Abstract
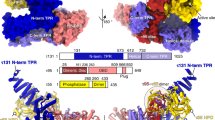
The crystal structure of the transcription factorIIB (TFIIB)/TATA box-binding protein (TBP)/ TATA-element ternary complex is described at 2.7 Å resolution. Core TFIIB resembles cyclin A, and recognizes the preformed TBP–DNA complex through protein–protein and protein–DNA interactions. The amino-terminal domain of core TFIIB forms the downstream surface of the ternary complex, where it could fix the transcription start site. The remaining surfaces of TBP and the TFIIB can interact with TBP-associated factors, other class II initiation factors, and transcriptional activators and coactivators.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 51 print issues and online access
$199.00 per year
only $3.90 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout
Similar content being viewed by others
References
Roeder, R. G. Trends biochem. Sci. 16, 402–408 (1991).
Hori, R. & Carey, M. Curr. Opin. Genet. Dev. 4, 236–244 (1994).
Moldonado, E. & Reinberg, D. Curr. Opin. Cell Biol. 7, 352–361 (1995).
Zawel, L. & Reinberg, D. Prog. Nucleic Acid Res. molec. Biol. 44, 67–108 (1993).
Nikolov, D. B. & Burley, S. K. Nature struct. Biol. 1, 621–637 (1994).
Parvin, J. & Sharp, P. Cell 73, 533–540 (1993).
Ha, I. et al. Genes Dev. 7, 1021–1032 (1993).
Goodrich, J. & Tjian, R. Curr. Opin. Cell Biol. 6, 403–409 (1994).
Zawel, L., Kumar, K. & Reinberg, D. Genes Dev. 9, 1479–1490 (1995).
Hoopes, B., LeBlanc, J. & Hawley, D. J. biol. Chem. 267, 11539–11546 (1992).
Kim, J. L. & Burley, S. K. Nature struct. Biol. 1, 638–653 (1994).
Koleske, A. & Young, R. Trends biochem. Sci. 20, 113–116 (1995).
Qureshi, S., Khoo, B., Baumann, P. & Jackson, S. P. Proc. natn. Acad. Sci. U.S.A. 92, 6077–6081 (1995).
Hisatake, K., Roeder, R. & Horikoshi, M. Nature 363, 744–747 (1993).
Barberis, A., Muller, C., Harrison, S. & Ptashne, M. Proc. natn. Acad. Sci. U.S.A. 90, 5628–5632 (1993).
Buratowski, S. & Zhou, H. Proc. natn. Acad. Sci. U.S.A. 90, 5633–5637 (1993).
Yamashita, S. et al. Science 261, 463–466 (1993).
Malik, S., Lee, D. & Roeder, R. G. Molec. cell. Biol. 13, 6253–6259 (1993).
Kim, T. et al. Nature 369, 252–255 (1994).
Nikolov, D. B. et al. Nature 360, 40–46 (1992).
Kim, J. L., Nikolov, D. B. & Burley, S. K. Nature 365, 520–527 (1993).
Brünger, A. T. X-PLOR v. 3.1 Manual (Yale Univ., New Haven, CT, 1992).
Laskowski, R. J., Macarthur, M. W., Moss, D. S. & Thornton, J. M. J. appl. Crystallogr. 26, 283–290 (1993).
Lüthy, R., Bowie, J. U. & Eisenberg, D. Nature 356, 83–85 (1992).
Kassavetis, G. et al. Cell 71, 1055–1064 (1992).
Buratowski, S. & Zhou, H. Cell 71, 221–230 (1992).
Colbert, T. & Hahn, S. Genes Dev. 6, 1940–1949 (1992).
Wang, Z. & Roeder, R. G. Proc. natn. Acad. Sci. U.S.A. 92, 7026–7030 (1995).
Gibson, T., Thompson, J., Blocker, A. & Kouzarides, T. Nucleic Acids Res. 22, 946–952 (1994).
Jeffrey, P. et al. Nature 376, 313–320 (1995).
Janin, J. Proteins 21, 30–39 (1995).
Kim, Y., Geiger, J. H., Hahn, S. & Sigler, P. B. Nature 365, 512–520 (1993).
Imbalzano, A., Zaret, K. & Kingston, R. J. biol. Chem. 269, 8280–8286 (1994).
Coulombe, B., Li, J. & Greenblatt, J. J. biol. Chem. 269, 19962–19967 (1994).
Buratowski, S., Hahn, S., Guarente, L. & Sharp, P. A. Cell 56, 549–561 (1989).
Lee, D., DeJong, J., Hashimoto, S., Horikoshi, M. & Roeder, R. Molec. cell. Biol. 12, 5189–5196 (1992).
Hori, R., Pyo, S. & Carey, M. Proc. natn. Acad. Sci. U.S.A. 92, 6047–6061 (1995).
Pinto, I., Wu, W.-H., Na, J. & Hampsey, M. J. biol. Chem. 269, 30569–30573 (1994).
Li, Y., Flanagan, P., Tschochner, H. & Kornberg, R. Science 263, 805–807 (1994).
Berroteran, R., Ware, D. & Hampsey, M. Molec. cell. Biol. 14, 226–237 (1994).
Choy, B. & Green, M. Nature 366, 531–536 (1993).
Roberts, S., Ha, I., Maldonado, E., Reinberg, D. & Green, M. Nature 363, 741–744 (1993).
Goodrich, J., Hoey, T., Thut, C., Admon, A. & Tjian, R. Cell 75, 519–530 (1993).
Klemm, R., Goodrich, J., Zhou, S. & Tjian, R. Proc. natn. Acad. Sci. U.S.A. 92, 5788–5792 (1995).
Feaver, W., Svejstrup, J., Henry, N. & Kornberg, R. Cell 79, 1103–1109 (1994).
Kraluis, P. J. J. appl. Crystallogr. 24, 946–950 (1991).
Lavery, R. & Sklenar, H. J. biomolec. Struct. Dyn. 6, 655–667 (1989).
Nicholls, A., Sharp, K. & Honig, B. Proteins 11, 281–296 (1991).
Studier, F. W., Rosenberg, A. H., Dunn, J. J. & Dubendorff, J. W. Meth. Enzym. 185, 60–89 (1990).
Terwilliger, T. C. & Eisenberg, D. Acta Crystallogr. A39, 813–817 (1983).
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Nikolov, D., Chen, H., Halay, E. et al. Crystal structure of a TFIIB–TBP–TATA-element ternary complex. Nature 377, 119–128 (1995). https://doi.org/10.1038/377119a0
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1038/377119a0
This article is cited by
-
Beyond the canonical role of TFIIB in eukaryotic transcription
Current Genetics (2022)
-
The bacterial multidrug resistance regulator BmrR distorts promoter DNA to activate transcription
Nature Communications (2020)
-
Molecular determinants underlying functional innovations of TBP and their impact on transcription initiation
Nature Communications (2020)
-
Mechanism of head-to-head MCM double-hexamer formation revealed by cryo-EM
Nature (2019)
-
CanTF, a Novel Transcription Factor in Pepper, Is Involved in Resistance to Phytophthora capsici as well as Abiotic Stresses
Plant Molecular Biology Reporter (2018)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.