Abstract
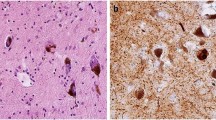
The abnormal aggregation of proteins into fibrillar lesions is a neuropathological hallmark of several sporadic and hereditary neurodegenerative diseases. For example, Lewy bodies (LBs) are intracytoplasmic filamentous inclusions that accumulate primarily in subcortical neurons of patients with Parkinson's disease (PD), or predominantly in neocortical neurons in a subtype of Alzheimer's disease (AD) known as the LB variant of AD (LBVAD) and in dementia with LBs (DLB). Aggregated neurofilament subunits and α-synuclein are major protein components of LBs, and these inclusions may contribute mechanistically to the degeneration of neurons in PD, DLB and LBVAD. Here we review recent studies of the protein building blocks of LBs, as well as the role LBs play in the onset and progression of PD, DLB and LBVAD. Increased understanding of the protein composition and pathological significance of LBs may provide insight into mechanisms of neuron dysfunction and death in other neurodegenerative disorders characterized by brain lesions containing massive deposits of proteinacious fibrils.
Similar content being viewed by others
Article PDF
Author information
Authors and Affiliations
Corresponding author
Additional information
Edited by G. Melino
Rights and permissions
About this article
Cite this article
Trojanowski, J., Goedert, M., Iwatsubo, T. et al. Fatal attractions: abnormal protein aggregation and neuron death in Parkinson's disease and Lewy body dementia. Cell Death Differ 5, 832–837 (1998). https://doi.org/10.1038/sj.cdd.4400432
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/sj.cdd.4400432
Keywords
This article is cited by
-
Involvement of heat shock proteins and parkin/α-synuclein axis in Parkinson’s disease
Molecular Biology Reports (2022)
-
Collusion of α-Synuclein and Aβ aggravating co-morbidities in a novel prion-type mouse model
Molecular Neurodegeneration (2021)
-
Computational insights into the role of α-strand/sheet in aggregation of α-synuclein
Scientific Reports (2019)
-
Cyclized NDGA modifies dynamic α-synuclein monomers preventing aggregation and toxicity
Scientific Reports (2019)
-
Membrane binding, internalization, and sorting of alpha-synuclein in the cell
Acta Neuropathologica Communications (2018)