Abstract
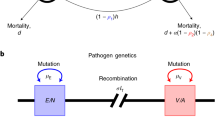
Vaccines rarely provide full protection from disease. Nevertheless, partially effective (imperfect) vaccines may be used to protect both individuals and whole populations1,2,3. We studied the potential impact of different types of imperfect vaccines on the evolution of pathogen virulence (induced host mortality) and the consequences for public health. Here we show that vaccines designed to reduce pathogen growth rate and/or toxicity diminish selection against virulent pathogens. The subsequent evolution leads to higher levels of intrinsic virulence and hence to more severe disease in unvaccinated individuals. This evolution can erode any population-wide benefits such that overall mortality rates are unaffected, or even increase, with the level of vaccination coverage. In contrast, infection-blocking vaccines induce no such effects, and can even select for lower virulence. These findings have policy implications for the development and use of vaccines that are not expected to provide full immunity, such as candidate vaccines for malaria4.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 51 print issues and online access
$199.00 per year
only $3.90 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout



Similar content being viewed by others
References
Anderson, R. M. & May, R. M. Infectious Diseases of Humans (Oxford Univ. Press, Oxford, 1991).
McLean, S. A. & Blower, S. M. Imperfect vaccines and herd immunity to HIV. Proc. R. Soc. Lond. B 256, 9–13 (1993).
Halloran, M. E., Struchiner, C. J. & Spielman, A. Modeling malaria vaccines II: Population effects of stage-specific malaria vaccines dependent on natural boosting. Math. Biosci. 96, 115–149 (1989).
Hoffman, S. L. Malaria Vaccine Development: A Multi-immune Response Approach (American Society for Microbiology Press, Washington DC, 1996).
McLean, A. R. Vaccination, evolution and changes in the efficacy of vaccines: a theoretical framework. Proc. R. Soc. Lond. B 266, 389–393 (1995).
Gupta, S., Ferguson, N. M. & Anderson, R. M. Vaccination and the population structure of antigenically diverse pathogens that exchange genetic material. Proc. R. Soc. Lond. B 266, 1435–1443 (1997).
Lipsitch, M. Vaccination against colonizing bacteria with multiple serotypes. Proc. Natl Acad. Sci. USA 96, 6571–6576 (1997).
CDC. Public health dispatch: outbreak of poliomyelitis—Dominican Republic and Haiti, 2000. Morb. Mortal. Wkly Rep. 46, 1094 (2000).
Zuckerman, A. J. Effect of hepatitis B virus mutants on efficacy of vaccination. Lancet 356, 1382–1384 (2000).
Anderson, R. M. & May, R. M. Co-evolution of hosts and parasites. Parasitology 86, 411–426 (1982).
Levin, S. A. & Pimentel, D. Selection of intermediate rates of increase in parasite-host systems. Am. Nat. 116, 308–315 (1981).
Frank, S. A. Models of parasite virulence. Q. Rev. Biol. 76, 37–78 (1996).
Gandon, S., Jansen, V. A. A. & van Baalen, M. Host life-history and the evolution of parasite virulence. Evolution 56, 1056–1062 (2001).
Nowak, M. A. & May, R. M. Superinfection and the evolution of parasite virulence. Proc. R. Soc. Lond. B 256, 81–89 (1994).
Lipsitch, M. & Moxon, E. R. Virulence and transmissibility of pathogens: what is the relationship? Trends Microbiol. 6, 31–36 (1997).
Mackinnon, M. J. & Read, A. F. Genetic relationships between parasite virulence and transmission in the rodent malaria Plasmodium chabaudi. Evolution 56, 689–703 (1999).
Gandon, S. & Michalakis, Y. Evolution of parasite virulence against qualitative or quantitative host resistance. Proc. R. Soc. Lond. B 266, 985–990 (2000).
van Baalen, M. & Sabelis, M. W. The dynamics of multiple infection and the evolution of virulence. Am. Nat. 146, 881–910 (1995).
May, R. M. & Nowak, M. A. Superinfection, metapopulation dynamics, and the evolution of diversity. J. Theor. Biol. 176, 95–114 (1994).
Caswell, H. Matrix Population Models: Construction, Analysis, and Interpretation (Sinauer, Sunderland, 2001).
Regoes, R. R., Nowak, M. A. & Bonhoeffer, S. Evolution of virulence in a heterogeneous host population. Evolution 56, 64–71 (2000).
Ronce, O. & Kirkpatrick, M. When sources become sinks: migrational meltdown in heterogeneous habitats. Evolution 56, 1520–1531 (2001).
Woolhouse, M. E. J., Taylor, L. H. & Haydon, D. T. Population biology of multihost pathogens. Science 296, 1109–1112 (2001).
Tachado, S. D. et al. Signal transduction in macrophages by glycophosphatidylinositols of Plasmodium, Trypanosoma, and Leishmania: activation of protein tyrosine kinases and protein kinase C by inositolglycan and diacylglycerol moieties. Proc. Natl Acad. Sci. USA 96, 4022–4027 (1997).
Read, A. F. & Taylor, L. H. The ecology of genetically diverse infections. Science 296, 1099–1102 (2001).
Ebert, D. Experimental evolution of parasites. Science 286, 1432–1435 (1998).
Williams, G. & Nesse, R. M. The dawn of Darwinian medicine. Q. Rev. Biol. 66, 1–22 (1991).
Dieckmann, U., Metz, J. A. J., Sabelis, M. W. & Sigmund, K. (eds) Virulence Management: the Adaptive Dynamics of Pathogen-host Interactions (Cambridge Univ. Press, Cambridge, 2001).
Witter, R. L. Avian tumor viruses: persistent and evolving pathogens. Acta Vet. Hung. 46, 251–266 (1997).
Chotivanich, K. T. et al. Parasite multiplication potential and the severity of falciparum malaria. J. Infect. Dis. 186, 1206–1209 (2000).
Acknowledgements
We thank M. van Baalen, R. Carter, D. Ebert, V. Jansen, T. Little, Y. Michalakis, F. Rousset, and S. West for discussions, and the Leverhulme Trust, BBSRC and the Wellcome Trust for support.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Supplementary information

Figure 4
(GIF 3.52 KB)
Schematic representation of the epidemiological model described in equation (5).

Figure 5
(GIF 7.12 KB)
Invasion dynamics of a virulence mutant after the start of vaccination campaign using an anti-growth rate vaccine (r2=0.8 and ri=r3=r4=0) with a vaccination coverage of 90%. In this simulation the virulence of the resident strain is the ES virulence in the absence of vaccination (αN=0.0153). We present the invasion dynamics of invasion of a mutant with virulence equal to the ES level after vaccination (αN=0.0418). At the start of the vaccination campaign we assumed that the mutant was at an initial frequency of 1% (full line), 0.1% (dashed line) or 0.01% (dotted line). We further assumed that its distribution among naïve and immune hosts was identical to the distribution of the resident strain before vaccination.
Rights and permissions
About this article
Cite this article
Gandon, S., Mackinnon, M., Nee, S. et al. Imperfect vaccines and the evolution of pathogen virulence. Nature 414, 751–756 (2001). https://doi.org/10.1038/414751a
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/414751a
This article is cited by
-
Pathogen evasion of social immunity
Nature Ecology & Evolution (2023)
-
Evolutionary implications of SARS-CoV-2 vaccination for the future design of vaccination strategies
Communications Medicine (2023)
-
Generating simple classification rules to predict local surges in COVID-19 hospitalizations
Health Care Management Science (2023)
-
Adapting to vaccination
Nature Ecology & Evolution (2022)
-
Infectious disease in an era of global change
Nature Reviews Microbiology (2022)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.