Abstract
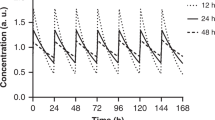
The use of insulin as an injected therapeutic agent for the treatment of diabetes has been one of the outstanding successes of modern medicine. The therapy has, however, had its associated problems, not least because injection of insulin does not lead to normal diurnal concentrations of insulin in the blood. This is especially true at meal times when absorption from subcutaneous tissue is too slow to mimic the normal rapid increments of insulin in the blood. In the neutral solutions used for therapy, insulin is mostly assembled as zinc-containing hexamers1 and this self-association, which under normal physiological circumstances functions to facilitate proinsulin transport, conversion and intracellular storage2, may limit the rate of absorption. We now report that it is possible, by single amino-acid substitutions, to make insulins which are essentially monomeric at pharmaceutical concentrations (0.6 mM) and which have largely preserved their biological activity. These monomeric insulins are absorbed two to three times faster after subcutaneous injection than the present rapid-acting insulins. They are therefore capable of giving diabetic patients a more physiological plasma insulin profile at the time of meal consumption.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 51 print issues and online access
$199.00 per year
only $3.90 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout
Similar content being viewed by others
References
Blundell, T. et al. Adv. Protein Chem. 26, 279–402 (1972).
Emdin, S. O. et al. Diabetologia 19, 174–182 (1980).
Brange, J. et al. Protein Engng. 1, 238 (1987).
Brange, J. et al. Diabetes 26, Suppl. 1, 77A (1987).
Hansen, J. F. & Brange, J. Protein Engn. 1, 250 (1987).
Melberg, S. G. Protein Engn. 1, 255–256 (1987).
Brange, J. et al. Diabetologia 30, 5O3A (1987).
Baker, E. N. et al. Phil. Trans. R. Soc. (in the press).
Gammeltoft, S. Physiol. Rev. 64, 1321–1378 (1984).
Fischer, W. H. et al. Biol. Chem. Hoppe-Seyler 366, 521–525 (1985).
Horuk, R. et al. Horon. Cell Reg. 4, 123–139 (1980).
Jones, D. H. et al. Biochemistry 24, 5852–5857 (1985).
Schwartz, G. P. et al. Proc. natn. Acad. Sci. U.S.A. 84, 6408–6411 (1987).
Derewenda, U. et al. Protein Engn. 1, 238–239 (1987).
Skyler, J. S. Diabetes Care 9, 666–667 (1986).
Thim, L. et al. Proc. natn. Acad. Sci. U.S.A. 83, 6766–6770 (1986).
Markussen, J. et al. in Peptides 1986. Proceedings of the Nineteenth European Peptide Symposium (ed. Theodoropoulos, D.) 189–194 (Walter de Gruyter, Berlin, 1987).
Markussen, J. et al. in Hormone Drugs (eds Gueriguian, J. L., Bransome, E.D. & Outschoorn, A. S.) 116–126 (United States Pharmacopeial Convention, Rockville, Maryland, 1982).
Maxam, A. & Gilbert, W. Meth. Enzym. 65, 499–560 (1980).
Elias, H.-G. in Characterization of Macromolecular Structure (ed. F. McIntyre) 28–50 (National Academy of Sciences, Washington, DC, 1968).
Moody, A. J. et al. Hormone Metab. Res. 6, 12–16 (1974).
Jones, T. A. J. appl. Chrystallogr. 11, 268–272 (1978).
Hubbard, R. E. in Computer Aided Molecular Design (Proceedings of a 2-day conference held in London October 1984) 99–106 (Oyez, London, 1985).
Wood, S. P. et al. Eur. J. Biochem. 55, 531–542 (1975).
Strickland, E. H. & Mercola, D. A. Biochemistry 15, 3875–3884 (1976).
Binder, C. Acta pharmac. tox. 27, (Suppl. 2) 1–87 (1969).
Ribel, U., et al. in Diabetes 1985 (eds Serrano-Rios M. & Lefèbvre, P. J.) 891–896 (Elsevier, Amsterdam, 1986).
Jørgensen, K. H. & Larsen, U. D. Diabetologia 19, 546–554 (1980).
Heding, L. G. Diabetologia 8, 260–266 (1972).
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Brange, J., Ribel, U., Hansen, J. et al. Monomeric insulins obtained by protein engineering and their medical implications. Nature 333, 679–682 (1988). https://doi.org/10.1038/333679a0
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1038/333679a0
This article is cited by
-
One hundred years of insulin therapy
Nature Reviews Endocrinology (2021)
-
Insulin at 100: still central in protein-based therapy for chronic disease
Communications Medicine (2021)
-
Elucidating the Mechanism of Absorption of Fast-Acting Insulin Aspart: The Role of Niacinamide
Pharmaceutical Research (2019)
-
Pursuit of a perfect insulin
Nature Reviews Drug Discovery (2016)
-
Structural basis for the poisonous activity of a predator's venom insulin
Nature Structural & Molecular Biology (2016)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.