Abstract
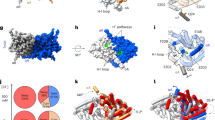
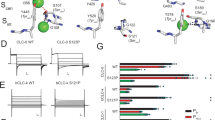
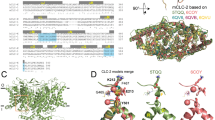
The ClC chloride channels catalyse the selective flow of Cl- ions across cell membranes, thereby regulating electrical excitation in skeletal muscle and the flow of salt and water across epithelial barriers. Genetic defects in ClC Cl- channels underlie several familial muscle and kidney diseases. Here we present the X-ray structures of two prokaryotic ClC Cl- channels from Salmonella enterica serovar typhimurium and Escherichia coli at 3.0 and 3.5 Å, respectively. Both structures reveal two identical pores, each pore being formed by a separate subunit contained within a homodimeric membrane protein. Individual subunits are composed of two roughly repeated halves that span the membrane with opposite orientations. This antiparallel architecture defines a selectivity filter in which a Cl- ion is stabilized by electrostatic interactions with α-helix dipoles and by chemical coordination with nitrogen atoms and hydroxyl groups. These findings provide a structural basis for further understanding the function of ClC Cl- channels, and establish the physical and chemical basis of their anion selectivity.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 51 print issues and online access
$199.00 per year
only $3.90 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout







Similar content being viewed by others
References
Doyle, D. A. et al. The structure of the potassium channel: molecular basis of K+ conduction and selectivity. Science 280, 69–77 (1998).
Zhou, Y., Cabral, J. M., Kaufman, A. & MacKinnon, R. Chemistry of ion coordination and hydration revealed by a K+ channel–Fab complex at 2.0 Å resolution. Nature 414, 43–48 (2001).
Jentsch, T. J., Friedrich, T., Schriever, A. & Yamada, H. The CLC chloride channel family. Pflugers Arch. 437, 783–795 (1999).
Maduke, M., Miller, C. & Mindell, J. A. A decade of CLC chloride channels: structure, mechanism, and many unsettled questions. Annu. Rev. Biophys. Biomol. Struct. 29, 411–438 (2000).
Fahlke, C. Ion permeation and selectivity in ClC-type chloride channels. Am. J. Physiol. Renal Physiol. 280, F748–F757 (2001).
Steinmeyer, K., Lorenz, C., Pusch, M., Koch, M. C. & Jentsch, T. J. Multimeric structure of ClC-1 chloride channel revealed by mutations in dominant myotonia congenita (Thomsen). EMBO J. 13, 737–743 (1994).
White, M. M. & Miller, C. A voltage-gated anion channel from the electric organ of Torpedo californica. J. Biol. Chem. 254, 10161–10166 (1979).
Miller, C. Open-state substructure of single chloride channels from Torpedo electroplax. Phil. Trans. R. Soc. Lond. B 299, 401–411 (1982).
Miller, C. & White, M. M. Dimeric structure of single chloride channels from Torpedo electroplax. Proc. Natl Acad. Sci. USA 81, 2772–2775 (1984).
Jentsch, T. J., Steinmeyer, K. & Schwarz, G. Primary structure of Torpedo marmorata chloride channel isolated by expression cloning in Xenopus oocytes. Nature 348, 510–514 (1990).
Ludewig, U., Pusch, M. & Jentsch, T. J. Two physically distinct pores in the dimeric ClC-0 chloride channel. Nature 383, 340–343 (1996).
Middleton, R. E., Pheasant, D. J. & Miller, C. Homodimeric architecture of a ClC-type chloride ion channel. Nature 383, 337–340 (1996).
Weinreich, F. & Jentsch, T. J. Pores formed by single subunits in mixed dimers of different CLC chloride channels. J. Biol. Chem. 276, 2347–2353 (2001).
Maduke, M., Pheasant, D. J. & Miller, C. High-level expression, functional reconstitution, and quaternary structure of a prokaryotic ClC-type chloride channel. J. Gen. Physiol. 114, 713–722 (1999).
Mindell, J. A., Maduke, M., Miller, C. & Grigorieff, N. Projection structure of a ClC-type chloride channel at 6.5 Å resolution. Nature 409, 219–223 (2001).
White, M. M. & Miller, C. Probes of the conduction process of a voltage-gated Cl- channel from Torpedo electroplax. J. Gen. Physiol. 78, 1–18 (1981).
Rychkov, G. Y., Pusch, M., Roberts, M. L., Jentsch, T. J. & Bretag, A. H. Permeation and block of the skeletal muscle chloride channel, ClC-1, by foreign anions. J. Gen. Physiol. 111, 653–665 (1998).
Rychkov, G., Pusch, M., Roberts, M. & Bretag, A. Interaction of hydrophobic anions with the rat skeletal muscle chloride channel ClC-1: effects on permeation and gating. J. Physiol. 530, 379–393 (2001).
Richard, E. A. & Miller, C. Steady-state coupling of ion-channel conformations to a transmembrane ion gradient. Science 247, 1208–1210 (1990).
Pusch, M., Ludewig, U., Rehfeldt, A. & Jentsch, T. J. Gating of the voltage-dependent chloride channel CIC-0 by the permeant anion. Nature 373, 527–531 (1995).
Chen, T. Y. & Miller, C. Nonequilibrium gating and voltage dependence of the ClC-0 Cl- channel. J. Gen. Physiol. 108, 237–250 (1996).
Ludewig, U., Jentsch, T. J. & Pusch, M. Inward rectification in ClC-0 chloride channels caused by mutations in several protein regions. J. Gen. Physiol. 110, 165–171 (1997).
Ludewig, U., Jentsch, T. J. & Pusch, M. Analysis of a protein region involved in permeation and gating of the voltage-gated Torpedo chloride channel ClC-0. J. Physiol. 498 (part 3), 691–702 (1997).
Maduke, M., Williams, C. & Miller, C. Formation of CLC-0 chloride channels from separated transmembrane and cytoplasmic domains. Biochemistry 37, 1315–1321 (1998).
Maduke, M., Pheasant, D. J. & Miller, C. High-level expression, functional reconstitution, and quaternary structure of a prokaryotic ClC-type chloride channel. J. Gen. Physiol. 114, 713–722 (1999).
Kleywegt, G. J. & Read, R. J. Not your average density. Structure 5, 1557–1569 (1997).
Cowan, S. W. et al. Crystal structures explain functional properties of two E. coli porins. Nature 358, 727–733 (1992).
Murata, K. et al. Structural determinants of water permeation through aquaporin-1. Nature 407, 599–605 (2000).
Fu, D. et al. Structure of a glycerol-conducting channel and the basis for its selectivity. Science 290, 481–486 (2000).
Grigorieff, N., Ceska, T. A., Downing, K. H., Baldwin, J. M. & Henderson, R. Electron-crystallographic refinement of the structure of bacteriorhodopsin. J. Mol. Biol. 259, 393–421 (1996).
Fahlke, C., Yu, H. T., Beck, C. L., Rhodes, T. H. & George, A. L. Jr Pore-forming segments in voltage-gated chloride channels. Nature 390, 529–532 (1997).
Fahlke, C., Beck, C. L. & George, A. L. Jr A mutation in autosomal dominant myotonia congenita affects pore properties of the muscle chloride channel. Proc. Natl Acad. Sci. USA 94, 2729–2734 (1997).
Roux, B. & MacKinnon, R. The cavity and pore helices in the KcsA K+ channel: electrostatic stabilization of monovalent cations. Science 285, 100–102 (1999).
Andersen, O. S. & Fuchs, M. Potential energy barriers to ion transport within lipid bilayers. Biophys. J. 15, 795–830 (1975).
McLaughlin, S. Electrostatic potentials at membrane-solution interfaces. Curr. Top. Membr. Transp. 9, 71–144 (1977).
Jordan, P. C. Electrostatic modeling of ion pores. II. Effects attributable to the membrane dipole potential. Biophys. J. 41, 189–195 (1983).
Otwinowski, Z. & Minor, W. Processing of X-ray diffraction data collected in oscillation mode. Methods Enzymol. 276, 307–326 (1997).
Collaborative Computational Project No. 4. The CCP4 Suite: Programs for X-ray crystallography. Acta Crystallogr. D 50, 760–763 (1994).
de La Fortelle, E. & Bricogne, G. in Methods in Enzymology (eds Carter, C. W. & Sweet, R. M.) 472–494 (Academic, New York, 1997).
Abrahams, J. P. & Leslie, A. G. W. Methods used in the structure determination of bovine mitochondrial F1 ATPase. Acta Crystallogr. D 52, 30–42 (1996).
Vonrhein, C. & Schulz, G. E. Locating proper non-crystallographic symmetry in low-resolution electron-density maps with the program GETAX. Acta Crystallogr. D. Biol. Crystallogr. 55 (part 1), 225–229 (1999).
Kleywegt, G. J. & Jones, T. A. in Proc. CCP4 Study Weekend (eds Bailey, S.. Hubbard, R. & Waller, D.) 59–66 (Daresbury Laboratory, Daresbury, UK, 1994).
Navaza, J. AMoRe: an automated package for molecular replacement. Acta Crystallogr. A 50, 157–163 (1994).
Jones, T. A., Zou, J. Y., Cowan, S. W. & Kjeldgaard, M. Improved methods for building protein models in electron density maps and the location of errors in these models. Acta Crystallogr. A 47, 110–119 (1991).
Brunger, A. T. et al. Crystallography & NMR system: new software suite for macromolecular structure determination. Acta Crystallogr. D 54, 905–921 (1998).
Cowtan, K. DM: an automated procedure for phase improvement by density modification. Joint CCP4 ESF-EACBM Newslett. Protein Crystallogr. 31, 34–38 (1994).
Rocchia, W. et al. Rapid grid-based construction of the molecular surface and the use of induced surface charge to calculate reaction field energies: Application to the molecular systems and geometric objects. J. Comp. Chem. 23, 128–137 (2002).
MacKerell, A. D. J. et al. All-atom empirical potential for molecular modeling and dynamics studies of proteins. J. Phys. Chem. B 102, 3586–3616 (1998).
Thompson, J. D., Higgins, D. G. & Gibson, T. J. CLUSTAL W: improving the sensitivity of progressive multiple sequence alignment through sequence weighting, positions-specific gap penalties and weight matrix choice. Nucleic Acids Res. 22, 4673–4680 (1994).
Sanner, M. F., Olson, A. J. & Spehner, J. C. Reduced surface: an efficient way to compute molecular surfaces. Biopolymers 38, 305–320 (1996).
Acknowledgements
We thank M. Becker and L. Berman at the National Synchrotron Light Source X-25, the staff at the Cornell High Energy Synchrotron Source A1 and F1, and C. Petosa and A. Perrakis of the European Molecular Biology Laboratory at the European Synchrotron Radiation Facility ID-13 for assistance in data collection; J. Bonanno for technical assistance; members of the MacKinnon laboratory for assistance at all stages of the project; and S. Harrison, D. Jeruzalmi, A. Phillippsen and O. Andersen for discussions. This work was supported by grants from the National Institutes of Health (NIH) to R.M. and from the National Center for Research Resources, NIH, to B.T.C. R.M. is an investigator in the Howard Hughes Medical Institute.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Supplementary information
Rights and permissions
About this article
Cite this article
Dutzler, R., Campbell, E., Cadene, M. et al. X-ray structure of a ClC chloride channel at 3.0 Å reveals the molecular basis of anion selectivity. Nature 415, 287–294 (2002). https://doi.org/10.1038/415287a
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1038/415287a
This article is cited by
-
Structural basis of pH-dependent activation in a CLC transporter
Nature Structural & Molecular Biology (2024)
-
Exploring the biosynthetic gene clusters in Brevibacterium: a comparative genomic analysis of diversity and distribution
BMC Genomics (2023)
-
A mechanical-coupling mechanism in OSCA/TMEM63 channel mechanosensitivity
Nature Communications (2023)
-
De novo CLCN3 variants affecting Gly327 cause severe neurodevelopmental syndrome with brain structural abnormalities
Journal of Human Genetics (2023)
-
Aromatic pentaamide macrocycles bind anions with high affinity for transport across biomembranes
Nature Chemistry (2023)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.