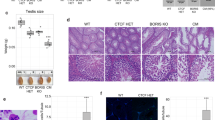
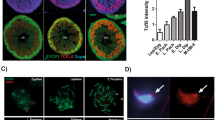
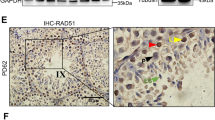
Abstract
SPERMATOGENESIS is a complex developmental process that occurs in several phases. A large number of genes have been identified that are expressed during spermatogenesis1,2, but the biological significance of many of these is not yet known. We have used gene targeting to selectively eliminate the transcription factor CREM (cyclic AMP-responsive element modulator), which is thought to e important for mammalian spermatogenesis3–5. Male mice deficient for all CREM proteins are sterile, as their developing spermatids fail to differentiate into sperm, and postmeiotic gene expression in the testis declines dramatically. The cessation of sperm development is not accompanied by decreases in the levels of follicle-stimulating hormone or testosterone. Our findings indicate that the CREM gene is essential for spermatogenesis, and mice deficient for this transcription factor could serve as a model system for the study of idiopathic infertility in men.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 51 print issues and online access
$199.00 per year
only $3.90 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout
Similar content being viewed by others
References
Wolgemuth, D. J. & Watrin, F. Mamm. Genome 1, 283–288 (1991).
Erikson, R. P. Trends Genet. 6, 264–269 (1990).
Foulkes, N. S., Mellström, B., Benusiglio, E. & Sassone-Corsi, P. Nature 355, 80–84 (1992).
Foulkes, N. S., Borreli, E. & Sassone-Corsi, P. Cell 64, 739–749 (1991).
Foulkes, N. S., Schlotter, F., Pevet, P. & Sassone-Corsi, P. Nature 362, 264–267 (1993).
Hurst, H. Protein Profile 2, 105–168 (1995).
Weinbauer, G. F. & Nieschlag. E. in Molecular Biology of Reproduction (ed. de Kretser, D. M.) 99–142 (Academic, New York, 1993).
Nantel, F. et al. Nature 380, 159–162 (1996).
Kashiwabara, S., Arai, Y., Kodaira, K. & Bab, T. Biochem. biophys. Res. Commun. 173, 240–245 (1990).
Anakwe, O. O., Sharma, S., Hardy, D. M. & Gerton, G. L. Molec. Reprod. Dev. 29, 172–179 (1991).
Escalier, D. et al. Development 133, 779–788 (1991).
Bellve, A. R. in Oxford Review of Reproductive Biology Vol. 1 (ed. Finne, C. A.) 159–260 (Oxford Univ. Press, 1979).
Kleene, K. C. Development 106, 367–373 (1989).
Heidaran, M. A., Showman, R. M. & Kistler, W. S. J. biol. Chem. 106, 1427–1433 (1988).
Kleene, K. C. et al. Devl Biol. 137, 395–402 (1990).
Heidaran, M. A. & Kistler, W. S. Gene 54, 281–284 (1987).
Zambrowicz, B. P., Harendza, C. J., Zimmermann, J. W., Brinster, R. L. & Palmiter, R. D. Proc. natn. Acad. Sci. U.S.A. 90, 5071–5075 (1993).
Kistler, M. K., Sassone-Corsi, P. & Kistler, W. S. Biol. Reprod. 51, 1322–1329 (1994).
Shin, D. M. & Kleene, K. C. Molec. Reprod. Dev. 33, 222–227 (1992).
Van der Hoorn, F. A. & Tarnasky, H. A. Proc. natn. Acad. Sci. U.S.A. 89, 703–707 (1992).
Delmas, V., van der Hoorn, F., Mellström, B., Jegou, B. & Sassone-Corsi, P. Molec. Endocr. 7, 1502–1514 (1993).
Nieschlag, E. Clin. Endocr. 38, 123–133 (1993).
Chandolia, R. K., Weinbauer, G. F., Simoni, S., Behre, H. M. & Nieschlag, E. Acta endocr. 125, 547–555 (1991).
Tamura, T. et al. Nucleic Acids Res. 19, 3861–3865 (1991).
Kaestner, K. H., Montoliu, L., Kern, H., Thulke, M. & Schütz, G. Gene 148, 67–70 (1994).
Russell, L. D., Ettlin, R. A., Sinha Hikim, A. P. & Clegg, E. D. in Histological and histopathological evaluation of the testis 119–161 (Cache River, Clearwater, FL, 1990).
Klemm, U. et al. Differentiation 42, 160–168 (1990).
Adham, I. M. et al. DNA Cell Biol. (in the press).
Kleen, K. C., Distel, R. J. & Hecht, N. B. Biochemistry 24, 719–722 (1985).
Kleen, K. C., Borzorgzadeh, A., Flynn, J. F., Yelick, P. C. & Hecht, N. B. Biochim. biophys. Acta 950, 215–220 (1988).
Bibb, M. J., VanEtten, R. A., Wright, C. T., Walberg, M. W. & Clayton, D. A. Cell 26, 167–180 (1981).
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Blendy, J., Kaestner, K., Weinbauer, G. et al. Severe impairment of permatogenesis in mice lacking the CREM gene. Nature 380, 162–165 (1996). https://doi.org/10.1038/380162a0
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1038/380162a0
This article is cited by
-
CaMK4 controls follicular helper T cell expansion and function during normal and autoimmune T-dependent B cell responses
Nature Communications (2024)
-
The deacetylase SIRT2 contributes to autoimmune disease pathogenesis by modulating IL-17A and IL-2 transcription
Cellular & Molecular Immunology (2022)
-
Genome-wide occupancy reveals the localization of H1T2 (H1fnt) to repeat regions and a subset of transcriptionally active chromatin domains in rat spermatids
Epigenetics & Chromatin (2021)
-
Retinoid-related orphan nuclear receptor alpha (RORα)-deficient mice display morphological testicular defects
Laboratory Investigation (2019)
-
The transcription factor CREM drives an inflammatory phenotype of T cells in oligoarticular juvenile idiopathic arthritis
Pediatric Rheumatology (2018)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.