expression in doomed larval salivary glands. Moreover, cell death occurred normally in Df(3R)E20 mutant salivary glands (data not shown).
There are no redundant functions between Drosophila Rel/NF-κB family members in salivary gland cell death
One final possibility is that the three Drosophila Rel/NF-κB family members could exert redundant functions in larval salivary gland cell death. Thus, for example, removing either Dif or Relish function alone might not reveal the role of a Rel/NF-κB response in this pathway. Accordingly, we examined the phenotypes associated with a triple mutant that is missing all members of the Drosophila Rel/NF-κB family. Animals carrying the Df(2L)J4 deletion were crossed to Df(3R)E20 animals and homozygotes were established for both deletions. Consistent with the expected weakness of this genetic background and, in particular, its susceptibility to infection, very few of the triple mutants survived until puparium formation. Eleven such animals were selected, staged until 14.5–16 h after puparium formation, and their salivary glands were examined for signs of impending cell death. Of these animals, 18% had intact salivary glands, 18% had no salivary glands, and 64% had salivary glands that were being degraded. These numbers are comparable to those seen in wild-type animals at this stage in development indicating that there is no contribution of Rel/NF-κB family members to larval salivary gland cell death.
To confirm the genetic identity of the stock we had established, RNA was isolated from the triple mutants and analyzed for Dif and Relish expression by Northern blot hybridization (Figure 4). As expected, control flies show normal levels of Dif and Relish (Figure 4, lanes 1,5). In addition, Df(3R)E20 mutants and Df(3R)E20 mutants that are missing one copy of the Dif locus lack Relish mRNA but continue to express Dif (Figure 4, lanes 2,4). Finally, flies carrying both homozygous deficiencies express neither Dif nor Relish (Figure 4, lane 3). These observations confirm the identity of the double deficiency stock and support the conclusion that Rel/NF-κB family members are not required for salivary gland cell death.
Dif and Relish are not expressed in Df(2L)J4; Df(3R)E20 mutants. RNA was isolated from adult flies of the depicted genotypes and Dif and Relish mRNA were detected by Northern blot hybridization. The control samples that express all Rel/NF-κB family members were derived from either w1118 or E23 animals (lanes 1 and 5, respectively). Df(3R)E20 Relish mutants with either one copy (lane 2) or two copies (lane 4) of the dorsal and Dif genes were also tested, as was the Df(2L)J4; Df(3R)E20 double mutant that is missing all Rel/NF-κB family members (lane 3). Hybridization to detect rp49 mRNA was used as a control for loading and transfer
Dfos mutants display defects in larval salivary gland cell death as well as rpr and hid induction
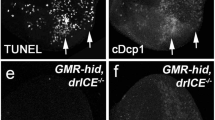
Both halves of the Drosophila AP-1 transcription factor, Dfos and Djun, are expressed in the salivary glands of late prepupae, with a brief burst of Dfos expression immediately preceding the induction of rpr and hid (Figure 1). We therefore tested whether these factors might contribute to salivary gland cell death through regulating rpr and/or hid transcription. Djun mutants die during embryogenesis and thus could not be used for this study.60,61,62 Similarly, most kay alleles lead to embryonic lethality. However, kay2 is a hypomorphic Dfos mutation that allows a few animals (∼1%) to survive to adulthood,63,66 allowing us to analyze the role of Dfos in larval salivary gland cell death. Control kay2/TM6B pupae displayed no defects in salivary gland cell death, with glands either partially or completely destroyed by 16 h after puparium formation (Figure 5). However, 23% of 16 h kay2 mutant pupae and 5% of 17–20 h kay2 mutant pupae have persistent salivary glands (Figure 5). These kay2 mutants underwent ecdysone-triggered adult head eversion at approximately the same time as the control pupae, indicating that there is no overall effect on hormone signaling in this genetic background, or a general developmental delay. We thus conclude that Dfos is required for the proper timing of this death response.
Salivary gland cell death is delayed in kay2 mutants. Salivary glands were dissected from either control kay2/TM6B 16 h pupae, or 16 h and 17–20 h kay2 mutant pupae. kay2 mutant pupae that were delayed in their overall development were discarded. Fully intact, partially destroyed (dissolving), and completely destroyed salivary glands were scored for each genotype and the numbers are presented in graphic format
The observed delay in the destruction of kay2 mutant larval salivary glands suggests that there might be a detectable effect on rpr and/or hid expression in this tissue. To test this possibility, salivary glands were isolated from staged kay2/TM6B control pupae at either 12 or 14 h after puparium formation and kay2 mutant pupae at 14 and 16 h after puparium formation. RNA was extracted from these tissues and the patterns of rpr and hid transcription were analyzed by Northern blot hybridization (Figure 6A). Both rpr and hid are induced normally in control salivary glands, and normal levels of rpr and hid mRNA are detected in 14 h kay2 mutant salivary glands (Figure 6A, lanes 1–3). In salivary glands isolated from 16 h kay2 mutants, however, little or no hid mRNA is detectable and the levels of rpr mRNA appear to be reduced (Figure 6A, lane 4). The absence of any effects on rpr and hid mRNA levels in 14 h kay2 mutant salivary glands is most likely due to the fact that many of these salivary glands would have died on time (Figure 5). In contrast, kay2 mutant glands that have survived to the 16 h timepoint, when all salivary glands from the control genotype have been destroyed (Figure 5), are more likely to reveal a role for Dfos in death gene expression.
Figure 6 Reduced Dfos activity results in lower levels of rpr and hid transcription. (A) Total RNA was isolated from salivary glands of either kay2/TM6B control animals at 12 or 14 h after puparium formation (hours after ppf) or kay2 homozygotes at 14 or 16 h after puparium formation, and the levels of rpr and hid mRNA were determined by Northern blot hybridization. Hybridization to detect rp49 mRNA was used as a control for loading and transfer. (B) Total RNA was isolated from 13 h pupae carrying either one copy of UAS-Fbz (control) or one copy of the D59 salivary gland GAL4 driver and one copy of the UAS-Fbz transgene (Fbz). The levels of rpr and hid mRNA were determined by Northern blot hybridization. Hybridization to detect rp49 mRNA was used as a control for loading and transfer
To confirm this result, we used a salivary gland GAL4 driver to express a dominant negative form of Dfos, Fbz, in the salivary glands of late prepupae.65 RNA was isolated from salivary glands at 13 h after puparium formation and the levels of rpr and hid mRNA were analyzed by Northern blot hybridization (Figure 6B). In agreement with the kay2 loss-of-function study, salivary glands expressing the dominant negative form of Dfos have reduced levels of hid mRNA. Moreover, correcting for the levels of the internal rp49 control, the levels of rpr mRNA also appear to be reduced. Similar results were seen in an independent duplicate experiment (data not shown). We thus conclude that Dfos is required for the proper timing of salivary gland cell death and that it exerts this effect by directing maximal levels of rpr and hid transcription.
Discussion
Several studies have shown that vertebrate steroid hormones can regulate programmed cell death through NF-κB and AP-1 activity.71,72,73,74,75 In all of these cases, however, the downstream targets of NF-κB and AP-1 are not known, and thus it remains unclear how hormonal regulation of these transcription factors initiates a death response. In this study, we ask whether Rel/NF-κB and/or AP-1 family members contribute to the steroid-triggered destruction of the larval salivary glands during Drosophila metamorphosis. Although we find no direct role for Rel/NF-κB family members in this pathway, we do show that AP-1 is required for efficient salivary gland cell death at the prepupal-pupal transition, and that it can mediate this effect through rpr and hid induction. This work provides a foundation for integrating AP-1 activity into the genetic cascade that controls larval salivary gland cell death during Drosophila metamorphosis.
NF-κB and AP-1 family members are induced in a stage-specific manner before the onset of cell death
Drosophila Rel/NF-κB and AP-1 family members all show some apparent regulation by ecdysone in doomed larval salivary glands. Both Relish and Djun are induced in late third instar larval glands, in synchrony with the E74A early ecdysone-inducible gene (Figure 1). Both peak in 2 h prepupae, are down-regulated in 4 h prepupae, and are repressed at 12–14 h after puparium formation, immediately following the prepupal ecdysone pulse. In contrast, Dif, cactus, and Dfos are all induced in a stage-specific manner, in synchrony with the prepupal ecdysone pulse and immediately preceding the induction of rpr and hid and the onset of larval salivary gland cell death (Figure 1). Whereas Dif and cactus are induced in parallel with E74A, Dfos is expressed somewhat earlier, just as E74A and Dif mRNA are first detected (Figure 1). This early induction could be due to increased sensitivity of the Dfos promoter to ecdysone, resulting in induction by a lower hormone concentration than that required for E74A.76
The stage-specific induction of Dif and Dfos provides a model for explaining the stage-specificity of rpr and hid expression in larval salivary glands. The rpr and hid death genes show no response to the late larval pulse of ecdysone but rather are selectively induced by the prepupal ecdysone pulse, ∼12 h after puparium formation (Figure 1).45 The similar stage-specific expression patterns of Dif and Dfos raise the possibility that these factors could direct the timing of rpr and hid induction, providing a molecular mechanism whereby the repetitive ecdysone signal is refined into a stage-specific death response. We test this model and provide evidence of a role for Dfos in larval salivary gland cell death.
A potential role for Rel/NF-κB binding sites in controlling hid expression and cell death
We used five different genetic approaches to determine whether Rel/NF-κB family members might play a role in larval salivary gland cell death. These included ectopic expression of either wild-type Dif, a truncated form of Dif carrying only the DNA binding and dimerization domain, or a dominant-negative form of Cactus that should inactivate all NF-κB responses. In addition we examined larval salivary gland cell death in loss-of-function mutants for either dorsal and Dif, Relish, or all three NF-κB family members. Of these, only ectopic expression of the truncated Dif protein resulted in a delay in larval salivary gland cell death and reduced levels of hid transcription (Figure 2). Unfortunately, the mechanism of action of this mutant form of Dif remains unclear. The absence of any clear effect on salivary gland cell death in the different mutant backgrounds, however, as well as in the presence of dominant-negative Cactus, strongly argues against a direct role for these transcriptional regulators in the salivary gland death response.
The observation that ectopic Dif(RD) expression can lead to death defects while no effect is seen in Rel/NF-κB mutants argues that this truncated protein is functioning in a different pathway. One possibility is that Dif(RD) can occupy Rel/NF-κB binding sites in hid regulatory sequences, thereby blocking the binding of other critical factors. These factors could either recognize the NF-κB site directly or bind to an overlapping target sequence. The latter is more likely since we have tested all three genes predicted by the Drosophila genome sequence to have canonical Rel DNA binding domains – dorsal, Dif, and Relish. The presence of four optimal Rel/NF-κB binding sites within a 28 kb region encompassing the ∼20 kb hid gene supports this model. It would be interesting to determine whether these sites are required for maximal hid induction by ecdysone. Regardless of the mechanism of Dif(RD) action, however, the key conclusion from this study is that Rel/NF-κB family members are not required for the steroid-triggered cell death of larval salivary glands during Drosophila metamorphosis.
Morphological and genetic studies have demonstrated that Drosophila larval salivary glands die by autophagy rather than apoptosis.46,47 Autophagy is characterized by the formation of multiple acidic autophagic vacuoles within the doomed cells followed by massive cellular degeneration. Studies in vertebrate cells have shown that Rel/NF-κB and AP-1 family members play a critical role in controlling apoptosis.1,5,6,9,14,15,16 No studies, however, have addressed a possible role for these factors in autophagic cell death. The results of this work indicate that Rel/NF-κB family members have no function in at least one autophagic death response, and suggest that the activity of this pathway may be specific to apoptotic programmed cell death. Further studies of Rel/NF-κB function in other autophagic death responses should provide insight into its role in regulating programmed cell death during development.
Dfos is required for efficient larval salivary gland cell death
In contrast to our results with Rel/NF-κB family members, we find that the Drosophila homolog of Fos is required for maximal rpr and hid transcription as well as the proper timing of larval salivary gland cell death. Intact larval salivary glands can be detected in 16–20 h kay2 mutant pupae, a time when glands in the control genotype have all entered cell death (Figure 5). In addition, the levels of both rpr and hid are reduced in persistent kay2 mutant salivary glands (Figure 6A, lane 4) as well as salivary glands that express a dominant negative form of Dfos (Figure 6B). It is important to note, however, that this is a partially penetrant phenotype. In 16 h kay2 pupae, 77% of the larval salivary glands are either completely or partially destroyed, and only 5% of the salivary glands persist until 17–20 h after puparium formation (Figure 5). This is similar to the partial salivary gland death defects observed in E74A mutants and supports the proposal that multiple independent pathways are required to direct efficient larval salivary gland cell death.48 We thus conclude that Dfos is required for directing efficient larval salivary gland cell death in response to the prepupal pulse of ecdysone. It is interesting to note that there are three AP-1 binding sites in the 7 kb region of the rpr promoter that confers maximal expression, suggesting that Dfos may directly regulate death gene expression.
Ecdysone triggers two apparently opposite biological responses at the onset of metamorphosis – the death of larval tissues and the growth and differentiation of adult tissues.77 It is interesting to note that AP-1 functions in both of these pathways. The late larval ecdysone pulse triggers morphogenesis of the leg and wing imaginal discs resulting in the formation of rudimentary adult appendages.78 The thoracic portions of the wing discs then fuse during prepupal development to form a smooth intact thorax that is evident on the dorsal surface of the adult fly. This fusion is dependent on AP-1 signaling.67,68 Thus, it appears that AP-1 exerts distinct stage- and tissue-specific functions during the onset of metamorphosis. Initially required for fusion of the thoracic imaginal discs, AP-1 is later required for efficient destruction of the larval salivary glands in response to ecdysone.
It is interesting to note that the results reported here for Dfos in larval salivary gland cell death are similar to those seen in mice. The expression of c-Fos precedes programmed cell death during normal mouse development.79 In spite of this correlation, however, mutant mice lacking c-Jun alone or c-Jun and c-Fos show only minor effects on programmed cell death responses.80,81 But, unlike Drosophila, the presence of multiple AP-1 family members encoded by the mouse genome complicates the interpretation of these results. Further functional studies of AP-1 should provide a better understanding of its roles in regulating programmed cell death during fly and mouse development.
Materials and Methods
Drosophila stocks
Canton S was used as a wild-type stock. The dlP01313 dorsal mutant and Df(2L)J4 dl, Dif double mutant stocks57 were maintained as yw, dlP01313/Cyo, y+ and yw, Df(2L)J4/Cyo, y+ or Df(2L)J4/Cyo GFP. Homozygous dlP01313 and Df(2L)J4 mutant third instar larvae were identified by their yellow mouth hooks. The Relish mutant Df(3R)E20 and its control line E23 have been described and were kindly provided by Dan Hultmark. E23 is a precise excision of a P element inserted in the 5′ end of the Relish gene.56 Df(3R)E20 is an imprecise excision of this P element that removes all four Relish transcription start sites and some 5′ untranslated sequences.56 Both E23 and Df(3R)E20 are homozygous viable. The GAL4/UAS system was used to direct ectopic expression in larval salivary glands.82 hs-GAL4 transformants were obtained from the Bloomington stock center. D59 was used to drive GAL4-dependent expression in larval salivary glands.83 The dominant negative cactus BP4-ΔN125cactΔPEST cDNA was kindly provided by D Stein,70 the kay2 mutant stock was provided by D Bohmann63 and UAS-Fbz transformants were provided by M Bienz.65
Developmental staging
Late third instar larvae were staged by maintaining them on food containing 0.1% bromophenol blue, as described.84 Blue gut mid-third instar larvae are referred as −18 h relative to puparium formation, partially clear gut late third instar larvae are referred to as −8 h, and clear gut late third instar larvae are referred to as −4 h. Prepupae and pupae were staged relative to puparium formation (0 h) as described.84
Ectopic expression studies
The P[hs-Dif(RD)] construct was generated by cutting the Dif cDNA with NdeI (artificially generated at codon 16)53 and EcoRV (at codon 432). The 1.25 kb fragment was inserted into the StuI site of the pCaSpeR-hs vector by blunt end ligation. This construct removes the C-terminal 235 amino acids of Dif which should function as the transcriptional activation domain. This construct was introduced into yw67 flies by P element-mediated germline transformation.85 To ectopically express Dif(RD), P[hs-Dif(RD)] transformants were synchronized at puparium formation and allowed to develop at 25°C for 10.5 h. They were incubated in a 37°C water bath for 30 min, and allowed to recover at 25°C for 3 or 5 h, after which their salivary glands were dissected. As a control, transformants were selected at puparium formation and allowed to develop at 25°C for 14 h before salivary glands were dissected. Total RNA was then extracted from the salivary glands and analyzed by Northern blot hybridization. The cactusΔN125cactΔPEST cDNA70 was inserted into both pCaSpeR-hs-act and pUAST. The vectors were cut with EcoRI, filled-in, and then cut with NotI, and the ΔN125cactΔPEST cDNA was inserted as a 1.5 kb HindIII (filled-in)-NotI fragment. Transgenic lines were established for these constructs by P element-mediated germline transformation of the recipient strain w1118.
Northern blot hybridization
Total RNA isolated from 12 pairs of dissected salivary glands was fractionated by formaldehyde gel electrophoresis and transferred to nylon membranes as described.69 For analysis of the dl, Dif, and Relish triple mutant (Figure 4), 20 μg total RNA from adult flies was loaded per lane. Each blot was sequentially hybridized with one or more radioactive probes and stripped for rehybridization.69 The E74A and rp49 probes were prepared as described;86 rpr and hid probes were prepared as described.45 The following DNA fragments were used to detect Djun, Dfos, Relish, Dif and cactus – Djun: a 300 bp fragment corresponding to the coding region for the first 100 amino acids of the Djun protein; Dfos: a 1195 bp BamHI fragment; Relish: an 804 bp BamHI–HindIII fragment; Dif: an 855 bp BamHI–EcoRI fragment; cactus: an 1162 bp BglII fragment.
Abbreviations
- JNKs:
-
Jun N-terminal kinases
- MAPK:
-
MAP kinase, hid, head involution defective
- RPR:
-
Drosophila grim reaper
References
Van Antwerp D, Martin S, Verma I, Green DR . 1998 Inhibition of TNF-induced apoptosis by NF-κB Trends Cell. Biol. 8: 107–111
Mercurio F, Manning AM . 1999 Multiple signals converging on NF-κB Curr. Biol. 11: 226–232
Baeuerle PA, Baltimore D . 1996 NF-κB: ten years after Cell 87: 13–20
May MJ, Ghosh S . 1997 NF-κB and IκB proteins: An overview Semin. Cancer Biol. 8: 63–73
Foo S, Nolan G . 1999 NF-kB to the rescue: RELs, apoptosis and cellular transformation Trends Genet. 15: 229–235
Baichwal VR, Baeuerle PA . 1997 Activate NF-kappa B or die? Curr. Biol. 7: R94–R96
Chen C, Edelstein LC, Gélinas C . 2000 The Rel/NF-κB family directly activates expression of the apoptosis inhibitor Bcl-xL Mol. Cell. Biol. 20: 2687–2695
Grumont RJ, Rourke IJ, Gerondakis S . 1999 Rel-dependent induction of A1 transcription is required to protect B cells from antigen receptor ligand-induced apoptosis Genes Dev. 13: 400–411
Kasibhatla S, Brunner T, Genestier L, Echeverri F, Mahboubi A, Green DR . 1998 DNA damaging agents induce expression of Fas ligand and subsequent apoptosis in T lymphocytes via the activation of NF-κB and AP-1 Mol. Cell. 1: 543–551
Kasibhatla S, Genestier L, Green DR . 1999 Regulation of Fas-ligand expression during activation-induced cell death in T lymphocytes via nuclear factor κB J. Biol. Chem. 274: 987–992
Matsui K, Fine A, Zhu B, Marshak-Rothstein A, Ju ST . 1998 Identification of two NF-κB sites in mouse CD95 ligand (Fas ligand) promoter: functional analysis in T cell hybridoma J. Immunol. 161: 3469–3473
Schneider A, Martin-Villalba A, Weih F, Vogel J, Wirth T, Schwaninger M . 1999 NF-κB is activated and promotes cell death in focal cerebral ischemia Nat. Med. 5: 554–559
Zong WX, Edelstein LC, Chen C, Bash J, Gelinas C . 1999 The prosurvival Bcl-2 homolog Bfl-1/A1 is a direct transcriptional target of NF-κB that blocks TNFα-induced apoptosis Genes Dev. 13: 382–387
Herdegen T, Skene P, Bahr M . 1997 The c-Jun transcription factor-bipotential mediator of neuronal death, survival and regeneration Trends Neurosci. 20: 227–231
Jehn BM, Osborne BA . 1997 Gene regulation associated with apoptosis Crit. Rev. Eukaryot. Gene Expr. 7: 179–193
Tournier C, Hess P, Yang D, Xu J, Turner T, Nimnual A, Bar-Sagi D, Jones S, Flavell R, Davis R . 2000 Requirement of JNK for stress-induced activation of the cytochrome c-mediated death pathway Science 288: 870–874
Karin M, Liu Z, Zandi E . 1997 AP-1 function and regulation Curr. Opin. Cell. Biol. 9: 240–246
Davis RJ . 2000 Signal transduction by the JNK group of MAP kinases Cell 103: 239–252
Basu S, Kolesnick R . 1998 Stress signals for apoptosis: ceramide and c-Jun kinase Oncogene 17: 3277–3285
Verheij M, Bose R, Lin XH, Yao B, Jarvis WD, Grant S, Birrer MJ, Szabo E, Zon LI, Kyriakis JM, Haimovitz-Friedman A, Fuks Z, Kolesnick RN . 1996 Requirement for ceramide-initiated SAPK/JNK signalling in stress-induced apoptosis Nature 380: 75–79
Evans-Storms RB, Cidlowski JA . 1995 Regulation of apoptosis by steroid hormones J. Steroid Biochem. Mol. Biol. 53: 1–8
Kiess W, Gallaher B . 1998 Hormonal control of programmed cell death/apoptosis Eur. J. Endocrinol. 138: 482–491
Chun SY, Eisenhauer KM, Minami S, Billig H, Perlas E, Hsueh AJ . 1996 Hormonal regulation of apoptosis in early antral follicles: follicle-stimulating hormone as a major survival factor Endocrinology 137: 1447–1456
Gould E, Cameron HA . 1996 Regulation of neuronal birth, migration and death in the rat dentate gyrus Dev. Neurosci. 18: 22–35
Michna H, Nishino Y, Neef G, McGuire WL, Schneider MR . 1992 Progesterone antagonists: tumor-inhibiting potential and mechanism of action J. Steroid Biochem. Mol. Biol. 41: 339–348
Moss HB . 1992 Endocrine therapy for advanced breast cancer: a review Breast Cancer Res. Treat. 21: 15–26
Schulman CC . 1994 Neoadjuvant androgen blockade prior to prostatectomy: a retrospective study and critical review Prostate 5 Suppl: 9–14
Gibbons GH, Dzau VJ . 1996 Molecular therapies for vascular diseases Science 272: 689–693
Rudin CM, Thompson CB . 1997 Apoptosis and disease: regulation and clinical relevance of programmed cell death Annu. Rev. Med. 48: 267–281
Thompson CB . 1995 Apoptosis in the pathogenesis and treatment of disease Science 267: 1456–1462
Abrams JM . 1999 An emerging blueprint for apoptosis in Drosophila Trends Cell. Biol. 9: 435–440
Aravind L, Dixit VM, Koonin EV . 2001 Apoptotic molecular machinery: vastly increased complexity in vertebrates revealed by genome comparisons Science 291: 1279–1284
Bangs P, White K . 2000 Regulation and execution of apoptosis during Drosophila development Dev. Dyn. 218: 68–79
Chen P, Nordstrom W, Gish B, Abrams JM . 1996 grim, a novel cell death gene in Drosophila Genes Dev. 10: 1773–1782
Grether ME, Abrams JM, Agapite J, White K, Steller H . 1995 The head involution defective gene of Drosophila melanogaster functions in programmed cell death Genes Dev. 9: 1694–1708
White K, Grether ME, Abrams JM, Young L, Farrell K, Steller H . 1994 Genetic control of programmed cell death in Drosophila Science 264: 677–683
Robinow S, Draizen T, Truman J . 1997 Genes that induce apoptosis: Transcriptional regulation in identified, doomed neurons of the Drosophila CNS Dev. Biol. 190: 206–213
Zhou L, Schnitzler A, Agapite J, Schwartz LM, Steller H, Nambu JR . 1997 Cooperative functions of the reaper and head involution defective genes in the programmed cell death of Drosophila central nervous system midline cells Proc. Natl. Acad. Sci. USA 94: 5131–5136
Bergmann A, Agapite J, McCall K, Steller H . 1998 The Drosophila gene hid is a direct molecular target of Ras-dependent survival signaling Cell 95: 331–341
Kurada P, White K . 1998 Ras promotes cell survival in Drosophila by downregulating hid expression Cell 95: 319–329
Chai J, Du C, Wu JW, Kyin S, Wang X, Shi Y . 2000 Structural and biochemical basis of apoptotic activation by Smac/DIABLO Nature 406: 855–862
Ekert P, Silke J, Hawkins C, Verhagen A, Vaux D . 2001 Diablo promotes apoptosis by removing MIHA/XIAP from processed caspase 9 J. Cell. Biol. 152: 483–490
Riddiford LM . 1993 Hormones and Drosophila development. In The Development of Drosophila melanogaster Bate M and Martinez Arias A, eds Cold Spring Harbor, NY: Cold Spring Harbor Laboratory Press pp 899–940
Robertson CW . 1936 The metamorphosis of Drosophila melanogaster, including an accurately timed account of the principal morphological changes J. Morph. 59: 351–399
Jiang C, Baehrecke EH, Thummel CS . 1997 Steroid regulated programmed cell death during Drosophila metamorphosis Development 124: 4673–4683
Lee CY, Baehrecke EH . 2001 Steroid regulation of autophagic programmed cell death during development Development 128: 1443–1455
von Gaudecker B, Schmale EM . 1974 Substrate-histochemical investigations and ultrahistochemical demonstrations of acid phosphatase in larval and prepupal salivary glands of Drosophila melanogaster Cell Tiss. Res. 155: 75–89
Jiang C, Lamblin A-F, Steller H, Thummel CS . 2000 A steroid-triggered transcriptional hierarchy controls salivary gland cell death during Drosophila metamorphosis Mol. Cell. 5: 445–455
Lee CY, Wendel DP, Reid P, Lam G, Thummel CS, Baehrecke EH . 2000 E93 directs steroid-triggered programmed cell death in Drosophila Mol. Cell. 6: 433–443
Han ZS, Ip YT . 1999 Interaction and specificity of Rel-related proteins in regulating Drosophila immunity gene expression J. Biol. Chem. 274: 21355–21361
Dushay MS, Asling B, Hultmark D . 1996 Origins of immunity: Relish, a compound Rel-like gene in the antibacterial defense of Drosophila Proc. Natl. Acad. Sci. USA 93: 10343–10347
Belvin MP, Anderson KV . 1996 A conserved signaling pathway: the Drosophila toll-dorsal pathway Annu. Rev. Cell. Dev. Biol. 12: 393–416
Ip YT, Reach M, Engstrom Y, Kadalayil L, Cai H, Gonzalez-Crespo S, Tatei K, Levine M . 1993 Dif, a dorsal-related gene that mediates an immune response in Drosophila Cell 75: 753–763
Stöven S, Ando I, Kadalayil L, Engström Y, Hultmark D . 2000 Activation of the Drosophila NF-κB factor Relish by rapid endoproteolytic cleavage EMBO Rep. 1: 347–352
Belvin MP, Jin Y, Anderson KV . 1995 Cactus protein degradation mediates Drosophila dorsal-ventral signaling Genes Dev. 9: 783–793
Hedengren M, Asling B, Dushay MS, Ando I, Ekengren S, Wihlborg M, Hultmark D . 1999 Relish, a central factor in the control of humoral but not cellular immunity in Drosophila Mol. Cell. 4: 827–837
Meng X, Khanuja BS, Ip YT . 1999 Toll receptor-mediated Drosophila immune response requires Dif, an NF-κB factor Genes Dev. 13: 792–797
Rutschmann S, Jung AC, Hetru C, Reichhart JM, Hoffmann JA, Ferrandon D . 2000 The Rel protein DIF mediates the antifungal but not the antibacterial host defense in Drosophila Immunity 12: 569–580
Perkins KK, Admon A, Patel N, Tjian R . 1990 The Drosophila Fos-related AP-1 protein is a developmentally regulated transcription factor Genes Dev. 4: 822–834
Sluss HK, Han Z, Barrett T, Goberdhan DC, Wilson C, Davis RJ, Ip YT . 1996 A JNK signal transduction pathway that mediates morphogenesis and an immune response in Drosophila Genes Dev. 10: 2745–2758
Hou XS, Goldstein ES, Perrimon N . 1997 Drosophila Jun relays the Jun amino-terminal kinase signal transduction pathway to the Decapentaplegic signal transduction pathway in regulating epithelial cell sheet movement Genes Dev. 11: 1728–1737
Kockel L, Zeitlinger J, Staszewski LM, Mlodzik M, Bohmann D . 1997 Jun in Drosophila development: redundant and nonredundant functions and regulation by two MAPK signal transduction pathways Genes Dev. 11: 1748–1758
Zeitlinger J, Kockel L, Peverali FA, Jackson DB, Mlodzik M, Bohmann D . 1997 Defective dorsal closure and loss of epidermal decapentaplegic expression in Drosophila fos mutants EMBO J. 16: 7393–7401
Riesgo-Escovar JR, Jenni M, Fritz A, Hafen E . 1996 The Drosophila Jun-N-terminal kinase is required for cell morphogenesis but not for DJun-dependent cell fate specification in the eye Genes Dev. 10: 2759–2768
Eresh S, Riese J, Jackson DB, Bohmann D, Bienz M . 1997 A CREB-binding site as a target for decapentaplegic signalling during Drosophila endoderm induction EMBO J. 16: 2014–2022
Riesgo-Escovar JR, Hafen E . 1997 Common and distinct roles of DFos and DJun during Drosophila development Science 278: 669–672
Agnes F, Suzanne M, Noselli S . 1999 The Drosophila JNK pathway controls the morphogenesis of imaginal discs during metamorphosis Development 126: 5453–5462
Zeitlinger J, Bohmann D . 1999 Thorax closure in Drosophila: involvement of Fos and the JNK pathway Development 126: 3947–3956
Karim FD, Thummel CS . 1991 Ecdysone coordinates the timing and amounts of E74A and E74B transcription in Drosophila Genes Dev. 5: 1067–1079
Bergmann A, Stein D, Geisler R, Hagenmaier S, Schmid B, Fernandez N, Schnell B, Nüsslein-Volhard C . 1996 A gradient of cytoplasmic Cactus degradation establishes the nuclear localization gradient of the dorsal morphogen in Drosophila Mech. Dev. 60: 109–123
Cerillo G, Rees A, Manchanda N, Reilly C, Brogan I, White A, Needham M . 1998 The oestrogen receptor regulates NF-κB and AP-1 activity in a cell-specific manner J. Steroid Biochem. Mol. Biol. 67: 79–88
Ivanov VN, Nikolic-Zugic J . 1997 Transcription factor activation during signal-induced apoptosis of immature CD4(+)CD8(+) thymocytes. A protective role of c-Fos J. Biol. Chem. 272: 8558–8566
McKay LI, Cidlowski JA . 1998 Cross-talk between NF-κB and the steroid hormone receptors: mechanisms of mutual antagonism Mol. Endocrinol. 12: 45–56
Wissink S, van Heerde EC, vand der Burg B, van der Saag PT . 1998 A dual mechanism mediates repression of NF-κB activity by glucocorticoids Mol. Endocrinol. 12: 355–363
Zhou F, Thompson EB . 1996 Role of c-jun induction in the glucocorticoid-evoked apoptotic pathway in human leukemic lymphoblasts Mol. Endocrinol. 10: 306–316
Karim FD, Thummel CS . 1992 Temporal coordination of regulatory gene expression by the steroid hormone ecdysone EMBO J. 11: 4083–4093
Bodenstein D . 1965 The postembryonic development of Drosophila In Biology of Drosophila Demerec M, ed New York: Hafner Publishing Co. pp 275–367
Fristrom D, Fristrom JW . 1993 The metamorphic development of the adult epidermis In The Development of Drosophila melanogaster Bate M and Martinez Arias A, ed Cold Spring Harbor, NY: Cold Spring Harbor Laboratory Press pp 843–897
Smeyne RJ, Vendrell M, Hayward M, Baker SJ, Miao GG, Schilling K, Robertson LM, Curran T, Morgan JI . 1993 Continuous c-fos expression precedes programmed cell death in vivo Nature 363: 166–169
Eferl R, Sibilia M, Hilberg F, Fuchsbichler A, Kufferath I, Guertl B, Zenz R, Wagner EF, Zatloukal K . 1999 Functions of c-Jun in liver and heart development J. Cell. Biol. 145: 1049–1061
Roffler-Tarlov S, Brown JJ, Tarlov E, Stolarov J, Chapman DL, Alexiou M, Papaioannou VE . 1996 Programmed cell death in the absence of c-Fos and c-Jun Development 122: 1–9
Brand AH, Perrimon N . 1993 Targeted gene expression as a means of altering cell fates and generating dominant phenotypes Development 118: 401–415
Gustafson K, Boulianne GL . 1996 Distinct expression patterns detected within individual tissues by the GAL4 enhancer trap technique Genome 39: 174–182
Andres AJ, Thummel CS . 1994 Methods for quantitative analysis of transcription in larvae and prepupae In Drosophila melanogaster: Practical Uses in Cell and Molecular Biology Goldstein LSB and Fyrberg EA, ed New York: Academic Press pp. 565–573
Rubin GM, Spradling AC . 1982 Genetic transformation of Drosophila with transposable element vectors Science 218: 348–353
Andres AJ, Fletcher JC, Karim FD, Thummel CS . 1993 Molecular analysis of the initiation of insect metamorphosis: a comparative study of Drosophila ecdysteroid-regulated transcription Dev. Biol. 160: 388–404
Karim FD, Guild GM, Thummel CS . 1993 The Drosophila Broad-Complex plays a key role in controlling ecdysone-regulated gene expression at the onset of metamorphosis Development 118: 977–988
Acknowledgements
We thank D Hultmark for providing the E23 control and Df(3R)E20 Relish mutant, D Stein for providing the cactusΔ125 construct, D Bohmann for the kay2 mutant, M Bienz for the UAS-Fbz stock, and the Bloomington Stock Center for providing GAL4 driver stocks. M Lehmann is the recipient of a research fellowship (Le870/4-1) from the Deutsche Forschungsgemeinschaft. This work was supported by National Institutes of Health grant RO1 GM53269 to YT Ip and RO1 GM60954 to CS Thummel.
Author information
Authors and Affiliations
Corresponding author
Additional information
Edited by G. Nunez
Rights and permissions
About this article
Cite this article
Lehmann, M., Jiang, C., Ip, Y. et al. AP-1, but not NF-κB, is required for efficient steroid-triggered cell death in Drosophila. Cell Death Differ 9, 581–590 (2002). https://doi.org/10.1038/sj.cdd.4401003
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/sj.cdd.4401003