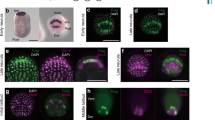
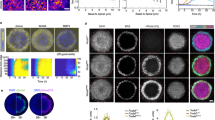
Abstract
During neural induction, the ‘organizer’ of the vertebrate embryo instructs neighbouring ectodermal cells to become nervous system rather than epidermis. This process is generally thought to occur around the mid-gastrula stage of embryogenesis1. Here we report the isolation of ERNI, an early response gene to signals from the organizer (Hensen's node). Using ERNI as a marker, we present evidence that neural induction begins before gastrulation—much earlier in development than previously thought. We show that the organizer and some of its precursor cells produce a fibroblast growth factor signal, which can initiate, and is required for, neural induction.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 51 print issues and online access
$199.00 per year
only $3.90 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
References
Harland, R. & Gerhart, J. Formation and function of Spemann's organizer. Annu. Rev. Cell Dev. Biol. 13, 611–667 (1997).
Wilson, P. A. & Hemmati-Brivanlou, A. Vertebrate neural induction: inducers, inhibitors, and a new synthesis. Neuron 18 , 699–710 (1997).
Schier, A. F. & Talbot, W. S. The zebrafish organizer. Curr. Opin. Genet. Dev. 8, 464–471 (1998).
Streit, A. et al. Chordin regulates primitive streak development and the stability of induced neural cells, but is not sufficient for neural induction in the chick embryo. Development 125, 507– 519 (1998).
Baker, J. C., Beddington, R. S. & Harland, R. M. Wnt signaling in Xenopus embryo inhibits BMP4 expression and activates neural development. Genes Dev. 13, 3149–3159 (1999).
Klingensmith, J., Ang, S. L., Bachiller, D. & Rossant, J. Neural induction and patterning in the mouse in the absence of the node and its derivatives. Dev. Biol. 216, 535– 549 (1999).
Streit, A. & Stern, C. D. Neural induction. A bird's eye view. Trends Genet. 15, 20– 24 (1999).
Gurdon, J. B. Embryonic induction—molecular prospects. Development 99, 285–306 (1987).
Streit, A. et al. Preventing the loss of competence for neural induction: HGF/SF, L5 and Sox-2. Development 124, 119– 1202 (1997).
Hatada, Y. & Stern, C. D. A fate map of the epiblast of the early chick embryo. Development 120, 2879 –2889 (1994).
Garcia-Martinez, V., Alvarez, I. S. & Schoenwolf, G. C. Locations of the ectodermal and nonectodermal subdivisions of the epiblast at stages 3 and 4 of avian gastrulation and neurulation. J. Exp. Zool. 267, 431–446 (1993).
Streit, A. & Stern, C. D. Establishment and maintenance of the border of the neural plate in the chick: involvement of FGF and BMP activity. Mech. Dev. 82, 51–66 (1999).
Chambers, D. & Mason, I. Expression of sprouty-2 during early development of the chick embryo is coincident with known sites of FGF signalling. Mech. Dev. 91, 361–364 (2000).
Storey, K. G. et al. Early posterior neural tissue is induced by FGF in the chick embryo. Development 125, 473– 484 (1998).
Mohammadi, M. et al. Structures of the tyrosine kinase domain of fibroblast growth factor receptor in complex with inhibitors. Science 276, 955–960 (1997).
Ye, W., Shimamura, K., Rubenstein, J. L., Hynes, M. A. & Rosenthal, A. FGF and Shh signals control dopaminergic and serotonergic cell fate in the anterior neural plate. Cell 93, 755–659 (1993).
Izpisua-Belmonte, J. C., De Robertis, E. M., Storey, K. G. & Stern, C. D. The homeobox gene goosecoid and the origin of organizer cells in the early chick blastoderm. Cell 74, 645– 659 (1993).
Launay, C., Fromentoux, V., Shi, D. L. & Boucaut, J. C. A truncated FGF receptor blocks neural induction by endogenous Xenopus inducers. Development 122, 869– 880 (1996).
Sasai, Y., Lu, B., Piccolo, S. & De Robertis, E. M. Endoderm induction by the organizer-secreted factors chordin and noggin in Xenopus animal caps. EMBO J. 15, 4547– 4555 (1996).
Amaya, E., Musci, T. J. & Kirschner, M. W. Expression of a dominant negative mutant of the FGF receptor disrupts mesoderm formation in Xenopus embryos. Cell 66, 257–270 ( 1991).
Lamb, T. M. & Harland, R. M. Fibroblast growth factor is a direct neural inducer, which combined with noggin generates anterior–posterior neural pattern. Development 121, 3627– 3636 (1995).
Alvarez, I. S., Araujo, M. & Nieto, M. A. Neural induction in whole chick embryo cultures by FGF. Dev. Biol. 199, 42–54 (1998).
Hongo, I., Kengaku, M. & Okamoto, H. FGF signaling and the anterior neural induction in Xenopus. Dev. Biol. 216, 561– 581 (1999).
Biehs, B., Francois, V. & Bier, E. The Drosophila short gastrulation gene prevents Dpp from autoactivating and suppressing neurogenesis in the neuroectoderm. Genes Dev. 10, 2922–2934 (1996).
Dulac, C. & Axel, R. A novel family of genes encoding putative pheromone receptors in mammals. Cell 83, 195–206 (1995).
Eyal-Giladi, H. & Kochav, S. From cleavage to primitive streak formation: a complementary normal table and a new look at the first stages of the development of the chick. I. General morphology. Dev. Biol. 49, 321–337 ( 1976).
Hamburger, V. & Hamilton, H. L. A series of normal stages in the development of the chick embryo. J. Morphol. 88 , 49–92 (1951).
Stern, C. D. & Ireland, G. W. An integrated experimental study of endoderm formation in avian embryos. Anat. Embryol. 163, 245–263 (1981).
Streit, A. & Stern, C. D. Mesoderm patterning and somite formation during node regression: differential effects of chordin and noggin. Mech. Dev. 85, 85–96 (1999).
Stern, C. D. Detection of multiple gene products simultaneously by in situ hybridization and immunohistochemistry in whole mounts of avian embryos. Curr. Top. Dev. Biol. 36, 223–243 (1998).
Acknowledgements
We thank A. Rosenthal and W. Ye (Genentech) for the FGFR1-IgG construct; R. Lovell-Badge for Sox3 and Sox2 and T. Jessell for S17; C. Dulac for advice on the differential screen; B. Cigich for technical assistance; I. Skromne for Fig. 4b, c; C. Ang for zebrafinch tissue; and T. Jessell, G. Sheng, K. Storey and D. Vasiliauskas for helpful comments on the manuscript. Supported by the National Institute of Mental Health.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Streit, A., Berliner, A., Papanayotou, C. et al. Initiation of neural induction by FGF signalling before gastrulation . Nature 406, 74–78 (2000). https://doi.org/10.1038/35017617
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1038/35017617
This article is cited by
-
Recent advances in the developmental origin of neuroblastoma: an overview
Journal of Experimental & Clinical Cancer Research (2022)
-
Exploring the roles of FGF/MAPK and cVG1/GDF signalling on mesendoderm induction and convergent extension during chick primitive streak formation
Development Genes and Evolution (2022)
-
hReg-CNCC reconstructs a regulatory network in human cranial neural crest cells and annotates variants in a developmental context
Communications Biology (2021)
-
Cadherins in early neural development
Cellular and Molecular Life Sciences (2021)
-
Brain organoids for the study of human neurobiology at the interface of in vitro and in vivo
Nature Neuroscience (2020)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.