Abstract
Most mesenchymal tumors of the gastrointestinal tract are currently classified as specific gastrointestinal stromal tumors. However, true leiomyomas are more common in the esophagus, and they have been occasionally noted in the colon and rectum, but the small number of reported cases does not allow for clinicopathologic profiling. This study was undertaken to characterize 88 tumors of the muscularis mucosae of the colon and rectum. Seventy tumors were obtained form the files of AFIP and 18 cases from the Department of Pathology of the Haartman Institute of the University of Helsinki. The lesions, except one, were removed by snare polypectomy as incidental lesions at cancer or polyp surveillance; one small tumor was an incidental finding in the rectal resection specimen. The tumors had a significant male predominance in both institutions (overall 2.4:1). They occurred in age range of 38 to 85 years (median 62 years). The lesions were typically small (range 1 to 22 mm, median 4 mm) and located predominantly in the rectum and sigmoid (72%). All tumors were composed of well-differentiated, eosinophilic smooth muscle cells that were seen immediately beneath the mucosa obliterating the muscularis mucosae layer and merging with it. Two tumors had significant atypia (“symplastic leiomyoma”); mitotic activity was seen in one of these tumors, but not in others. The lesional cells were uniformly positive for smooth muscle actin and desmin and negative for CD34, CD117 and S100-protein, based on immunohistochemical studies on 20 to 24 cases with each marker. No gastrointestinal stromal tumors were identified among the tumors of muscularis mucosae, and no CD117-positive cells, except mast cells, were seen in the muscularis mucosae layer. None of the patients had morbidity related to the tumor. Based on follow-up data on 29 patients, leiomyomas of muscularis mucosae are benign. They should be separated from gastrointestinal stromal tumors that have a clinicopathologic spectrum including frequent disease-related mortality. Snare polypectomy is an adequate treatment, but ensuring the complete removal and follow-up are necessary precautions for tumors with any atypia or mitotic activity.
Similar content being viewed by others
INTRODUCTION
Gastrointestinal stromal tumors (GIST) constitute the largest subset of mesenchymal tumors of the stomach and small intestine, whereas typical leiomyomas are more common in the esophagus (1, 2, 3, 4, 5). Typical leiomyomas have also been occasionally mentioned as colorectal tumors (6, 7, 8, 9, 10, 11, 12, 13), but all these studies antedate the use of CD117 (KIT) as a marker to separate true smooth muscle tumors and GISTs, and sufficient data are not available to establish their clinicopathologic profile. In this study we analyzed 88 tumors of the muscularis mucosae of colon and rectum, and show that they are all leiomyomas different from GISTs. Uniformly benign behavior was documented in this study, even when atypia was present.
MATERIALS AND METHODS
Case Material and Follow-Up
Mesenchymal tumors from the colon and rectum that were coded as leiomyomas, leiomyosarcomas, smooth muscle tumors, or stromal tumors were retrieved from the files of the Armed Forces Institute of Pathology and the Haartman Institute of the University of Helsinki, Helsinki, Finland from 1970 to 1998 (total of 522 tumors).
Eighty-eight tumors arose from or involved the muscularis mucosae only, and they all were composed of well-differentiated smooth muscle cells. There were no GISTs among the tumors limited to muscularis mucosae. In the same material in the two institutions, there were 37 colonic and 133 rectal primary GISTs, and 51 additional GISTs secondarily involving these sites. The series of colonic GISTs has been previously published (14). All except 2 of the 88 leiomyomas originally were diagnosed as leiomyomas; two tumors with atypia were diagnosed as smooth muscle tumors of uncertain malignant potential. Hematoxylin and eosin-stained slides (one to two slides of each case) were reviewed. Mitoses were searched for in each lesion.
Clinical data were reviewed from the records, and the follow-up information was obtained from the contributing pathologists, hospital records, tumor registries and sometimes from the patients themselves. If clinical records were not available, the cause of death was obtained from the state death certificates.
Immunohistochemistry
Immunostains were performed on freshly cut sections on 15 tumors and of variable numbers of available unstained slides on 9 tumors; of the latter group, partial results were reviewed from the files. Also, normal colon segments from surgery for diverticulitis or carcinoma (n = 12) were evaluated immunohistochemically to find the normal distribution of CD117-, actin, desmin and CD34-positive cells. Antibodies to the following antigens were used: CD34 (QBEND-10, monoclonal, diluted at 1:80, Biogenex laboratories, San Ramon, Ca), CD117 (c-kit pro-oncogene product, a polyclonal antibody sc-168 at 1:400, Santa Cruz Biotechnology, Santa Cruz, Ca), ∝-smooth muscle actin (1A4 monoclonal at 1:800 [Sigma Chemicals, St. Louis, Mo], desmin D33, monoclonal at 1:100 [DAKO]), and S100-protein (polyclonal antibody at 1:1600, DAKO). Studies were performed on one representative block of each case by using the avidin biotin peroxidase complex detection system with diaminobenzidine as the chromogen. Microwave epitope retrieval in sodium EDTA-buffer (372 mg/L, pH 8.0) was used before immunostaining for CD34 and CD117. The staining for CD34, CD117, smooth muscle actin and S100-protein were interpreted only when positive normal cells were present (endothelial cells, mast cells or Cajal cells, vascular pericytes, dendritic cells or nerves, respectively). This rule was not followed in the case of desmin, because normal muscle tissue was sometimes absent in the available slides. Three authors evaluated the results, and consensus was reached in each case.
RESULTS
Clinical Features
There were 88 patients with leiomyoma of muscularis mucosae. There was a significant, nearly identical male predominance in both institutions (AFIP M:F 2.6:1 and Helsinki M:F 2.4:1) suggesting that the male predominance of the military population did not skew the gender distribution. The patients' ages ranged 38 to 85 years (median 62 years). Only two patients were under 40 years of age. The age and sex distribution of the 85 cases with complete data is shown in Figure 1.
The leiomyomas were predominantly distally located: 21 in rectum, 36 in sigmoid, 12 in descending colon, two in transverse colon and two in hepatic flexure. The exact location in colon of 15 tumors was not available. The single hospital material from Helsinki had a ratio of colonic to rectal tumors of 12:6, whereas this ratio in AFIP material was 55:15, indicating that more tumors were located in the colon than in the rectum.
The ratio of leiomyomas of muscularis mucosae to GISTs in colon was 12:1 in the Helsinki single hospital material and 55:36 in the AFIP material. The ratio of leiomyomas of muscularis mucosae to GISTs in the rectum was 6:6 in the Helsinki single hospital material and 15:127 in the AFIP material indicating the leiomyomas of muscularis mucosae are more common than GISTs in the colon, but equally or less common than GISTs in the rectum (although referral bias likely caused relative overrepresentation of the GISTs in the AFIP material).
The lesions typically appeared as small sessile polyps, and were removed by endoscopic snare polypectomy. Sometimes they had a pedicle, which occasionally was as long as 7 cm. Endoscopic snare polypectomy resulted in complete tumor removal in virtually all cases. The lesional diameter ranged from 1 to 22 mm (median 4 mm). The three largest tumors measured 22 mm, 14 mm, and 13 mm. There was no evidence of specific symptoms. The colonoscopy indications were not clear from the records in all cases, but most commonly included adenoma surveillance, cancer screening or follow-up of occult blood in stool. One tumor was incidentally detected in a surgical specimen of rectum that was excised because of a mucinous adenocarcinoma.
Pathologic Features
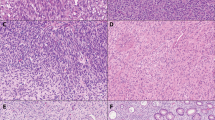
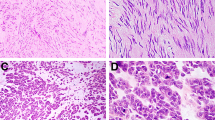
The lesions were grossly described as white, firm, circumscribed nodules that formed the majority of the polyps covered by a grossly intact colonic mucosa. Histologically the lesions were composed of well-differentiated smooth muscle cells that were immediately bordering the overlying epithelium (Fig. 2). The epithelium often showed slight pressure atrophy, with focal ulceration in one case. The lesions completely merged with the muscularis mucosae layer and appeared to originate from it (Fig. 3). In almost all specimens, there was normal submucosa underneath the lesions, but elements of muscularis propria were never present in the endoscopic specimens. Two tumors showed marked atypia with hyperchromatic nuclei (Fig. 4), and one of these lesions also showed a single mitosis.
Immunohistochemically all 24 tumors that were studied were positive for smooth muscle actin and desmin with global, diffuse staining (Fig. 5). All 20 tumors studied were negative for CD34 and CD117 (KIT), but moderate numbers of intralesional mast cells were positive for the latter. S100-protein positive elements included only small nerve twigs adjacent to tumors and scattered dendritic antigen presenting cells.
Normal colonic tissue contained CD117-positive Cajal cell-like elements only in and around the myenteric plexus; such cells were not identified in the muscularis mucosae layer. Both muscularis mucosae and muscularis propria were uniformly positive for smooth muscle actin and desmin. In addition to endothelia, scattered fibroblasts between the smooth muscle fibers were CD34-positive.
Follow-Up
Information available on 29 patients showed no recurrences (median follow-up of 46 months [range 2 to 145 months]). Thirteen patients died of unrelated causes. One of the two patients whose leiomyoma had significant atypia and a mitosis, was alive and well after 5 years. Another patient whose leiomyoma showed significant atypia died of hypertensive heart disease 43 months later.
DISCUSSION
In this study we analyzed a series of 88 leiomyomas of the muscularis mucosae of colon and rectum. These tumors are currently increasingly recognized as incidental findings in colorectal endoscopies performed for cancer screening or polyp surveillance. In some instances they have been referred to as submucosal leiomyomas, but they actually arise in the muscularis mucosae, which is obliterated, and the lesion then is located immediately beneath the surface epithelium. The restriction of the lesions to the muscularis mucosae makes them amenable to complete endoscopic removal, because grasping the lesion with an instrument draws up the uninvolved submucosa.
All mesenchymal tumors arising from or limited to the muscularis mucosae were leiomyomas composed of well-differentiated smooth muscle cells that are uniformly positive for actin and desmin, and negative for CD34 and CD117, similar to normal colonic muscularis mucosae and muscularis propria. These tumors differ from the specific GISTs clinically, histologically, and immunophenotypically.
Whereas the leiomyomas are positive for actin and desmin and negative for CD34 and CD117 (KIT), the GISTs have the opposite patterns. We have seen only four colonic GISTs smaller than 1 cm. All were involving the outer muscular layer and were removed from the serosal side (14). No KIT-positive GISTs limited to the muscularis mucosae layer were identified in this study. The normal muscularis mucosae layer does not contain CD117-positive Cajal cells or multipotential stem cells, which probably explains why GISTs do not occur in the muscularis mucosae layer or present as small intraluminal polyps.
It was difficult to ascertain specific symptoms for these tumors, and they probably mostly were incidental findings. According to our experience based on combination of single hospital material and AFIP referral material, the leiomyomas of the muscularis mucosae are at least twice as common as GISTs in the colon, whereas equal number of these tumors were seen in the rectum in the single hospital material.
Another location where leiomyomas are seen with a high relative frequency is esophagus, where intramural leiomyomas are the most common mesenchymal tumors and outnumber GISTs by 48:17 = 2.8:1) (15). However, intramural leiomyomas are extremely rare in colon and rectum, if they occur. Similarly, prototypic leiomyomas are very rare in the stomach and small intestine, according to our experience.
The leiomyomas of the muscularis mucosae seem to predominantly occur in older people (median age 62 years). Although this may partly reflect the patient population undergoing colonoscopy, also young patients are frequently colonoscoped for the surveillance of inflammatory bowel disease, yet only two patients under 40 years had a leiomyoma of muscularis mucosae.
The leiomyomas of the muscularis mucosae have a significant male predominance 2.5:1, similar to what has been found in esophageal leiomyoma (3:1) (15). Although the genetic mechanism for the development of muscularis mucosae leiomyoma is unknown, deletions in collagen type IV alpha 5 and alpha 6 genes located in the X-chromosome have been reported in both familial and sporadic esophageal leiomyomas (16, 17). In males, any deletions involving the X-chromosome are homozygous thereby potentially explaining the male predominance of diseases with such a genetic involvement.
Significant atypia was a rare finding in leiomyomas of the muscularis mucosae. This appears to be comparable with the atypia seen in other leiomyomas, such as those in the uterus and male genitalia and commonly referred to as “symplastic leiomyomas.” Without mitotic activity, such atypia appears to have little clinical significance, as none of the lesions showed tumor recurrence despite conservative snare polypectomy. However, we believe that it is prudent to ensure that the atypical lesion is limited to mucosa, or, if in doubt, perform an additional endoscopic excision of the polyp base for histologic examination, or at least maintain a close follow-up.
Typical actin and/or desmin-positive leiomyosarcomas are very rare in colon, and they often appear as intraluminal polypoid lesions. Although these tumors could theoretically be of muscularis mucosae origin, such an origin could not be demonstrated in our recent review of true colonic leiomyosarcomas (14). Our data do not support an association between the leiomyomas of the muscularis mucosae and true colonic leiomyosarcomas. However, the possibility that the true colonic leiomyosarcomas originate from the muscularis mucosae cannot be ruled out.
CONCLUSION
In summary, we presented 88 cases of leiomyomas of the muscularis mucosae of colon and rectum. These tumors typically present as incidental intraluminal polyps at endoscopy, and are clinically harmless lesions. They are actin and desmin-positive and are not related to the KIT-positive specific GISTs.
References
Appelman HD . Mesenchymal tumors of the gut: histological perspectives, new approaches, new results, and does it make any difference. Monogr Pathol 1990; 31: 220–246.
Kindblom LG, Remotti HE, Aldenborg F, Meis-Kindblom JM . Gastrointestinal pacemaker cell tumor (GIPACT). Gastrointestinal stromal tumors show phenotypic characteristics of the interstitial cells of Cajal. Am J Pathol 1998; 152: 1259–1269.
Miettinen M, Lasota J . Gastrointestinal stromal tumors—definition, clinical, histologic, immunohistochemical and molecular genetic features and differential diagnosis. A review. Virchows Arch 2001; 438: 1–12.
Sircar K, Hewlett BR, Huizinga JD, Chorneyko K, Berezin I, Riddell RH . Interstitial cells of Cajal as precursors for gastrointestinal stromal tumors. Am J Surg Pathol 1999; 23: 377–389.
Ueyama T, Guo K-J, Hashimoto H, Daimaru Y, Enjoji M . A clinicopathologic and immunohistochemical study of gastrointestinal stromal tumors. Cancer 1992; 69: 947–955.
Haque S, Dean PJ . Stromal neoplasms of the rectum and anal canal. Hum Pathol 1992; 23: 762–767.
Hishida Y, Ishida M . Smooth-muscle tumors of the rectum in Japanese. Dis Colon Rectum 1974; 17: 226–234.
Kusminsky RE, Bailey W . Leiomyomas of the rectum and anal canal: report of six cases and review of the literature. Dis Colon Rectum 1977; 20: 580–599.
Moyana TN, Friesen R, Tan LK . Colorectal smooth-muscle tumors: a pathologic study with immunohistochemistry and histomorphometry. Arch Pathol Lab Med 1991; 115: 1016–1021.
Nemer FD, Stoeckinger JM, Evans OT . Smooth-muscle rectal tumors: a therapeutic dilemma. Dis Colon Rectum 1977; 20: 405–413.
Tworek JA, Goldblum JR, Weiss SW, Greenson JK, Appelman HD . Stromal tumors of the abdominal colon. A clinicopathologic study of 20 cases. Am J Surg Pathol 1999; 23: 937–945.
Tworek JA, Goldblum JR, Weiss SW, Greenson JK, Appelman HD . Stromal tumors of the anorectum. A clinicopathologic study of 22 cases. Am J Surg Pathol 1999; 23: 946–954.
Walsh TH, Mann CV . Smooth muscle neoplasms of the rectum and anal canal. Br J Surg 1987; 71: 597–599.
Miettinen M, Sarlomo-Rikala M, Sobin LH, Lasota J . Gastrointestinal stromal tumors and leiomyosarcomas in the colon—a clinicopathologic, immunohistochemical and molecular genetic study of forty-four cases. Am J Surg Pathol 2000; 24: 1339–1352.
Miettinen M, Sarlomo-Rikala M, Sobin LH, Lasota J . Esophageal stromal tumors—a clinicopathologic, immunohistochemical and molecular genetic study of seventeen cases and comparison with esophageal leiomyomas and leiomyosarcoma. Am J Surg Pathol 2000; 24: 211–222.
Heidet L, Boye E, Cai Y, Sado Y, Zhang X, Flejou JF, et al. Somatic deletion of the 5′ ends of both the COL4A5 and COL4A6 genes in a sporadic leiomyoma of the esophagus. Am J Pathol 1998; 152: 673–678.
Ueki Y, Naito I, Oohashi T, Sugimoto M, Seki T, Yoshioka H, et al. Topoisomerase I, and II consensus sequences in a 17-kb deletion junction of the COL4A5 and COL4A6 genes and immunohisto-chemical analysis of esophageal leiomyomatosis associated with Alport syndrome. Am J Hum Genet 1998; 62: 253–261.
Acknowledgements
The cases contributed of Veterans Affair Hospitals are gratefully acknowledged. Supported by American Registry of Pathology.
Author information
Authors and Affiliations
Additional information
The opinions and assertions contained herein are the expressed views of the authors and are not to be construed as official or reflecting the views of the Departments of the Army or Defense.
Rights and permissions
About this article
Cite this article
Miettinen, M., Sarlomo-Rikala, M. & Sobin, L. Mesenchymal Tumors of Muscularis Mucosae of Colon and Rectum are Benign Leiomyomas that Should be Separated from Gastrointestinal Stromal Tumors—A Clinicopathologic and Immunohistochemical Study of Eighty-Eight Cases. Mod Pathol 14, 950–956 (2001). https://doi.org/10.1038/modpathol.3880417
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/modpathol.3880417
Keywords
This article is cited by
-
Colonic perineurioma (benign fibroblastic polyp): case report and review of the literature
Diagnostic Pathology (2018)
-
Recurrent fusion of the genes FN1 and ALK in gastrointestinal leiomyomas
Modern Pathology (2016)
-
Primary leiomyosarcoma of the colon
Clinical Journal of Gastroenterology (2015)