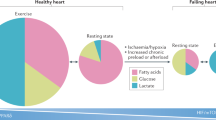
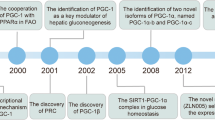
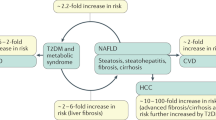
Abstract
In developed societies, chronic diseases such as diabetes, obesity, atherosclerosis and cancer are responsible for most deaths. These ailments have complex causes involving genetic, environmental and nutritional factors. There is evidence that a group of closely related nuclear receptors, called peroxisome proliferator-activated receptors (PPARs), may be involved in these diseases. This, together with the fact that PPAR activity can be modulated by drugs such as thiazolidinediones and fibrates, has instigated a huge research effort into PPARs1. Here we present the latest developments in the PPAR field, with particular emphasis on the physiological function of PPARs during various nutritional states, and the possible role of PPARs in several chronic diseases.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 51 print issues and online access
$199.00 per year
only $3.90 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout



Similar content being viewed by others
References
Desvergene, B. & Wahli, W. Peroxisome proliferator-activated receptors: nuclear control of metabolism. Endocr. Rev. 20, 649–688 (1999).
Lin, Q., Ruuska, S. E., Shaw, N. S., Dong, D. & Noy, N. Ligand selectivity of peroxisome proliferator activated receptor alpha. Biochemistry 38, 185–190 (1999).
Ellinghaus, P., Wolfrum, C., Assmann, G., Spencer, F. & Seedorf, U. Phytanic acid activates the peroxisome proliferator-activated receptor alpha (PPARalpha) in sterol carrier protein 2-/sterol carrier protein x-deficient mice. J. Biol. Chem. 274, 2766 –2772 (1999).
Moya-Camarena, S. Y., Vanden Heuvel, J. P., Blanchard, S. G., Leesnitzer, L. A. & Belury, M. A. Conjugated linoleic acid is a potent naturally occurring ligand and activator of PPARalpha. J. Lipid Res. 40, 426–1433 (1999).
Rosen, E. D. et al. PPAR is required for the differentiation of adipose tissue in vivo and in vitro. Mol. Cell 4, 611–617 (1999).
Basu-Modak, S. et al. Peroxisome proliferator-activated receptor beta regulates acyl-CoA synthetase 2 in reaggregated rat brain cell cultures. J. Biol. Chem. 274, 35881–35888 (1999).
Lim, H. et al. Cyclo-oxygenase-2-derived prostacyclin mediates embryo implantation in the mouse via PPARδ. Genes Dev. 13, 1561–1574 (1999).
Foretz, M. et al. ADD1/SREBP-1c is required in the activation of hepatic lipogenic gene expression by glucose. Mol. Cell. Biol. 19, 3760–3768 (1999).
Foretz, M., Guichard, C., Ferre, P. & Foufelle, F. Sterol regulatory element binding protein-1c is a major mediator of insulin action on the hepatic, expression of glucokinase and lipogenesis-related genes. Proc. Natl Acad. Sci. USA 96, 12737–12742 (1999).
Rieusset, J. et al. Insulin acutely regulates the expression of the peroxisome proliferator-activated receptor-gamma in human adipocytes. Diabetes 48, 699–705 ( 1999).
Fajas, L. et al. Regulation of peroxisome proliferator-activated receptor gamma expression by adipocyte differentiation and determination factor 1/sterol regulatory element binding protein 1: implications for adipocyte differentiation and metabolism. Mol. Cell. Biol. 19, 5495 –5503 (1999).
Wang, M. Y., Lee, Y. & Unger, R. H. Novel form of lipolysis induced by leptin. J. Biol. Chem. 274, 17541–17544 (1999).
Kubota, N. et al. PPAR mediates high-fat diet-induced adipocyte hypertrophy and insulin resistance. Mol. Cell 4, 597 –609 (1999).
Kersten, S. et al. Peroxisome proliferator activated receptor alpha mediates the adaptive response to fasting. J. Clin. Invest. 103, 1489–1498 (1999).
Leone, T. C., Weinheimer, C. J. & Kelly, D. P. A critical role for the peroxisome proliferator-activated receptor alpha (PPARalpha) in the cellular fasting response: the PARalpha-null mouse as a model of fatty acid oxidation disorders. Proc. Natl Acad. Sci. USA 96, 7473–7478 (1999).
Wang, M., Wise, S. C., Leff, T. & Su, T. Z. Troglitazone, an antidiabetic agent, inhibits cholesterol biosynthesis through a mechanism independent of peroxisome proliferator-activated receptor-gamma. Diabetes 48, 254–260 ( 1999).
Kockx, M. F. et al. Fibrates suppress fibrinogen gene expression in rodents via activation of the peroxisome proliferator-activated receptor-alpha. Blood 93, 2991–2998 ( 1999).
Devchand, P. R. et al. Chemical probes that differentially modulate peroxisome proliferator-activated receptor alpha and BLTR, nuclear and cell surface receptors for leukotriene B(4). J. Biol. Chem. 274, 23341– 23348 (1999).
Delerive, P. et al. Peroxisome proliferator-activated receptor alpha negatively regulates the vascular inflammatory gene response by negative cross-talk with transcription factors NF-kappaB and AP-1. J. Biol. Chem. 274, 32048–32054 (1999).
Hill, M. R. et al. Effect of peroxisome proliferator-activated receptor alpha activators on tumor necrosis factor expression in mice during endotoxemia. Infect. Immun. 67, 3488– 3493 (1999).
Ricote, M., Huang, J. T., Welch, J. S. & Glass, C. K. The peroxisome proliferator-activated receptor (PPARgamma) as a regulator of monocyte/macrophage function. J. Leukoc. Biol. 66 , 733–739 (1999).
Thieringer, R. et al. Activation of peroxisome proliferator-activated receptor gamma does not inhibit IL-6 or TNF-α responses of macrophages to lipopolysaccharide in vitro and in vivo. J. Immunol. 164, 1046–1054 (2000).
Demetri, G. D. et al. Induction of solid tumor differentiation by the peroxisome proliferator-activated receptor-gamma ligand troglitazone in patients with liposarcoma. Proc. Natl Acad. Sci. USA 96, 3951–3956 (1999).
Suh, N. et al. A new ligand for the peroxisome proliferator-activated receptor-gamma (PPAR-gamma), GW7845, inhibits rat mammary carcinogenesis. Cancer Res. 59, 5671–5673 ( 1999).
He, T. C., Chan, T. A., Vogelstein, B. & Kinzler, K. W. PPARdelta is an APC-regulated target of nonsteroidal anti-inflammatory drugs. Cell 99, 335–345 (1999).
Barak, Y. et al. PPAR gamma is required for placental, cardiac, and adipose tissue development. Mol. Cell 4, 585– 595 (1999).
Valve, R. et al. Two polymorphisms in the peroxisome proliferator-activated receptor-gamma gene are associated with severe overweight among obese women. J. Clin. Endocrinol. Metab. 84, 3708– 3712 (1999).
Ek, J. et al. Homozygosity of the Pro12A1a variant of the peroxisome proliferation-activated receptor-gamma2 (PPAR-gamma2): divergent modulating effects on body mass index in obese and lean Caucasian men. Diabetologia 42, 892–895 (1999).
Mancini, F. P. et al. Pro12A1a substitution in the peroxisome proliferator-activated receptor-gamma2 is not associated with type 2 diabetes. Diabetes 48, 1466–1468 ( 1999).
Barroso, I. et al. Dominant negative mutations in human PPARgamma associated with severe insulin resistance, diabetes mellitus and hypertension. Nature 402, 880–883 ( 1999).
Acknowledgements
S.K. was supported by fellowships from the European Molecular Biology Organization and the Roche Research Foundation. Work in the author's laboratory is financed by the Swiss National Science Foundation, the Etat de Vaud, and Human Frontier Science Programme.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Kersten, S., Desvergne, B. & Wahli, W. Roles of PPARs in health and disease. Nature 405, 421–424 (2000). https://doi.org/10.1038/35013000
Issue Date:
DOI: https://doi.org/10.1038/35013000
This article is cited by
-
Prostaglandin and prostaglandin receptors: present and future promising therapeutic targets for pulmonary arterial hypertension
Respiratory Research (2023)
-
Genetic and metabolic aspects of non-alcoholic fatty liver disease (NAFLD) pathogenicity
Egyptian Journal of Medical Human Genetics (2023)
-
Hydrogen alleviated cognitive impairment and blood‒brain barrier damage in sepsis-associated encephalopathy by regulating ABC efflux transporters in a PPARα-dependent manner
BMC Neuroscience (2023)
-
Empagliflozin induces the transcriptional program for nutrient homeostasis in skeletal muscle in normal mice
Scientific Reports (2023)
-
Utility of the burmese Python as a model for studying plasticity of extreme physiological systems
Journal of Muscle Research and Cell Motility (2023)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.