Abstract
Placental site trophoblastic tumor is a neoplasm of extravillous intermediate trophoblast at the implantation site, preceded in the majority of cases by a female gestational event. Our pilot investigation suggested that the development of this tumor might require a paternally derived X chromosome and the absence of a Y chromosome. Twenty cases of placental site trophoblastic tumor were included in this study. Genotyping at 15 polymorphic loci and one sex determination locus was performed by multiplex PCR followed by capillary electrophoresis. X chromosome polymorphisms were determined by PCR amplification of exon 1 of the human androgen receptor gene using primers flanking the polymorphic CAG repeats within this region. Genotyping at 15 polymorphic loci was informative and paternal alleles were present in all tumors, confirming the trophoblastic origin of the tumors. The presence of an X chromosome and the absence of a Y chromosome were observed in all tumors. Among 13 cases in which analysis of the X chromosome polymorphism was informative, all but one demonstrated at least two X alleles and seven cases showed one identifiable paternal X allele. These results confirm a unique pathogenetic mechanism in placental site trophoblastic tumor, involving an exclusion of the Y chromosome from the genome and, therefore, a tumor arising from the trophectoderm of a female conceptus. As epigenetic regulations of imprinting during X chromosome inactivation are of significant biological implications, placental site trophoblastic tumor may provide an important model for studying the sex chromosome biology and the proliferative advantage conferred by the paternal X chromosome.
Similar content being viewed by others
Main
Placental site trophoblastic tumor is a rare neoplastic proliferation of extravillous intermediate trophoblasts at the implantation site.1 It belongs to an extraordinary group of neoplasms that are fetal in origin and associated with infiltration of the maternal endometrium and mytometrium. The clinical presentation is generally that of a young woman who has a history of full term pregnancy, an abortion or uncommonly, a complete mole. The majority of the tumors pursue a benign clinical course; however, clinically malignant cases exist with an estimated mortality rate of 15%.2 Treatment of placental site trophoblastic tumor is mainly surgical, but successful multidrug chemotherapy has been recently reported in some aggressive cases.3
At cytogenetic level, most of the tumors analyzed have been diploid4, 5 and rarely showed genetic imbalances analyzed by comparative genomic hybridization.6, 7 We have previously noted through literature reviews that a majority of the tumors were preceded by a female gestational event and our pilot molecular investigation suggested that the tumorigenesis likely requires the presence of a paternally derived X chromosome in the tumor genome.8 We now present an analysis of a larger series of cases to confirm these earlier observations.
Materials and methods
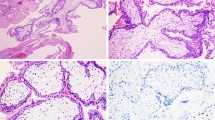
The current study consisted of 20 archival cases of placental site trophoblastic tumor, obtained from eight medical centers in the United States. All cases were diagnosed by the primary institutions based on histology and immunohistochemistry. Representative hematoxylin–eosin-stained slides of each tumor were reviewed and the diagnosis was confirmed according to well-established histological criteria.1 Histological review of sections from all tumors showed sheets and cords of large polygonal cells having moderate to abundant pink to clear cytoplasm and large nuclei, and permeating surrounding myometrial smooth muscle (Figure 1).
Histological features of placental site trophoblastic tumor (hematoxylin–eosin stain). The tumor consists of sheets of neoplastic trophoblasts characterized by large polygonal cells with abundant cytoplasm and pleomorphic nuclei, invading the adjacent myometrium. Note the intimate association of tumor cells with myometrial smooth muscle cells, contributing to the contaminating PCR products seen in Figures 2 and 3.
DNA was extracted from unstained tumor and paired normal tissue sections from the same case. Briefly, 10 serial 10 μm sections were cut with the first one stained with hematoxylin–eosin to verify the presence of tumor tissue, and the remaining nine used for DNA extraction using Qiagen DNA tissue kits (Qiagen, Chatsworth, CA, USA) according to the manufacturer's instruction. The concentration of DNA preparation was determined by absorbance at 260 nm. Tumor genotyping was analyzed by AmpFlSTR® Identifiler™ PCR amplification system (Applied Biosystems, Inc.). The reaction consists of a multiplex assay that amplifies 15 tetranucleotide tandem repeat loci in a single PCR amplification, with amplicons ranging from 100 to 350 base pairs. All 13 loci for the combined DNA index system (CODIS) loci are included with two additional loci, D2S1338 and D19S433. The combination of these loci is consistent with several worldwide database recommendations for reliable determination of identity.9 In addition, the presence of X and Y chromosome was determined by amplification using the primers complementary to DNA sequence of the amelogenin locus shared by both X and Y chromosomes and flanking a six-base pair deletion within intron 1 of this gene.10 Amplification of this region results in 107-and 113-bp products from the X and Y chromosomes, respectively. Genomic DNA of 0.5–1.25 ng was amplified in a 25-μl reaction mix containing 10.0 μl of AmpFlSTR reaction mix, 5.0 μl of AmpFlSTR primer mix and 0.5 μl AmpliTaq Gold DNA polymerase. The PCR was as carried out as follows: 11 min at 95°C, followed by 28 cycles of 94°C for 1 min; 59°C for 1 min and 72°C for 1 min, completed at 60°C for 60 min. A 1 μl volume of the PCR product was mixed with 13 μl and 0.5 μl sizing marker (GeneScan-500LIZ; Applied Biosystems) and capillary electrophoresis was performed using an ABI3130 instrument. Data collection and analysis were performed using GeneMapper™ software version 3.7 (Applied Biosystems).
X chromosome polymorphism analysis was performed by PCR amplification of exon 1 of the human androgen receptor gene using primers flanking the polymorphic CAG repeats. Genomic DNA (200 ng) was subject to a 25 μl PCR containing 1 × PCR buffer (Roche), 25 mM MgCl2, 5 mM dNTP, 0.4 μ M forward primer (Hex-5′-CCTAGGAGCTTTCCAGAATC-3′) and reverse primer (5′-TACGATGGGCTTGGGGAGAA-3′). The PCR was carried out for 8 min 95°C, 30 cycles of 95°C for 40 s, 57°C for 30 s and 72°C for 40 s, with a final extension at 72°C for 45 min. A 1 μl volume of the PCR product was analyzed by the capillary electrophoresis as described above.
Results
Although AmpFlSTR® Identifiler™ PCR failed to produce some larger allelic products in some cases due probably to partial DNA degradation, genotyping was overall informative in all 20 tumors (Table 1). Unique paternal alleles were found in all tumors, confirming the trophoblastic origin of the tumors. Amplification at the amelogenin loci was successful in every case (Table 1). The presence of X chromosome and the absence of Y chromosome were consistently observed (Figure 2).
AmpFlSTR® Identifiler™ PCR amplifies the amelogenin, D5S818 and FGA (α-fibrinogen) tetranucleotide polymorphic loci in placental site trophoblastic tumor (upper panel), paired normal myometrium (middle panel) and unrelated, normal male control tissue (lower panel). The PCR products were analyzed by capillary electrophoresis (y-axis—fluorescence intensity of labeled product and x-axis—PCR product size in base pairs). The tumor demonstrates unique paternal alleles at D5S818 and FAG loci together with the maternal alleles detected in the paired myometrium. One of the maternal alleles at both D5S818 and FAG loci (upper panel) represents the maternal smooth muscle cells within the tumor. Normal male control sample shows both X and Y products (107 and 113 bp, respectively) at the amelogenin locus, while only the X product is seen in the tumor and paired myometrium.
Analyses of polymorphisms in the X-linked human androgen receptor gene were unsuccessful in seven cases, largely due to the degradation of the template DNA and the relatively larger amplicons of the PCR (250–320 bp). Since the tumor cells were infiltrative and intimately associated with normal maternal myometrial tissue (Figure 1), the presence of contaminating maternal genome was anticipated. Nevertheless, among 13 informative cases, all but one demonstrated at least two X chromosomes in the tumor samples and seven cases showed an identifiable paternal X allele (Figure 3). The remaining six cases harbored two X alleles that were indistinguishable from those of the corresponding maternal tissue.
Representative X chromosome polymorphism analysis by PCR amplification of exon 1 of the human androgen receptor gene in placental site trophoblastic tumor (upper panel) and paired normal myometrium (lower panel). In addition to the two maternal X alleles (two peaks, lower panel), the tumor demonstrates a unique paternal X allele (middle peak, upper panel). One of the maternal peaks (upper panel) represents PCR amplification of DNA from the maternal smooth muscle cells within the tumor.
Discussion
During human gestation, the embryo implants into the specialized endometrium (decidua) and the placental forms as trophoblasts invade the endometrium to establish the maternal–fetal interface. Placental site trophoblastic tumor arises from neoplastic transformation of trophoblasts that invade the endometrium and the myometrium. At the genetic level, most placental site trophoblastic tumors are diploid and all are allogeneic, with one haploid set of chromosomes different from that of the maternal tissue. Thus, placental site trophoblastic tumor represents an extraordinary process, in which normal immunologic barriers to the overgrowth of allogeneic tissues are overcome. In this respect, placental site trophoblastic tumor is similar to two other forms of proliferative gestational diseases, that is, partial and complete hydatidiform moles, in which distinctive chromosomal abnormalities have long been recognized.11, 12 All complete moles contain a diploid or, less frequently, tetraploid genome consisting of two or four sets of paternal chromosomes, including a paternally derived X chromosome and over 95% of partial moles contain a diandric triploid genome consisting of two sets of paternal and one maternal genome. As demonstrated in the current study, placental site trophoblastic tumor is another trophoblastic neoplasm with a distinct genetic composition.
In our pilot investigation, the presence of a paternal X chromosome was seen in 4 of 5 cases, and the absence of a Y chromosomal element was found in all five tumors by the semi-nested PCR amplification of SRY (human sex-determining region on Y chromosome).8 Since then, additional case reports of placental site trophoblastic tumor have been published and again a female antecedent pregnancy was documented in 12 out of 14 new cases.13, 14, 15 One cell line from a placental site trophoblastic tumor was established and confirmed by karyotyping to have XX genome.16 However, the results of both our current and previous investigations contrast to those of a recent genetic analysis of 23 placental site trophoblastic tumors, in which 50% of the tumors were found to contain a Y chromosome.17 The reasons for this discrepancy are not clear but could be due to different methodologies chosen. In the paper published by Oldt et al,17 using DNA materials extracted from paraffin-embedded formalin-fixed tissues, the detection of SRY was carried out by a single, 50-cycle PCR amplification followed by gel electrophoresis. Such an approach carries the risk of technical artifacts due to high cycle number and potential amplification of contaminating template. Alternative methods (such as X chromosome polymorphism or the amelogenin sex homologue analysis, see below) were not performed to confirm their findings. AmpFlSTR® Identifiler™ PCR amplification assay is a short tandem repeat multiplex assay that amplifies in a single PCR amplification 15 tetranucleotide repeat loci as well as the amelogenin locus shared by the X and Y chromosomes. The small products generated from the X and Y amelogenin alleles permit efficient analysis of DNA template extracted from formalin-fixed paraffin-embedded tissue. In conjunction with capillary electrophoresis, which precisely sizes the two X and Y PCR products, this approach ensures a highly consistent and reliable assessment of sex chromosome content.
The presence of a haploid set of chromosomes without a Y chromosomal element in all of our 20 cases supports the concept that placental site trophoblastic tumor arises from the trophectoderm of a female conceptus. These results imply that the presence of paternal X chromosome is important for the neoplastic proliferation of placental site trophoblastic tumor. One unlikely explanation is that placental site trophoblastic tumor might derive from an antecedent complete hydatidiform mole, a comparatively more common gestational trophoblastic disease with uniparental disomy of the paternal genome and an exclusive paternal X chromosome. However, only a small percentage of placental site trophoblastic tumors have been reported to follow the development of complete mole1, 2. Alternatively, our data suggest an active role for the paternal X chromosome in the pathogenesis of placental site trophoblastic tumor, although the mechanistic contribution of this chromosome to tumor growth is unclear.
Epigenetic imprinting, the selective suppression of various genes derived from one parent or the other, has been proposed to be an important regulatory pathway in the development of the placenta in eutherian mammals. The ‘parental conflict hypothesis’ views that parents of opposite sex have conflicting interests in allocating resources to their offspring by the mother.18, 19, 20, 21 The intent of the paternal genome would be to maximize resources for the father's own progeny. In contrast, the interest of the maternal genome would be to distribute resources equally among the offspring. This implies that growth-promoting genes are mainly expressed from the paternally inherited genome and are silent in the maternally inherited counterpart. Analyses of many imprinted genes in mammals support this theory.22, 23
X chromosome inactivation plays a central role in compensating for the double dose of X-linked genes in cells of the female relative to cells of the male. In some species, X inactivation represents an example of epigenetic imprinting. The paternal X chromosome is preferentially imprinted and silent in mouse trophectoderm,24, 25, 26, 27, 28 a tissue type corresponding to that from which the placental site trophoblastic tumor and hydatidiform moles arise in humans. In marsupial mammals, the paternal X chromosome is preferentially inactivated in all lineages.29, 30 Unexpectedly, the mode of X inactivation appears random in human extra-embryonic tissue by recent data,31, 32 although this remains controversial.28, 33, 34 Nevertheless, the chromosomal composition of placental site trophoblastic tumor suggests a functional role of the paternal X chromosome in this tumor, for instance, by inappropriate expression of paternal X-linked genes. Further investigation is required to ascertain whether the persistence of a paternal X chromosome in trophectoderm provides a proliferative advantage, eventually leading to neoplastic transformation.
References
Shih IM, Kurman RJ . The pathology of intermediate trophoblastic tumors and tumor-like lesions. Int J Gynecol Pathol 2001;20:31–47.
Baergen RN, Rutgers JL, Young RH, et al. Placental site trophoblastic tumor: a study of 55 cases and review of the literature emphasizing factors of prognostic significance. Gynecol Oncol 2006;100:511–520.
Ajithkumar TV, Abraham EK, Rejnishkumar R, et al. Placental site trophoblastic tumor. Obstet Gynecol Surv 2003;58:484–488.
Kotylo PK, Michael H, Davis TE, et al. Flow cytometric DNA analysis of placental-site trophoblastic tumors. Int J Gynecol Pathol 1992;11:245–252.
Fukunaga M, Ushigome S . Malignant trophoblastic tumor: immunohistochemical and flow cytometric comparison of choriocarcinoma and placental site trophoblastic tumors. Hum Pathol 1993;24:1098–1106.
Hui P, Riba A, Pejovic T, et al. Comparative genomic hybridization study of placental site trophoblastic tumour: a report of four cases. Mod Pathol 2004;17:248–251.
Xue WC, Guan XY, Ngan HY, et al. Malignant placental site trophoblastic tumor: a cytogenetic study using comparative genomic hybridization and chromosome in situ hybridization. Cancer 2002;94:2288–2294.
Hui P, Parkash V, Perkins AS, et al. Pathogenesis of placental site trophoblastic tumor may require the presence of a paternally derived X chromosome. Lab Invest 2000;80:965–972.
Collins PJ, Hennessy LK, Leibelt C, et al. Developmental validation of a single-tube amplification of the 13 CODIS STR loci, D2S1338, D19S433, and amelogenin: the AmpFlSTR Identifiler PCR Amplification Kit. J Forensic Sci 2004;49:1265–1277.
Akane A . Sex determination by PCR analysis of the X–Y amelogenin gene. Methods Mol Biol 1998;98:245–249.
Szulman A . Syndromes of hydatidiform moles, partial vs complete. J Reprod Med 1984;29:788–791.
Kajii T, Ohama K . Androgenetic origin of hydatidiform mole. Nature 1977;268:633–634.
Hassadia A, Gillespie A, Tidy J, et al. Placental site trophoblastic tumour: Clinical features and management. Gynecol Oncol 2005;99:603–607.
Khan S, Dancey G, Lindsay I, et al. Placental site trophoblastic tumour derived from an oocyte donation pregnancy. BJOG 2006;113:344–346.
How J, Scurry J, Grant P, et al. Placental site trophoblastic tumor. Report of three cases and review of the literature. Int J Gynecol Cancer 1995;5:241–249.
Kobel M, Pohl G, Schmitt WD, et al. Activation of mitogen-activated protein kinase is required for migration and invasion of placental site trophoblastic tumor. Am J Pathol 2005;167:879–885.
Oldt RJ, Kurman R, Shih IM . Molecular genetic analysis of placental site trophoblastic tumors and epithelioid trophoblastic tumors confirms their trophoblastic origin. Am J Pathol 2002;161:1033–1037.
Barlow D . Gametic imprinting in mammals. Science 1995;270:1610–1613.
Haig D, Westoby M . An earlier formulation of the genetic conflict hypothesis of genomic imprinting. Nat Genet 2006;38:271.
Abu-Amero S, Monk D, Apostolidou S, et al. Imprinted genes and their role in human fetal growth. Cytogenet Genome Res 2006;113:262–270.
Solter D . Imprinting. Int J Dev Biol 1998;42:951–954.
Tilghman SM . The sins of the fathers and mothers: genomic imprinting in mammalian development. Cell 1999;96:185–193.
Constancia M, Kelsey G, Reik W . Resourceful imprinting. Nature 2004;432:53–57.
Li Y, Lemaire P, Behringer RR . Esx1, a novel X chromosome-linked homeobox gene expressed in mouse extraembryonic tissues and male germ cells. Dev Biol 1997;188:85–95.
Lin TP, Labosky PA, Grabel LB, et al. The Pem homeobox gene is X-linked and exclusively expressed in extraembryonic tissues during early murine development. Dev Biol 1994;166:170–179.
Salido EC, Yen PH, Mohandas TK, et al. Expression of the X-inactivation-associated gene XIST during spermatogenesis. Nat Genet 1992;2:196–199.
Sado T, Ferguson-Smith AC . Imprinted X inactivation and reprogramming in the preimplantation mouse embryo. Hum Mol Genet 2005;14:R59–R64.
Huynh KD, Lee JT . Inheritance of a pre-inactivated paternal X chromosome in early mouse embryos. Nature 2003;426:857–862.
Cooper DW, VandeBerg JL, Sharman GB, et al. Phosphoglycerate kinase polymorphism in kangaroos provides further evidence for paternal X inactivation. Nat New Biol 1971;230:155–157.
Graves JA . Mammals that break the rules: genetics of marsupials and monotremes. Annu Rev Genet 1996;30:233–236.
Looijenga L, Gillis AJM, Verkerk AJ, et al. Heterogeneous X inactivation in trophoblastic cells of human full-term female placentas. Am J Hum Genet 1999;64:1445–1452.
Migeon BR, Lee CH, Chowdhury AK, et al. Species differences in TSIX/Tsix reveal the roles of these genes in X-chromosome inactivation. Am J Hum Genet 2002;71:286–293.
Goto T, Wright E, Monk M . Paternal X-chromosome inactivation in human trophoblastic cells. Mol Hum Reprod 1997;3:77–80.
Lee JT . Molecular links between X-inactivation and autosomal imprinting: X-inactivation as a driving force for the evolution of imprinting. Curr Biol 2003;13:R242–R254.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Hui, P., Wang, H., Chu, P. et al. Absence of Y chromosome in human placental site trophoblastic tumor. Mod Pathol 20, 1055–1060 (2007). https://doi.org/10.1038/modpathol.3800941
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/modpathol.3800941
Keywords
This article is cited by
-
Gestational trophoblastic disease: an update
Abdominal Radiology (2023)
-
Historical, morphological and clinical overview of placental site trophoblastic tumors: from bench to bedside
Archives of Gynecology and Obstetrics (2017)