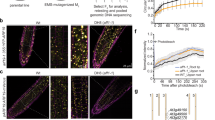
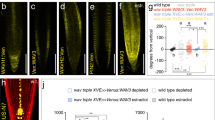
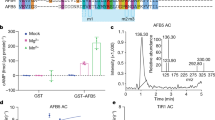
Abstract
The plant hormone auxin is central in many aspects of plant development. Previous studies have implicated the ubiquitin-ligase SCFTIR1 and the AUX/IAA proteins in auxin response. Dominant mutations in several AUX/IAA genes confer pleiotropic auxin-related phenotypes, whereas recessive mutations affecting the function of SCFTIR1 decrease auxin response. Here we show that SCFTIR1 is required for AUX/IAA degradation. We demonstrate that SCFTIR1 interacts with AXR2/IAA7 and AXR3/IAA17, and that domain II of these proteins is necessary and sufficient for this interaction. Further, auxin stimulates binding of SCFTIR1 to the AUX/IAA proteins, and their degradation. Because domain II is conserved in nearly all AUX/IAA proteins in Arabidopsis, we propose that auxin promotes the degradation of this large family of transcriptional regulators, leading to diverse downstream effects.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 51 print issues and online access
$199.00 per year
only $3.90 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout






Similar content being viewed by others
References
Gray, W. M. & Estelle, I. Function of the ubiquitin-proteasome pathway in auxin response. Trends Biochem. Sci. 25, 133–138 (2000).
Gray, W. M. et al. Identification of an SCF ubiquitin-ligase complex required for auxin response in Arabidopsis thaliana. Genes Dev. 13, 1678–1691 (1999).
Ruegger, M. et al. The TIR1 protein of Arabidopsis functions in auxin response and is related to human SKP2 and yeast grr1p. Genes Dev. 12, 198–207 (1998).
del Pozo, J. C., Timpte, C., Tan, S., Callis, J. & Estelle, M. The ubiquitin-related protein RUB1 and auxin response in Arabidopsis. Science 280, 1760–1763 (1998).
Lyapina, S. et al. Promotion of NEDD8-CUL1 conjugate cleavage by COP9 signalosome. Science 292, 1382–1385 (2001).
Osaka, F. et al. Covalent modifier NEDD8 is essential for SCF ubiquitin-ligase in fission yeast. EMBO J. 19, 3475–3484 (2000).
Podust, V. N. et al. A Nedd8 conjugation pathway is essential for proteolytic targeting of p27Kip1 by ubiquitination. Proc. Natl Acad. Sci. USA 97, 4579–4584 (2000).
del Pozo, J. C. & Estelle, M. The Arabidopsis cullin AtCUL1 is modified by the ubiquitin-related protein RUB1. Proc. Natl Acad. Sci. USA 96, 15342–15347 (1999).
Abel, S., Nguyen, M. D. & Theologis, A. The PS-IAA4/5-like family of early auxin-inducible mRNAs in Arabidopsis thaliana. J. Mol. Biol. 251, 533–549 (1995).
Kim, J., Harter, K. & Theologis, A. Protein–protein interactions among the Aux/IAA proteins. Proc. Natl Acad. Sci. USA 94, 11786–11791 (1997).
Ulmasov, T., Hagen, G. & Guilfoyle, T. J. ARF1, a transcription factor that binds to auxin response elements. Science 276, 1865–1868 (1997).
Ulmasov, T., Murfett, J., Hagen, G. & Guilfoyle, T. J. Aux/IAA proteins repress expression of reporter genes containing natural and highly active synthetic auxin response elements. Plant Cell 9, 1963–1971 (1997).
Nagpal, P. et al. AXR2 encodes a member of the Aux/IAA protein family. Plant Physiol. 123, 563–574 (2000).
Rogg, L. E., Lasswell, J. & Bartel, B. A gain-of-function mutation in iaa28 suppresses lateral root development. Plant Cell 13, 465–480 (2001).
Rouse, D., Mackay, P., Stirnberg, P., Estelle, M. & Leyser, O. Changes in auxin response from mutations in an AUX/IAA gene. Science 279, 1371–1373 (1998).
Tian, Q. & Reed, J. W. Control of auxin-regulated root development by the Arabidopsis thaliana SHY2/IAA3 gene. Development 126, 711–721 (1999).
Worley, C. K. et al. Degradation of Aux/IAA proteins is essential for normal auxin signalling. Plant J. 21, 553–562 (2000).
Ouellet, F., Overvoorde, P. J. & Theologis, A. IAA17/AXR3. Biochemical insight into an auxin mutant phenotype. Plant Cell 13, 829–842 (2001).
Smart, C. M., Scofield, S. R., Bevan, M. W. & Dyer, T. A. Delayed leaf senescence in tobacco plants transformed with TMR, a gene for cytokinin production in Agrobacterium. Plant Cell 3, 647–656 (1991).
Deshaies, R. J. SCF and Cullin/Ring H2-based ubiquitin ligases. Annu. Rev. Cell Dev. Biol. 15, 435–467 (1999).
Mockaitis, K. & Howell, S. H. Auxin induces mitogenic activated protein kinase (MAPK) activation in roots of Arabidopsis seedlings. Plant J. 24, 785–796 (2000).
Ramos, J. A., Zenser, N., Leyser, H. M. & Callis, J. Rapid degradation of Aux/IAA proteins requires conserved amino acids of domain II and is proteasome-dependent. Plant Cell 13, 2349–2360 (2001).
Ulmasov, T., Hagen, G. & Guilfoyle, T. J. Dimerization and DNA binding of auxin response factors. Plant J. 19, 309–319 (1999).
Ulmasov, T., Hagen, G. & Guilfoyle, T. J. Activation and repression of transcription by auxin-response factors. Proc. Natl Acad. Sci. USA 96, 5844–5849 (1999).
Berleth, T. & Jurgens, G. The role of the monopteros gene in organising the basal body region of the Arabidopsis embryo. Development 118, 575–587 (1993).
Karin, M. & Ben-Neriah, Y. Phosphorylation meets ubiquitination: the control of NF-κB activity. Annu. Rev. Immunol. 18, 621–663 (2000).
Abel, S., Oeller, P. W. & Theologis, A. Early auxin-induced genes encode short-lived nuclear proteins. Proc. Natl Acad. Sci. USA 91, 326–330 (1994).
Lincoln, C., Britton, J. H. & Estelle, M. Growth and development of the axr1 mutants of Arabidopsis. Plant Cell 2, 1071–1080 (1990).
Stomp, A.-M. in GUS Protocols (ed. Gallagher, S. R.) 103–113 (Academic, London, 1991).
Pringle, J. R. et al. Fluorescence microscopy methods for yeast. Methods Cell Biol. 31, 357–435 (1989).
Acknowledgements
This work was supported by grants from the National Institutes of Health (M.E.), the US Department of Energy (M.E.), the Texas Higher Education Coordinating Body Advance Research Program (M.E.) and the UK Biotechnology and Biological Science Research Council (O.L.).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Gray, W., Kepinski, S., Rouse, D. et al. Auxin regulates SCFTIR1-dependent degradation of AUX/IAA proteins. Nature 414, 271–276 (2001). https://doi.org/10.1038/35104500
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1038/35104500
This article is cited by
-
Regulation of endogenous hormone and miRNA in leaves of alfalfa (Medicago sativa L.) seedlings under drought stress by endogenous nitric oxide
BMC Genomics (2024)
-
Large-scale analysis of the ARF and Aux/IAA gene families in 406 horticultural and other plants
Molecular Horticulture (2024)
-
Analysis of co-expression and gene regulatory networks associated with sterile lemma development in rice
BMC Plant Biology (2023)
-
Nitrate stimulates adventitious rooting by increasing auxin content and regulating auxin- and root development-related genes expression in apple
Horticulture Advances (2023)
-
TaLBD1, a LOB transcription factor gene in T. aestivum, confers plant adaptation to low-N stress via modulating N acquisition-associated processes
Plant Cell, Tissue and Organ Culture (PCTOC) (2023)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.