Abstract
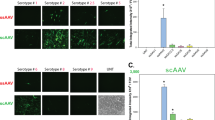
To date adeno-associated viral (AAV) vectors are the only gene therapy vectors that have been shown to efficiently transduce photoreceptor cells and have thus become the most commonly used vector for ocular transduction. Various AAV serotypes have been evaluated in the eye, the first of which was AAV2, which is able to transduce photoreceptors, retinal pigment epithelium (RPE) and retinal ganglion cells. AAV serotypes 1 and 4, as well as AAV2 pseudotyped with these capsids, only transduce the RPE. AAV serotype 5 and AAV2/5 transduce the photoreceptors as well as RPE, but not retinal ganglion cells. Here, we assessed the capacity of the novel serotype AAV2/8 to transduce various ocular tissues of the adult murine retina by administering AAV2/8 green fluorescent protein intravitreally, subretinally and intracamerally. We also determined the kinetics and efficiency of self-complementary AAV (scAAV) vectors of serotypes 2/2, 2/5 and 2/8 and compared them with single-stranded AAV (ssAAV). We found that ssAAV2/8 transduces photoreceptors and RPE more efficiently than ssAAV2/2 and ssAAV2/5, and that scAAV2/8 had faster onset and higher transgene expression than ssAAV2/8. This improved transduction efficiency might facilitate the development of improved gene therapy protocols for inherited retinal degenerations, particularly those caused by defects in photoreceptor-specific genes.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
References
Ali RR . Prospects for gene therapy. Novartis Found Symp 2004; 255: 165–172.
Bainbridge JW, Tan MH, Ali RR . Gene therapy progress and prospects: the eye. Gene Therapy 2006; 13: 1191–1197.
Goncalves MA . Adeno-associated virus: from defective virus to effective vector. Virol J 2005; 2: 43–44.
Sarra GM, Stephens C, Schlichtenbrede FC, Bainbridge JW, Thrasher AJ, Luthert PJ et al. Kinetics of transgene expression in mouse retina following sub-retinal injection of recombinant adeno-associated virus. Vision Res 2002; 42: 541–549.
Yang GS, Schmidt M, Yan Z, Lindbloom JD, Harding TC, Donahue BA et al. Virus-mediated transduction of murine retina with adeno-associated virus: effects of viral capsid and genome size. J Virol 2002; 76: 7651–7660.
Auricchio A, Kobinger G, Anand V, Hildinger M, O'Connor E, Maguire AM et al. Exchange of surface proteins impacts on viral vector cellular specificity and transduction characteristics: the retina as a model. Hum Mol Genet 2001; 10: 3075–3081.
Auricchio A, Hildinger M, O'Connor E, Gao GP, Wilson JM . Isolation of highly infectious and pure adeno-associated virus type 2 vectors with a single-step gravity-flow column. Hum Gene Ther 2001; 12: 71–76.
Auricchio A . Pseudotyped AAV vectors for constitutive and regulated gene expression in the eye. Vision Res 2003; 43: 913–918.
Weber M, Rabinowitz J, Provost N, Conrath H, Folliot S, Briot D et al. Recombinant adeno-associated virus serotype 4 mediates unique and exclusive long-term transduction of retinal pigmented epithelium in rat, dog, and nonhuman primate after subretinal delivery. Mol Ther 2003; 7: 774–781.
Ding W, Zhang L, Yan Z, Engelhardt JF . Intracellular trafficking of adeno-associated viral vectors. Gene Therapy 2005; 12: 873–880.
Thomas CE, Storm TA, Huang Z, Kay MA . Rapid uncoating of vector genomes is the key to efficient liver transduction with pseudotyped adeno-associated virus vectors. J Virol 2004; 78: 3110–3122.
Vincent-Lacaze N, Snyder RO, Gluzman R, Bohl D, Lagarde C, Danos O . Structure of adeno-associated virus vector DNA following transduction of the skeletal muscle. J Virol 1999; 73: 1949–1955.
Wang Z, Ma HI, Li J, Sun L, Zhang J, Xiao X . Rapid and highly efficient transduction by double-stranded adeno-associated virus vectors in vitro and in vivo. Gene Therapy 2003; 10: 2105–2111.
Broekman ML, Comer LA, Hyman BT, Sena-Esteves M . Adeno-associated virus vectors serotyped with AAV8 capsid are more efficient than AAV-1 or -2 serotypes for widespread gene delivery to the neonatal mouse brain. Neuroscience 2006; 138: 501–510.
Inagaki K, Fuess S, Storm TA, Gibson GA, Mctiernan CF, Kay MA et al. Robust systemic transduction with AAV9 vectors in mice: efficient global cardiac gene transfer superior to that of AAV8. Mol Ther 2006; 14: 45–53.
Wang Z, Zhu T, Rehman KK, Bertera S, Zhang J, Chen C et al. Widespread and stable pancreatic gene transfer by adeno-associated virus vectors via different routes. Diabetes 2006; 55: 875–884.
Davidoff AM, Gray JT, Ng CY, Zhang Y, Zhou J, Spence Y et al. Comparison of the ability of adeno-associated viral vectors pseudotyped with serotype 2, 5, and 8 capsid proteins to mediate efficient transduction of the liver in murine and nonhuman primate models. Mol Ther 2005; 11: 875–888.
Nathwani AC, Gray JT, Ng CY, Zhou J, Spence Y, Waddington SN et al. Self-complementary adeno-associated virus vectors containing a novel liver-specific human factor IX expression cassette enable highly efficient transduction of murine and nonhuman primate liver. Blood 2006; 107: 2653–2661.
Ali RR, Sarra GM, Stephens C, Alwis MD, Bainbridge JW, Munro PM et al. Restoration of photoreceptor ultrastructure and function in retinal degeneration slow mice by gene therapy. Nat Genet 2000; 25: 306–310.
Zhang X, De Alwis M, Hart SL, Fitzke FW, Inglis SC, Boursnell ME et al. High-titer recombinant adeno-associated virus production from replicating amplicons and herpes vectors deleted for glycoprotein H. Hum Gene Ther 1999; 10: 2527–2537.
Clark KR, Liu X, McGrath JP, Johnson PR . Highly purified recombinant adeno-associated virus vectors are biologically active and free of detectable helper and wild-type viruses. Hum Gene Ther 1999; 10: 1031–1039.
Davidoff AM, Ng CY, Sleep S, Gray J, Azam S, Zhao Y et al. Purification of recombinant adeno-associated virus type 8 vectors by ion exchange chromatography generates clinical grade vector stock. J Virol Methods 2004; 121: 209–215.
Akache B, Grimm D, Shen X, Fuess S, Yant SR, Glazer DS et al. A two-hybrid screen identifies cathepsins B and L as uncoating factors for adeno-associated virus 2 and 8. Mol Ther 2007; 15: 330–339.
Gao GP, Alvira MR, Wang L, Calcedo R, Johnston J, Wilson JM . Novel adeno-associated viruses from rhesus monkeys as vectors for human gene therapy. Proc Natl Acad Sci USA 2002; 99: 11854–11859.
Wang L, Cao O, Swalm B, Dobrzynski E, Mingozzi F, Herzog RW . Major role of local immune responses in antibody formation to factor IX in AAV gene transfer. Gene Therapy 2005; 12: 1453–1464.
Allocca M, Mussolino C, Hoyos MG, Sanges D, Iodice C, Petrillo M et al. Novel AAV serotypes efficiently transduce murine photoreceptors. J Virol 2007; 81: 11372–11380.
Ren C, Kumar S, Shaw DR, Ponnazhagan S . Genomic stability of self-complementary adeno-associated virus 2 during early stages of transduction in mouse muscle in vivo. Hum Gene Ther 2005; 16: 1047–1057.
Buch PK, MacLaren RE, Duran Y, Balaggan KS, MacNeil A, Schlichtenbrede FC et al. In contrast to AAV-mediated Cntf expression, AAV-mediated Gdnf expression enhances gene replacement therapy in rodent models of retinal degeneration. Mol Ther 2006; 14: 700–709.
Broderick CA, Smith AJ, Balaggan KS, Georgarias A, Buch PK, Trittibach PC et al. Local administration of an adeno-associated viral vector expressing IL-10 reduces monocyte infiltration and subsequent photoreceptor damage during experimental autoimmune uveitis. Mol Ther 2005; 12: 369–373.
Ali RR, Reichel MB, Thrasher AJ, Levinsky RJ, Kinnon C, Kanuga N et al. Gene transfer into the mouse retina mediated by an adeno-associated viral vector. Hum Mol Genet 1996; 5: 591–594.
Bennett J, Duan D, Engelhardt JF, Maguire AM . Real-time, noninvasive in vivo assessment of adeno-associated virus-mediated retinal transduction. Invest Ophthalmol Vis Sci 1997; 38: 2857–2863.
Grant CA, Ponnazhagan S, Wang XS, Srivastava A, Li T . Evaluation of recombinant adeno-associated virus as a gene transfer vector for the retina. Curr Eye Res 1997; 16: 949–956.
Lai YK, Rolling F, Baker E, Rakoczy PE . Kinetics of efficient recombinant adeno-associated virus transduction in retinal pigment epithelial cells. Exp Cell Res 2001; 267: 184–192.
Rolling F, Shen WY, Tabarias H, Constable I, Kanagasingam Y, Barry CJ et al. Evaluation of adeno-associated virus-mediated gene transfer into the rat retina by clinical fluorescence photography. Hum Gene Ther 1999; 10: 641–648.
Dudus L, Anand V, Acland GM, Chen SJ, Wilson JM, Fisher KJ et al. Persistent transgene product in retina, optic nerve and brain after intraocular injection of rAAV. Vision Res 1999; 39: 2545–2553.
Smith AJ, Schlichtenbrede FC, Tschernutter M, Bainbridge JW, Thrasher AJ, Ali RR . AAV-mediated gene transfer slows photoreceptor loss in the RCS rat model of retinitis pigmentosa. Mol Ther 2003; 8: 188–195.
Acland GM, Aguirre GD, Ray J, Zhang Q, Aleman TS, Cideciyan AV et al. Gene therapy restores vision in a canine model of childhood blindness. Nat Genet 2001; 28: 92–95.
Schlichtenbrede FC, Da Cruz L, Stephens C, Smith AJ, Georgiadis A, Thrasher AJ et al. Long-term evaluation of retinal function in Prph2Rd2/Rd2 mice following AAV-mediated gene replacement therapy. J Gene Med 2003; 5: 757–764.
Acknowledgements
This work was supported by grants from the European Commission (EVI-Genoret and Clinigene) and a grant from the department of health and from the Special Trustees of Moorfields Eye Hospital to RRA and a grant from the Katharine Dormandy Trust, UK to ACN.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Natkunarajah, M., Trittibach, P., McIntosh, J. et al. Assessment of ocular transduction using single-stranded and self-complementary recombinant adeno-associated virus serotype 2/8. Gene Ther 15, 463–467 (2008). https://doi.org/10.1038/sj.gt.3303074
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/sj.gt.3303074
Keywords
This article is cited by
-
X-Linked Retinitis Pigmentosa Gene Therapy: Preclinical Aspects
Ophthalmology and Therapy (2023)
-
PEG-mediated transduction of rAAV as a platform for spatially confined and efficient gene delivery
Biomaterials Research (2022)
-
Gene therapy in color vision deficiency: a review
International Ophthalmology (2021)
-
Focused Update on AAV-Based Gene Therapy Clinical Trials for Inherited Retinal Degeneration
BioDrugs (2020)
-
AAVrh-10 transduces outer retinal cells in rodents and rabbits following intravitreal administration
Gene Therapy (2019)