Abstract
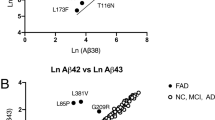
Alzheimer's disease is characterized by accumulation in the brain of a family of insoluble amyloid peptides (Aβ peptides)1, which are produced as a result of the normal processing of β-amyloid precursor protein (β-APP). Russo et al.2 claim that a truncated Aβ peptide that lacks the first ten amino acids accumulates in the brains of patients carrying a mutant form of pre-senilin 1 (PS1), a protein that is involved in cleavage of β-APP. However, we have found that this same species is also overrepresented in Alzheimer's patients with mutations in β-APP itself3. Our findings do not support the conclusion of Russo et al. that pathogenic PS1 mutations may control cleavage of β-APP by β-secretase4,5.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 51 print issues and online access
$199.00 per year
only $3.90 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout
Similar content being viewed by others
References
Masters, C. L. et al. Proc. Natl Acad. Sci. USA 82, 4245–4249 (1985).
Russo, C. et al. Nature 405, 531–532 (2000).
Naslund, J. et al. Proc. Natl Acad. Sci. USA 91, 8378–8382 (1994).
Li, Y. M. et al. Nature 405, 689–694 (2000).
Esler, W. P. et al. Nature Cell Biol. 2, 428–434 (2000).
Sherrington, R. et al. Nature 375, 754–760 (1995).
Cai, X. D., Golde, T. E. & Younkin, S. G. Science 259, 514–516 (1993).
Citron, M. et al. Nature 360, 672–674 (1992).
Gouras, G. K. et al. J. Neurochem. 71, 1920–1925 (1998).
Chen, F. et al. J. Biol. Chem. 275, 36794–36802 (2000).
Vassar, R. et al. Science 286, 735–741 (1999).
Miller, D. L. et al. Arch. Biochem. Biophys. 301, 41–52 (1993).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Gandy, S., Naslund, J. & Nordstedt, C. Molecular consequences of presenilin-1 mutation. Nature 411, 654–655 (2001). https://doi.org/10.1038/35079682
Published:
Issue Date:
DOI: https://doi.org/10.1038/35079682
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.