Abstract
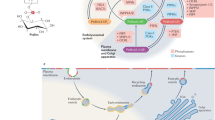
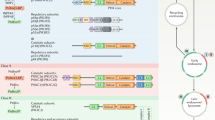
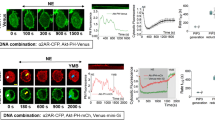
When a stimulatory agonist molecule binds at the exterior of the cell membrane, a second messenger transduces the signal to the interior of the cell. Second messengers can be derived from phospholipids in the membrane by the action of the enzymes phospholipase C or phosphoinositide-3-OH kinase (PI(3)K). PI(3)K is a key player in many cellular responses, including the movement of organelle membranes, shape alteration through rearrangement of cytoskeletal actin, transformation and chemotaxis. But how PI(3)K mediates these responses is only now becoming clear.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 51 print issues and online access
$199.00 per year
only $3.90 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout


Similar content being viewed by others
References
Whitman, M., Downes, C. P., Keeler, M., Keller, T. & Cantley, L. Type I phosphatidylinositol kinase makes a novel inositol phospholipid, phosphatidylinositol-3-phosphate. Nature 332, 644–646 (1988).
Traynor-Kaplan, A. E., Harris, A. L., Thompson, B. L., Taylor, P. & Sklar, L. A. An inositol tetrakisphosphate-containing phospholipid in activated neutrophils. Nature 334, 353–356 (1988).
Franke, T. F. et al. PI3K: downstream AKTion blocks apoptosis. Cell 88, 435–437 (1977).
Chang, H. W. et al. The retroviral oncogene p3k is homologus to the gene encoding for the catalytic subunit of phosphoinositide 3-kinase. Science (in the press).
Morris, J. Z., Tissenbaum, H. A. & Ruvkun, G. Aphosphatidylinositol-3-OH kinase family member regulating longevity and diapause in Caenorhabditis elegans. Nature 382, 536–539 (1996).
Rodriguez, V. P. et al. Phosphatidylinositol-3-OH kinase as a direct target of Ras. Nature 370, 527–532 (1994).
Rameh, L., Chen, C.-S. & Cantley, L. C. Phosphatidylinositol-3,4,5-P3 interacts with SH2 domains and modulates phosphoinositide 3-kinase association with tyrosine-phosphorylated proteins. Cell 83, 821–830 (1995).
Franke, T. F. et al. The protein kinase encoded the Akt proto-oncogene is a target of the PDGF-activated phosphatidylinositol 3-kinase. Cell 81, 1–20 (1995).
Burgering, B. M. & Coffer, P. J. Protein kinase B (c-Akt) in phosphatidylinositol-3-OH kinase signal transduction. Nature 376, 599–602 (1995).
Kohn, A. D., Takeuchi, F. & Roth, R. A. Akt, a pleckstrin homology domain containing kinase, is activated primarily by phosphorylation. J. Biol. Chem. 271, 21920–21926 (1996).
Franke, T. F., Kaplan, D. R., Cantley, L. C. & Toker, A. Direct regulation of the Akt protooncogene product by PI34P2. Science 275, 665–668 (1997).
Klippel, A., Kavanaugh, W. M., Pot, D. & Williams, L. T. Aspecific product of PI 3-K directly activates the protein kinase Akt through its pleckstrin homology domain. Mol. Cell. Biol. 17, 338–344 (1997).
Frech, M. et al. High affinity binding of inositol phosphates and phosphoinositides to the pleckstrin homology domain of RAC/protein kinase B and their influence on kinase activity. J. Biol. Chem. 272, 8474–8481 (1997).
Alessi, D. R. et al. Characterisation of a 3-phosphoinositide-dependent protein kinase which phosphorylates and activates PKB-α Curr. Biol. 7, 261–269 (1997).
Konishi, H. et al. Activation of RAC-protein kinase by heat shock and hyperosmolarity stress through a pathway independent of PI 3-K. Proc. Natl Acad. Sci. U.S.A. 93, 7639–7643 (1996).
Salim, K. et al. Distinct specificity in the recognition of phosphoinositides by the pleckstrin homology domains of dynamin and Btk. EMBO J. 15, 6241–6250 (1996).
Fukuda, M., Kojima, T., Kabayama, H. & Mikoshiba, K. Mutation of the pleckstrin homology domain of BTK in immunodeficiency impaired IP4 binding capacity. J. Biol. Chem. 271, 30303–30306 (1996).
Klarulund, J. K. et al. Signaling by phosphoinositide-3,4,5-trisphosphate through proteins containing pleckstrin and Sec7 Homology domains. Science 275, 1927–1930 (1997).
Kolanus, W. et al. Alpha L beta 2 integrin/LFA-1 binding to ICAM-1 induced by cytohesin-1, a cytoplasmic regulatory molecule. Cell 86, 233–242 (1996).
Chardin, P. et al. Ahuman exchange factor for ARF contains Sec7 and PH domains. Nature 384, 481–484 (1996).
Zhou, M.-M. et al. Structure and ligand recognition of the phosphotyrosine binding domain of Shc. Nature 378, 584–592 (1995).
Rameh, L. E. et al. Phosphoinositide binding specificity of plekcastrin homology domains; the Btk domain specifically binds to PIP3. J. Biol. Chem. (in the press).
Palmer, R. H. et al. Activation of PRK1 by phosphatidylinositol 4,5-bisphosphate and phosphatidylinositol 3,4,5-trisphospate. J. Biol. Chem. 270, 22412–22416 (1995).
Toker, A. et al. Activation of protein kinase C family members by the novel polyphosphoinositides PtdIns-3,4-P2 and PtdIns-3,4,5-P3. J. Biol. Chem. 269, 32358–32367 (1994).
Nakanishi, H., Brewer, K. A. & Exton, J. H. Activation of the zeta isozyme of protein kinase C by phosphatidylinositol 3,4,5-trisphosphate. J. Biol. Chem. 268, 13–16 (1993).
Moriya, S. et al. Platelet-derived growth factor activates protein kinase C-ε through redundant and independent signaling pathways involving phospholipase C-γ or phosphatidylinositol 3-kinase. Proc. Natl Acad. Sci. USA 93, 151–155 (1996).
Akimoto, K. et al. EGF or PDGF receptors activate atypical PKCλ through phosphatidylinositol 3-kinase. EMBO J. 15, 788–798 (1996).
Sossin, W. S., Chen, C.-S. & Toker, A. Stimulation of an insulin receptor activates and downregulates the Ca-independent protein kinase C, Apl II, through a wortmannin sensitive signaling pathway in Aplysia. J. Neurochem. 67, 220–228 (1996).
Toker, A. et al. Phosphorylation of the platelet p47 phosphorprotein is mediated by the lipid products of phosphoinositide 3-kinase. J. Biol. Chem. 270, 29525–29531 (1995).
Lu, P. J. & Chen, C.-S. Selective recognition of PIP3 by a synthetic peptide. J. Biol. Chem. 272, 466–472 (1997).
Derman, M. P. et al. The lipid products of phosphoinositide 3-kinase mediate chemotaxis through protein kinase. J. Biol. Chem. 272, 6465–6470 (1995).
Cross, D. A. et al. The inhibition of glycogen synthase kinase-3 by insulin or insulin-like growth factor 1 in the rat skeletal muscle cell line L6 is blocked by wortmannin, but not by rapamycin: evidence that wortmannin blocks activation of the mitogen-activated protein kinase pathway in L6 cells between Ras and Raf. Biochem. J. 303, 21–26 (1994).
Lopez-Ilasaca, M., Crespo, P., Pellici, P. G., Gutkind, J. S. & Wetzker, R. Linkage of G protein coupled receptors to the MAPK signaling pathway through PI 3-Kγ. Science 275, 394–397 (1997).
Klippel, A. et al. Membrane localization of phosphatidylinositol 3-kinase is sufficient to activate multiple signal-transducing kinase pathway. Mol. Cell. Biol. 16, 4117–4127 (1996).
Schu, P. V. et al. Phosphatidylinositol 3-kinase encoded by yeast VPS34 gene essential for protein sorting. Science 260, 88–91 (1993).
Rapaport, I. et al. Regulatory interactions in the recognition of endocytic sorting signals by AP-2 complexes. EMBO J. 16, 2240–2250 (1997).
Hao, W. et al. Regulation of AP-3 function by inositides: identification of PIP3 as a potent ligand. J.Biol. Chem. 272, 6393–6398 (1997).
Joly, M., Kazlauskas, A., Fay, F. S. & Corvera, S. Disruption of PDGF receptor trafficking by mutation of its PI-3 kinase binding sites. Science 263, 684–687 (1994).
Frevert, E. U. & Kahn, B. B. Differential effects of constitutively active PI 3-K on glucose transport, glycogen synthase activity and DNA synthesis in 3T3-L1 adipocytes. Mol. Cell. Biol. 17, 190–198 (1997).
Schiavo, G., Gu, Q.-M., Prestwich, G. D., Sollner, T. H. & Rothman, J. E. Calcium-dependent switching of the specificity of phosphoinositide binding to synaptotagmin. Proc. Natl Acad. Sci. USA 93, 13327–13332 (1996).
Hammonds-Odie, L. P. et al. Identification and cloning of centaurin-α. J. Biol. Chem. 271, 18859–18868 (1996).
Lu, P. J., Shieh, W.-R., Rhee, S. G., Yin, H. L. & Chen, C.-C. Lipid products of Pi 3-K bind profilin with high affinity. Biochemistry 35, 14027–14034 (1997).
Hartwig, J. H. et al. D3 phosphoinositides and outside-in integrin signaling by GPIIbIIIa mediate platelet actin assembly and filpidial extension induced by PMA. J. Biol. Chem. 271, 32986–32993 (1996).
Hartwig, J. H. et al. Thrombin receptor-ligation and activated Rac uncap actin filament barbed ends through phosphoinositide synthesis in permeabilized human platelets. Cell 82, 1–20 (1995).
Wennstrom, S. et al. Activation of phosphoinositide 3-kinase is required for PDGF-stimulated membrane ruffling. Curr. Biol. 4, 385–393 (1994).
Hawkins, P. T. et al. PDGF stimulates an increase in GTP-rac via activation of phosphoinositide 3-kinase. Curr. Biol. 5, 393–403 (1995).
Reif, K., Nobes, C. D., Thomas, G., Hall, A. & Cantrell, D. A. PI 3-K signals activate a selective subset of Rac/Rho-dependent effector pathways. Curr. Biol. 6, 1445–1455 (1996).
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Toker, A., Cantley, L. Signalling through the lipid products of phosphoinositide-3-OH kinase. Nature 387, 673–676 (1997). https://doi.org/10.1038/42648
Issue Date:
DOI: https://doi.org/10.1038/42648
This article is cited by
-
Wound healing effects of Asparagus lucidus Lindl extract through the phosphorylation of ERK1/2
BMC Complementary Medicine and Therapies (2023)
-
Preterm birth alters the feeding-induced activation of Akt signaling in the muscle of neonatal piglets
Pediatric Research (2023)
-
Development of selective inhibitors of phosphatidylinositol 3-kinase C2α
Nature Chemical Biology (2023)
-
Structural basis of phosphatidylinositol 3-kinase C2α function
Nature Structural & Molecular Biology (2022)
-
Class I PI3K regulatory subunits control differentiation of dendritic cell subsets and regulate Flt3L mediated signal transduction
Scientific Reports (2022)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.