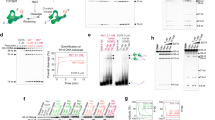
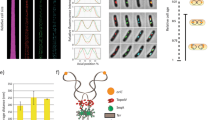
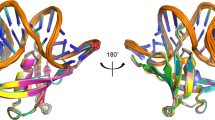
Abstract
THE DNA topoisomerases are ubiquitous enzymes that fulfil vital roles in the replication, transcription and recombination of DNA by carrying out DNA-strand passage reactions1–7. Here we characterize a prokaryotic counterpart to the eukaryotic topoisomerase I in the hyperthermophilic methanogen Methanopyrus handleri8–10. The new enzyme, called topoisomerase V, has the following properties in common with eukaryotic topoisomerase I, which distinguish it from all other known prokaryotic topoisomerases: (1) its activity is Mg2+-independent; (2) it relaxes both negatively and positively supercoiled DNA; (3) it makes a covalent complex with the 3' end of the broken DNA strand; and (4) it is recognized by antibody raised against human topoisomerase I. Eukaryotic-like enzymes have been discovered in some hyperthermophilic prokaryotes, namely the eocytes11 and the extremely thermophilic archaebacteria12, and hyperthermophilic homologues of eukaryotic DNA polymerase-α, transcription factor IIB and DNA ligase13–15 have all been reported. Thus our findings support the idea that some essential parts of the eukaryotic transcription–translation and replication machineries were in place before the emergence of eukaryotes, and that the closest living relatives of eukaryotes may be hyperthermophiles.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 51 print issues and online access
$199.00 per year
only $3.90 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout
Similar content being viewed by others
References
Gellert, M. A. Rev. Biochem. 50, 879–910 (1981).
Wang, J. C. A. Rev. Biochem. 54, 665–697 (1985).
Maxwell, A. & Gellert, M. Adv. Protein Chem. 38, 68–107 (1986).
Wang, J. C. J. biol. Chem. 266, 6659–6662 (1991).
Kornberg, A. & Baker, T. A. DNA Replication 379–401 (Freeman, New York, 1992).
Wang, J. C. & Liu, L. F. in DNA Topology and its Biological Effects (eds Cozzarelli, N. R. & Wang, J. C.) 321–340 (Cold Spring Harbor Laboratory Press, New York, 1990).
Wang, J. C., Caron, P. R. & Kim, R. A. Cell 62, 403–406 (1990).
Huber, R., Kurr, M., Jannasch, H. W. & Stetter, K. O. Nature 342, 833–834 (1989).
Kurr, M. et al. Arch. Microbiol. 156, 239–247 (1991).
Burggraf, S., Stetter, K. O., Rouviere, P. & Woese, C. R. Appl. Microbiol. 14, 346–351 (1991).
Rivera, M. C. & Lake, J. A. Science 257, 74–76 (1992).
Woese, C. R., Kandler, O. & Wheelis, M. L. Proc. natn. Acad. Sci. U.S.A. 87, 4576–4579 (1990).
Perler, F. B. et al. Proc. natn. Acad. Sci. U.S.A. 89, 5577–5581 (1992).
Ouzounis, C. & Sander, C. Cell 71, 189–190 (1992).
Kletzin, A. Nucleic Acids Res. 20, 5389–5386 (1992).
Slesarev, A. I., Zaitzev, D. A., Kopylov, V. M., Stetter, K. O. & Kozyavkin, S. A. J. biol. Chem. 266, 12321–12328 (1991).
Champoux, J. J. in DNA Topology and its Biological Effects (eds Cozzarelli, N. R. & Wang, J. C.) 217–242 (Cold Spring Harbor Laboratory Press, New York, 1990).
Bonven, B. J., Gocke, E. & Westergaard, O. Cell 41, 541–551 (1985).
Slesarev, A. I. Eur. J. Biochem. 173, 395–399 (1988).
Tse, Y.-C., Kirkegaard, K. & Wang, J. C. J. biol. Chem. 255, 5560–5565 (1980).
Kovalsky, O. I., Kozyavkin, S. A. & Slesarev, A. I. Nucleic Acids Res. 18, 2801–2805 (1990).
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Slesarev, A., Stetter, K., Lake, J. et al. DNA topoisomerase V is a relative of eukaryotic topoisomerase I from a hyperthermophilic prokaryote. Nature 364, 735–737 (1993). https://doi.org/10.1038/364735a0
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1038/364735a0
This article is cited by
-
Screening for natural inhibitors of human topoisomerases from medicinal plants with bio-affinity ultrafiltration and LC–MS
Phytochemistry Reviews (2020)
-
Structure of the N-terminal fragment of topoisomerase V reveals a new family of topoisomerases
The EMBO Journal (2006)
-
An eukaryotic-type DNA topoisomerase is present inHalobacterium halobium
Origins of life and evolution of the biosphere (1996)
-
Heat-stable enzymes from extremely thermophilic and hyperthermophilic microorganisms
World Journal of Microbiology & Biotechnology (1995)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.