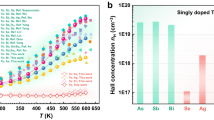
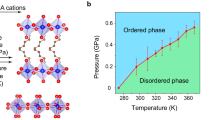
Abstract
ELECTRIDES are crystalline salts formed from complexed alkali-metal cations. There has been some dispute as to whether the valence electron from the alkali ion becomes a trapped interstitial anion1,2 or resides at or near the alkali-metal nucleus3. If the former description holds, electrides would represent stoichiometric counterparts of ionic insulators containing 'F-centre' electronic defects. Experiments1,2 have so far failed to resolve the question. Here we present ab initio self-consistent density-functional calculations4 of the electron distribution in the electride Cs+ (15-crown-5)2 · e-. We find that a spatially localized electron is located at the anion site, in accord with the F-centre model. Although the potential is in fact repulsive in this region, the electron is apparently forced to reside here by the need to lower its kinetic energy. We suggest that this picture may hold for other electrides as well.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 51 print issues and online access
$199.00 per year
only $3.90 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout
Similar content being viewed by others
References
Dye, J. L. & De Backer, M. G. A. Rev. Phys. Chem. 38, 271–301 (1987).
Dye, J. L. Science 247, 663–668 (1990).
Golden, S. & Tuttle, T. R. Jr Phys. Rev. B45, 13913–13918 (1992).
Singh, D. J., Krakauer, H., Haas, C. & Liu, A. Y. Phys. Rev. B46, 13065–13072 (1992).
Ward, D. L., Huang, R. H., Kuchenmeister, M. E. & Dye, J. L. Acta crystallogr. C46, 1831–1833 (1990).
Dawes, S. B., Eglin, J. L., Moeggenborg, K. J., Kim, J. & Dye, J. L. J. Am. chem. Soc. 113, 1605–1609 (1991).
Allan, G., De Backer, M. G., Lannoo, M. & Lefebvre, I. Europhys. Lett. 11, 49–53 (1990).
Rencsok, R., Kaplan, T. A. & Harrison, J. F. J. chem. Phys. 93, 5875–5882 (1990).
Ashcroft, N. W. & Mermin, N. D. Solid State Physics 685 (Holt, Rinehart & Winston, New York, 1976).
Pickett, W. E. Rev. mod. Phys. 61, 433–512 (1989).
Cohen, R. E., Pickett, W. E. & Krakauer, H. Phys. Rev. Lett. 62, 831–834 (1989).
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Singh, D., Krakauer, H., Haas, C. et al. Theoretical determination that electrons act as anions in the electride Cs+ (15-crown-5)2·e-. Nature 365, 39–42 (1993). https://doi.org/10.1038/365039a0
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1038/365039a0
This article is cited by
-
Electride characteristics of M2(η5-E5)2 (M = Be, Mg; E = Sb5-)
Structural Chemistry (2021)
-
Cavity-trapped electrons: lithium doped tetracyano-2,6-naphthoquinodimethane (TNAP) systems
Journal of Molecular Modeling (2020)
-
Identifying quasi-2D and 1D electrides in yttrium and scandium chlorides via geometrical identification
npj Computational Materials (2018)
-
Predicting the structure and stability of titanium oxide electrides
npj Computational Materials (2018)
-
Molecular graphs of \(\hbox {Mo}_{2n}\hbox {C}_n\) Mo 2 n C n (n = 1–10) clusters
Theoretical Chemistry Accounts (2016)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.