Abstract
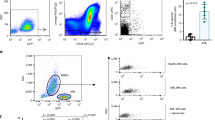
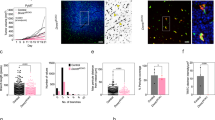
Human adult blood late outgrowth endothelial cells (BOECs) are potential yet untested cellular vehicles to target tumor-cytotoxic effectors to tumors. We show that, following intravenous injection into irradiated mice, BOECs home to Lewis lung carcinoma (LLC) lung metastases, but less so to liver or kidney metastases. BOECs targeted most but not all of the lung metastases, to a different degree. While most of the homed BOECs took up an extravascular position, some integrated into tumor vessels. Sequestration into normal tissue was low. Placental growth factor mediated both migration and invasion of BOECs into LLC spheroid masses in vitro, as did VEGF. When armed with a suicide gene, BOECs exerted a bystander effect on LLC cells in vitro and in vivo. Surprisingly, i.v. administration of armed BOECs into mice bearing multi-organ LLC metastases did not prolong survival. In addition to homing efficacy other parameters impacted upon the efficacy of BOECs. These include the ultimate susceptibility of BOECs to suicide gene-induced cell death, their paracrine proliferative effect on LLC cells and their low proliferation rate compared to LLC cells. Addressing these determinants may make BOECs a useful addition to the arsenal of tumor-targeting moieties.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout






Similar content being viewed by others
References
Asahara T, Takahashi T, Masuda H, Kalka C, Chen D, Iwaguro H et al. VEGF contributes to postnatal neovascularization by mobilizing bone marrow-derived endothelial progenitor cells. EMBO J 1999; 18: 3964–3972.
Arbab AS, Pandit SD, Anderson SA, Yocum GT, Bur M, Khuu HM et al. MRI and confocal microscopy studies of magnetically labeled endothelial progenitor cells trafficking to sites of tumor angiogenesis. Stem Cells 2006; 24: 671–678.
Asahara T, Masuda H, Takahashi T, Kalka C, Pastore C, Silver M et al. Bone marrow origin of endothelial progenitor cells responsible for postnatal vasculogenesis in physiological and pathological neovascularization. Circ Res 1999; 85: 221–228.
Coussens LM, Tinkle CL, Hanahan D, Werb Z . MMP-9 supplied by bone marrow-derived cells contributes to skin carcinogenesis. Cell 2000; 103: 481–490.
Davidoff AM, Ng CY, Brown P, Leary MA, Spurbeck WW, Zhou J et al. Bone marrow-derived cells contribute to tumor neovasculature and, when modified to express an angiogenesis inhibitor, can restrict tumor growth in mice. Clin Cancer Res 2001; 7: 2870–2879.
Lyden D, Hattori K, Dias S, Costa C, Blaikie P, Butros L et al. Impaired recruitment of bone-marrow-derived endothelial and hematopoietic precursor cells blocks tumor angiogenesis and growth. Nat Med 2001; 7: 1194–1201.
De Palma M, Venneri MA, Roca C, Naldini L . Targeting exogenous genes to tumor angiogenesis by transplantation of genetically modified hematopoietic stem cells. Nat Med 2003; 9: 789–795.
Garcia-Barros M, Paris F, Cordon-Cardo C, Lyden D, Rafii S, Haimovitz-Friedman A et al. Tumor response to radiotherapy regulated by endothelial cell apoptosis. Science 2003; 300: 1155–1159.
Machein MR, Renninger S, de Lima-Hahn E, Plate KH . Minor contribution of bone marrow-derived endothelial progenitors to the vascularization of murine gliomas. Brain Pathol 2003; 13: 582–597.
Ruzinova MB, Schoer RA, Gerald W, Egan JE, Pandolfi PP, Rafii S et al. Effect of angiogenesis inhibition by Id loss and the contribution of bone-marrow-derived endothelial cells in spontaneous murine tumors. Cancer Cell 2003; 4: 277–289.
Gothert JR, Gustin SE, van Eekelen JA, Schmidt U, Hall MA, Jane SM et al. Genetically tagging endothelial cells in vivo: bone marrow-derived cells do not contribute to tumor endothelium. Blood 2004; 104: 1769–1777.
Hilbe W, Dirnhofer S, Oberwasserlechner F, Schmid T, Gunsilius E, Hilbe G et al. CD133 positive endothelial progenitor cells contribute to the tumour vasculature in non-small cell lung cancer. J Clin Pathol 2004; 57: 965–969.
Li H, Gerald WL, Benezra R . Utilization of bone marrow-derived endothelial cell precursors in spontaneous prostate tumors varies with tumor grade. Cancer Res 2004; 64: 6137–6143.
Rajantie I, Ilmonen M, Alminaite A, Ozerdem U, Alitalo K, Salven P . Adult bone marrow-derived cells recruited during angiogenesis comprise precursors for periendothelial vascular mural cells. Blood 2004; 104: 2084–2086.
Strobel P, Hartmann M, Jakob A, Mikesch K, Brink I, Dirnhofer S et al. Thymic carcinoma with overexpression of mutated KIT and the response to imatinib. N Engl J Med 2004; 350: 2625–2626.
Yang L, DeBusk LM, Fukuda K, Fingleton B, Green-Jarvis B, Shyr Y et al. Expansion of myeloid immune suppressor Gr+CD11b+ cells in tumor-bearing host directly promotes tumor angiogenesis. Cancer Cell 2004; 6: 409–421.
Anderson SA, Glod J, Arbab AS, Noel M, Ashari P, Fine HA et al. Noninvasive MR imaging of magnetically labeled stem cells to directly identify neovasculature in a glioma model. Blood 2005; 105: 420–425.
De Palma M, Venneri MA, Galli R, Sergi LS, Politi LS, Sampaolesi M et al. Tie2 identifies a hematopoietic lineage of proangiogenic monocytes required for tumor vessel formation and a mesenchymal population of pericyte progenitors. Cancer Cell 2005; 8: 211–226.
Larrivee B, Niessen K, Pollet I, Corbel SY, Long M, Rossi FM et al. Minimal contribution of marrow-derived endothelial precursors to tumor vasculature. J Immunol 2005; 175: 2890–2899.
Peters BA, Diaz LA, Polyak K, Meszler L, Romans K, Guinan EC et al. Contribution of bone marrow-derived endothelial cells to human tumor vasculature. Nat Med 2005; 11: 261–262.
Shinde Patil VR, Friedrich EB, Wolley AE, Gerszten RE, Allport JR, Weissleder R . Bone marrow-derived lin(−)c-kit(+)Sca-1+ stem cells do not contribute to vasculogenesis in Lewis lung carcinoma. Neoplasia 2005; 7: 234–240.
Duda DG, Cohen KS, Kozin SV, Perentes JY, Fukumura D, Scadden DT et al. Evidence for bone marrow-derived endothelial cells incorporation into perfused blood vessels in tumors. Blood 2006; 107: 2774–2776.
Ferrari N, Glod J, Lee J, Kobiler D, Fine HA . Bone marrow-derived, endothelial progenitor-like cells as angiogenesis-selective gene-targeting vectors. Gene Ther 2003; 10: 647–656.
Arafat WO, Casado E, Wang M, Alvarez RD, Siegal GP, Glorioso JC et al. Genetically modified CD34+ cells exert a cytotoxic bystander effect on human endothelial and cancer cells. Clin Cancer Res 2000; 6: 4442–4448.
Moore XL, Lu J, Sun L, Zhu CJ, Tan P, Wong MC . Endothelial progenitor cells’ ‘homing’ specificity to brain tumors. Gene Therapy 2004; 11: 811–818.
Tabatabai G, Frank B, Mohle R, Weller M, Wick W . Irradiation and hypoxia promote homing of haematopoietic progenitor cells towards gliomas by TGF-beta-dependent HIF-1alpha-mediated induction of CXCL12. Brain 2006; 129: 2426–2435.
Jevremovic D, Gulati R, Hennig I, Diaz RM, Cole C, Kleppe L et al. Use of blood outgrowth endothelial cells as virus-producing vectors for gene delivery to tumors. Am J Physiol Heart Circ Physiol 2004; 287: H494–H500.
Le Ricousse-Roussanne S, Barateau V, Contreres JO, Boval B, Kraus-Berthier L, Tobelem G . Ex vivo differentiated endothelial and smooth muscle cells from human cord blood progenitors home to the angiogenic tumor vasculature. Cardiovasc Res 2004; 62: 176–184.
Lin Y, Weisdorf DJ, Solovey A, Hebbel RP . Origins of circulating endothelial cells and endothelial outgrowth from blood. J Clin Invest 2000; 105: 71–77.
Lin Y, Chang L, Solovey A, Healey JF, Lollar P, Hebbel RP . Use of blood outgrowth endothelial cells for gene therapy for hemophilia A. Blood 2002; 99: 457–462.
Hicklin DJ, Ellis LM . Role of the vascular endothelial growth factor pathway in tumor growth and angiogenesis. J Clin Oncol 2005; 23: 1011–1027.
Ferrara N, Gerber HP, LeCouter J . The biology of VEGF and its receptors. Nat Med 2003; 9: 669–676.
Gill M, Dias S, Hattori K, Rivera ML, Hicklin D, Witte L et al. Vascular trauma induces rapid but transient mobilization of VEGFR2(+)AC133(+) endothelial precursor cells. Circ Res 2001; 88: 167–174.
Heissig B, Hattori K, Dias S, Friedrich M, Ferris B, Hackett NR et al. Recruitment of stem and progenitor cells from the bone marrow niche requires MMP-9 mediated release of kit-ligand. Cell 2002; 109: 625–637.
Autiero M, Luttun A, Tjwa M, Carmeliet P . Placental growth factor and its receptor, vascular endothelial growth factor receptor-1: novel targets for stimulation of ischemic tissue revascularization and inhibition of angiogenic and inflammatory disorders. J Thromb Haemost 2003; 1: 1356–1370.
Clauss M, Weich H, Breier G, Knies U, Rockl W, Waltenberger J et al. The vascular endothelial growth factor receptor Flt-1 mediates biological activities. Implications for a functional role of placenta growth factor in monocyte activation and chemotaxis. J Biol Chem 1996; 271: 17629–17634.
Carmeliet P, Moons L, Luttun A, Vincenti V, Compernolle V, De Mol M et al. Synergism between vascular endothelial growth factor and placental growth factor contributes to angiogenesis and plasma extravasation in pathological conditions. Nat Med 2001; 7: 575–583.
Hiratsuka S, Maru Y, Okada A, Seiki M, Noda T, Shibuya M . Involvement of Flt-1 tyrosine kinase (vascular endothelial growth factor receptor-1) in pathological angiogenesis. Cancer Res 2001; 61: 1207–1213.
Adini A, Kornaga T, Firoozbakht F, Benjamin LE . Placental growth factor is a survival factor for tumor endothelial cells and macrophages. Cancer Res 2002; 62: 2749–2752.
Zhang L, Chen J, Ke Y, Mansel RE, Jiang WG . Expression of Placenta growth factor (PlGF) in non-Small cell Lung cancer (NSCLC) and the clinical and prognostic significance. World J Surg Oncol 2005; 3: 68.
Ikai T, Miwa H, Shikami M, Hiramatsu A, Tajima E, Yamamoto H et al. Placenta growth factor stimulates the growth of Philadelphia chromosome positive acute lymphoblastic leukemia cells by both autocrine and paracrine pathways. Eur J Haematol 2005; 75: 273–279.
Donnini S, Machein MR, Plate KH, Weich HA . Expression and localization of placenta growth factor and PlGF receptors in human meningiomas. J Pathol 1999; 189: 66–71.
Lacal PM, Failla CM, Pagani E, Odorisio T, Schietroma C, Falcinelli S et al. Human melanoma cells secrete and respond to placenta growth factor and vascular endothelial growth factor. J Invest Dermatol 2000; 115: 1000–1007.
Wei SC, Tsao PN, Yu SC, Shun CT, Tsai-Wu JJ, Wu CH et al. Placenta growth factor expression is correlated with survival of patients with colorectal cancer. Gut 2005; 54: 666–672.
Chen CN, Hsieh FJ, Cheng YM, Cheng WF, Su YN, Chang KJ et al. The significance of placenta growth factor in angiogenesis and clinical outcome of human gastric cancer. Cancer Lett 2004; 213: 73–82.
Matsumoto K, Suzuki K, Koike H, Okamura K, Tsuchiya K, Uchida T et al. Prognostic significance of plasma placental growth factor levels in renal cell cancer: an association with clinical characteristics and vascular endothelial growth factor levels. Anticancer Res 2003; 23: 4953–4958.
Taylor AP, Osorio L, Craig R, Raleigh JA, Ying Z, Goldenberg DM et al. Tumor-specific regulation of angiogenic growth factors and their receptors during recovery from cytotoxic therapy. Clin Cancer Res 2002; 8: 1213–1222.
Luttun A, Tjwa M, Moons L, Wu Y, Angelillo-Scherrer A, Liao F et al. Revascularization of ischemic tissues by PlGF treatment, and inhibition of tumor angiogenesis, arthritis and atherosclerosis by anti-Flt1. Nat Med 2002; 8: 831–840.
Aicher A, Brenner W, Zuhayra M, Badorff C, Massoudi S, Assmus B et al. Assessment of the tissue distribution of transplanted human endothelial progenitor cells by radioactive labeling. Circulation 2003; 107: 2134–2139.
Virgolini I, Angelberger P, Li SR, Koller F, Koller E, Pidlich J et al. Indium-111-labeled low-density lipoprotein binds with higher affinity to the human liver as compared to iodine-123-low-density-labeled lipoprotein. J Nucl Med 1991; 32: 2132–2138.
Erbs P, Regulier E, Kintz J, Leroy P, Poitevin Y, Exinger F et al. In vivo cancer gene therapy by adenovirus-mediated transfer of a bifunctional yeast cytosine deaminase/uracil phosphoribosyltransferase fusion gene. Cancer Res 2000; 60: 3813–3822.
Huber BE, Austin EA, Richards CA, Davis ST, Good SS . Metabolism of 5-fluorocytosine to 5-fluorouracil in human colorectal tumor cells transduced with the cytosine deaminase gene: significant antitumor effects when only a small percentage of tumor cells express cytosine deaminase. Proc Natl Acad Sci USA 1994; 91: 8302–8306.
Pasqualini R, Arap W, McDonald DM . Probing the structural and molecular diversity of tumor vasculature. Trends Mol Med 2002; 8: 563–571.
Rumpold H, Wolf D, Koeck R, Gunsilius E . Endothelial progenitor cells: a source for therapeutic vasculogenesis? J Cell Mol Med 2004; 8: 509–518.
Ishizawa K, Kubo H, Yamada M, Kobayashi S, Suzuki T, Mizuno S et al. Hepatocyte growth factor induces angiogenesis in injured lungs through mobilizing endothelial progenitor cells. Biochem Biophys Res Commun 2004; 324: 276–280.
Li H, Gerald WL, Benezra R . Utilization of bone marrow-derived endothelial cell precursors in spontaneous prostate tumors varies with tumor grade. Cancer Res 2004; 64: 6137–6143.
Gorski DH, Beckett MA, Jaskowiak NT, Calvin DP, Mauceri HJ, Salloum RM et al. Blockage of the vascular endothelial growth factor stress response increases the antitumor effects of ionizing radiation. Cancer Res 1999; 59: 3374–3378.
Sonveaux P, Brouet A, Havaux X, Gregoire V, Dessy C, Balligand JL et al. Irradiation-induced angiogenesis through the up-regulation of the nitric oxide pathway: implications for tumor radiotherapy. Cancer Res 2003; 63: 1012–1019.
Heissig B, Rafii S, Akiyama H, Ohki Y, Sato Y, Rafael T et al. Low-dose irradiation promotes tissue revascularization through VEGF release from mast cells and MMP-9-mediated progenitor cell mobilization. J Exp Med 2005; 202: 739–750.
Acknowledgements
We thank H Ribbert from the Department of Nuclear Medicine for help in preparing and detecting 111In-DTP-Ac-LDL-loaded BOECs, M Shibuya for the LLC and LLC-PLGF cell lines, P Vajkoczy for SF126 cells, W Wick for LNT-229 cells and for introducing us to stereotactic manipulations, S Zhou for providing human fibroblasts, S Miebach for introduction to spheroid assays and I Gastrock-Balitsch, U Kirchner and H Knauss for excellent technical help. Supported by a grant from the Wilhelm Sander-Stiftung (to CB).
Author information
Authors and Affiliations
Corresponding author
Additional information
Supplementary Information accompanies the paper on Gene Therapy website (http://www.nature.com/gt)
Rights and permissions
About this article
Cite this article
Wei, J., Jarmy, G., Genuneit, J. et al. Human blood late outgrowth endothelial cells for gene therapy of cancer: determinants of efficacy. Gene Ther 14, 344–356 (2007). https://doi.org/10.1038/sj.gt.3302860
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/sj.gt.3302860
Keywords
This article is cited by
-
Endothelial progenitor cells support tumour growth and metastatisation: implications for the resistance to anti-angiogenic therapy
Tumor Biology (2015)
-
Blood outgrowth endothelial cell-based systemic delivery of antiangiogenic gene therapy for solid tumors
Cancer Gene Therapy (2010)
-
Endothelial progenitor cells for cancer gene therapy
Gene Therapy (2008)
-
Targeted release of oncolytic measles virus by blood outgrowth endothelial cells in situ inhibits orthotopic gliomas
Gene Therapy (2007)