Abstract
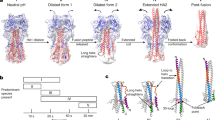
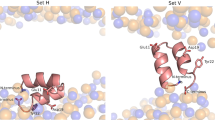
Low pH induces a conformational change in the influenza virus haemagglutinin, which then mediates fusion of the viral and host cell membranes. The three-dimensional structure of a fragment of the haemagglutinin in this conformation reveals a major refolding of the secondary and tertiary structure of the molecule. The apolar fusion peptide moves at least 100 Å to one tip of the molecule. At the other end a helical segment unfolds, a subdomain relocates reversing the chain direction, and part of the structure becomes disordered.
Similar content being viewed by others
Article PDF
References
Wiley, D. C. & Skehel, J. J. A. Rev. Biochem. 56, 365–394 (1987).
Maeda, T. & Ohnishi, S. FEBS Lett. 122, 283–287 (1980).
Huang, R. T. C., Rott, R. & Klenk, H.-D. Virology 110, 243–247 (1981).
White, J., Matlin, K. & Helenius, A. J. Cell Biol. 89, 674–679 (1982).
White, J. M. & Wilson, I. A. J. Cell Biol. 105, 2887–2896 (1987).
Godley, L. et al. Cell 68, 635–645 (1992).
Brand, C. M. & Skehel, J. J. Nature new Biol. 238, 145–147 (1972).
Skehel, J. J. & Waterfield, M. D. Proc. natn. Acad. Sci. U.S.A. 72, 93–97 (1975).
Wilson, I. A., Skehel, J. J. & Wiley, D. C. Nature 289, 366–373 (1981).
Weis, W. I., Brünger, A. T., Skehel, J. J. & Wiley, D. C. J. molec. Biol. 212, 737–761 (1990).
Skehel, J. J. et al. Proc. natn. Acad. Sci. U.S.A. 79, 968–972 (1982).
Gething, M.-J., Doms, R. W., York, D. & White, J. J. Cell Biol. 102, 11–23 (1986).
Daniels, R. S. et al. in The Origin of Pandemic Influenza Viruses (eds Laver, G. W. & Chu, C. M.) 1–7 (Elsevier-North Holland, New York, 1985).
Doms, R. W., Helenius, A. & White, J. J. biol. Chem. 260, 2973–2981 (1985).
Ruigrok, R. W. H. et al. J. gen. Virol. 69, 2785–2795 (1988).
Bullough, P. A. et al. J. molec. Biol. 236, 1262–1265 (1994).
Brünger, A. T. Nature 355, 472–475 (1992).
Wharton, S. A. et al. J. biol. Chem. 263, 4474–4480 (1988).
Carr, C. M. & Kim, P. S. Cell 73, 823–832 (1993).
Ward, C. W. & Dopheide, T. A. Aust. J. biol. Sci. 33, 441–447 (1980).
Graves, P. N., Schulman, J. L., Young, J. F. & Palese, P. Virology 126, 106–116 (1983).
Webster, R. G., Brown, L. E. & Jackson, D. C. Virology 126, 587–599 (1983).
Daniels, R. S. et al. in The Origin of Pandemic Influenza Viruses (eds Laver, W. G. & Chu, C. M.) 8–18 (Elsevier-North Holland, New York, 1985).
Ruigrok, R. W. H. et al. EMBO J. 5, 41–49 (1986).
Daniels, R. S. et al. EMBO J. 6, 1459–1465 (1987).
Yewdell, J. W., Gerhard, W. & Bachi, T. J. Virol. 48, 239–248 (1983).
Ruigrok, R. W. H. et al. Virology 155, 484–497 (1986).
Goldsmith, E. J. & Mottonen, J. Structure 2, 241–244 (1994).
Crick, F. H. C. Acta crystallogr. 6, 689–697 (1953).
McLachlan, A. D. & Karn, J. J. molec. Biol. 164, 605–626 (1983).
Daniels, R. S. et al. Cell 40, 431–439 (1985).
Sauter, N. K. et al. Biochemistry 31, 9609–9621 (1992).
Stegmann, T. & Helenius, A. in Viral Fusion Mechanisms (ed. Bentz, J.) 89–111 (CRC Press, Boca Raton, Florida, 1993).
Söllner, T., Bennett, M. K., Whiteheart, S. W., Scheller, R. H. & Rothman, J. E. Cell 75, 409–418 (1993).
Ellens, H., Bentz, J., Mason, D., Zhang, F. & White, J. M. Biochemistry 29, 9697–9707 (1990).
Spruce, A. E., Iwata, A., White, J. M. & Almers, W. Nature 342, 555–558 (1989).
Kemble, G. W., Danieli, T. & White, J. M. Cell 76, 383–391 (1994).
Blum, M., Metcalf, P., Harrison, S. C. & Wiley, D. C. J. appl. Crystallogr. 20, 235–242 (1987).
Leslie, A. G. W. in CCP4 and ESF-EACMB Newsletter on Protein Crystallography, No. 26 (Daresbury Laboratory, Warrington, 1992).
CCP4, A Suite of Programs for Protein Crystallography (SERC (UK) Collaborative Computing Project No. 4, Daresbury Laboratory, Warrington, 1979).
Terwilliger, T. C. & Eisenberg, D. Acta crystallogr. A39, 813–817 (1983).
Metcalf, P., Blum, M., Freymann, D., Turner, M. & Wiley, D. C. Nature 325, 84–86 (1987).
Bricogne, G. Acta crystallogr. A32, 832–847 (1976).
Jones, T. A., Zou, J.-Y., Cowan, S. W. & Kjeldgaard, M. Acta crystallogr. A47, 110–119 (1991).
Brünger, A. T. X-PLOR (Version 3.1): A System for X-ray Crystallography and NMR (Yale Univ. Press, New Haven, 1992).
Lüthy, R., Bowie, J. U. & Eisenberg, D. Nature 356, 83–85 (1992).
Colovos, C. & Yeates, T. O. Prot. Sci. 2, 1511–1519 (1993).
Read, R. J. Acta crystallogr. A42, 140–149 (1986).
Kraulis, P. J. appl. Crystallogr. 24, 924–950 (1991).
Carson, M. J. molec. Graphics 5, 103–106 (1987).
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Bullough, P., Hughson, F., Skehel, J. et al. Structure of influenza haemagglutinin at the pH of membrane fusion. Nature 371, 37–43 (1994). https://doi.org/10.1038/371037a0
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1038/371037a0
This article is cited by
-
A stapled lipopeptide platform for preventing and treating highly pathogenic viruses of pandemic potential
Nature Communications (2024)
-
M6PR interacts with the HA2 subunit of influenza A virus to facilitate the fusion of viral and endosomal membranes
Science China Life Sciences (2024)
-
A mosaic influenza virus-like particles vaccine provides broad humoral and cellular immune responses against influenza A viruses
npj Vaccines (2023)
-
Structural characterisation of hemagglutinin from seven Influenza A H1N1 strains reveal diversity in the C05 antibody recognition site
Scientific Reports (2023)
-
In vitro anti-influenza assessment of anionic compounds ascorbate, acetate and citrate
Virology Journal (2022)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.