Abstract
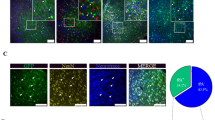
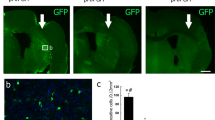
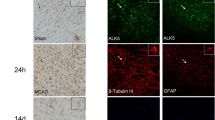
Gene therapy may be a promising approach for treatment of brain ischemia. In this study, we examined the effect of postischemic gene transfer of midkine, a heparin-binding neurotrophic factor, using a focal brain ischemia model with the photothrombotic occlusion method. At 90 min after induction of brain ischemia in spontaneously hypertensive rats, a replication-deficient recombinant adenovirus encoding mouse midkine (AdMK, n=7) or a control vector encoding β-galactosidase (Adβgal, n=7) was injected into the lateral ventricle ipsilateral to ischemia. At 2 days after ischemia, we determined infarct volume by 2,3,5-triphenyltetrazolium chloride staining. There were no significant differences in cerebral blood flow 1 h after ischemia between AdMK and Adβgal groups. Infarct volume of AdMK group was 51±27 mm3, which was significantly smaller than that of Adβgal group (86±27 mm3, P<0.05). TUNEL-positive and cleaved caspase-3-positive cells in the periischemic area of AdMK-treated rats were significantly fewer than those in Adβgal-treated rats, suggesting that the reduction of infarct volume by midkine was partly mediated by its antiapoptotic action. Thus, gene transfer of midkine to the ischemic brain may be effective in the treatment of brain ischemia.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
References
Tomomura M et al. A retinoic acid-responsive gene, MK, found in the teratocarcinoma system. Heterogeneity of the transcript and the nature of the translation product. J Biol Chem 1990; 265: 10765–10770.
Kadomatsu K, Tomomura M, Muramatsu T . cDNA cloning and sequencing of a new gene intensely expressed in early differentiation stages of embryonal carcinoma cells and in mid-gestation period of mouse embryogenesis. Biochem Biophys Res Commun 1988; 151: 1312–1318.
Muramatsu H, Muramatsu T . Purification of recombinant midkine and examination of its biological activities: functional comparison of new heparin binding factors. Biochem Biophys Res Commun 1991; 177: 652–658.
Muramatsu H et al. Midkine, a retinoic acid-inducible growth/differentiation factor: immunochemical evidence for the function and distribution. Dev Biol 1993; 159: 392–402.
Kikuchi S et al. Midkine, a novel neurotrophic factor, promotes survival of mesencephalic neurons in culture. Neurosci Lett 1993; 160: 9–12.
Michikawa M et al. Retinoic acid responsive gene product, midkine, has neurotrophic functions for mouse spinal cord and dorsal root ganglion neurons in culture. J Neurosci Res 1993; 35: 530–539.
Satoh J et al. Midkine that promotes survival of fetal human neurons is produced by fetal human astrocytes in culture. Brain Res Dev Brain Res 1993; 75: 201–205.
Unoki K et al. Rescue of photoreceptors from the damaging effects of constant light by midkine, a retinoic acid-responsive gene product. Invest Ophthalmol Vis Sci 1994; 35: 4063–4068.
Kojima S et al. Midkine enhances fibrinolytic activity of bovine endothelial cells. J Biol Chem 1995; 270: 9590–9596.
Yoshida Y et al. Midkine is present in the early stage of cerebral infarct. Brain Res Dev Brain Res 1995; 85: 25–30.
Yao H et al. DNA fragmentation in ischemic core and penumbra in focal cerebral ischemia in rats. Brain Res Mol Brain Res 2001; 91: 112–118.
Owada K et al. Midkine inhibits caspase-dependent apoptosis via the activation of mitogen-activated protein kinase and phosphatidylinositol 3-kinase in cultured neurons. J Neurochem 1999; 73: 2084–2092.
Yoshida Y et al. Intraventricular administration of the neurotrophic factor midkine ameliorates hippocampal delayed neuronal death following transient forebrain ischemia in gerbils. Brain Res 2001; 894: 46–55.
Heistad DD, Faraci FM . Gene therapy for cerebral vascular disease. Stroke 1996; 27: 1688–1693.
Ooboshi H et al. Adenovirus-mediated gene transfer in vivo to cerebral blood vessels and perivascular tissue. Circ Res 1995; 77: 7–13.
Betz AL, Yang GY, Davidson BL . Attenuation of stroke size in rats using an adenoviral vector to induce overexpression of interleukin-1 receptor antagonist in brain. J Cereb Blood Flow Metab 1995; 15: 547–551.
Xu DG et al. Elevation of neuronal expression of NAIP reduces ischemic damage in the rat hippocampus. Nat Med 1997; 3: 997–1004.
Yang GY et al. Attenuation of ischemia-induced mouse brain injury by SAG, a redox-inducible antioxidant protein. J Cereb Blood Flow Metab 2001; 21: 722–733.
Zhang WR et al. Therapeutic time window of adenovirus-mediated GDNF gene transfer after transient middle cerebral artery occlusion in rat. Brain Res 2002; 947: 140–145.
Hayashi K et al. Gene therapy for preventing neuronal death using hepatocyte growth factor: in vivo gene transfer of HGF to subarachnoid space prevents delayed neuronal death in gerbil hippocampal CA1 neurons. Gene Therapy 2001; 8: 1167–1173.
Kumon Y et al. Transient increase in endogenous basic fibroblast growth factor in neurons of ischemic rat brains. Brain Res 1993; 605: 169–174.
Kokaia Z et al. Rapid alterations of BDNF protein levels in the rat brain after focal ischemia: evidence for increased synthesis and anterograde axonal transport. Exp Neurol 1998; 154: 289–301.
Sugimori H, Speller H, Finklestein SP . Intravenous basic fibroblast growth factor produces a persistent reduction in infarct volume following permanent focal ischemia in rats. Neurosci Lett 2001; 300: 13–16.
Schäbitz WR et al. Intravenous brain-derived neurotrophic factor reduces infarct size and counterregulates Bax and Bcl-2 expression after temporary focal cerebral ischemia. Stroke 2000; 31: 2212–2217.
Relton JK et al. Peripheral administration of interleukin-1 receptor antagonist inhibits brain damage after focal cerebral ischemia in the rat. Exp Neurol 1996; 138: 206–213.
Chang ML et al. Nicotinamide and ketamine reduce infarct volume and DNA fragmentation in rats after brain ischemia and reperfusion. Neurosci Lett 2002; 322: 137–140.
Linnik MD et al. Expression of bcl-2 from a defective herpes simplex virus-1 vector limits neuronal death in focal cerebral ischemia. Stroke 1995; 26: 1670–1674; discussion 1675.
Yagi T et al. Rescue of ischemic brain injury by adenoviral gene transfer of glial cell line-derived neurotrophic factor after transient global ischemia in gerbils. Brain Res 2000; 885: 273–282.
Lawrence MS et al. Herpes simplex viral vectors expressing bcl-2 are neuroprotective when delivered after a stroke. J Cereb Blood Flow Metab 1997; 17: 740–744.
Padfield PJ, Elliott AC, Baldassare JJ . Adenovirus-mediated gene expression in isolated rat pancreatic acini and individual pancreatic acinar cells. Pflugers Arch 1998; 436: 782–787.
Tran QK et al. Midkine inhibits bradykinin-stimulated Ca(2+) signaling and nitric oxide production in endothelial cells. Biochem Biophys Res Commun 2000; 276: 830–836.
Takada T et al. Midkine, a retinoic acid-inducible heparin-binding cytokine in inflammatory responses: chemotactic activity to neutrophils and association with inflammatory synovitis. J Biochem (Tokyo) 1997; 122: 453–458.
Shibata Y et al. Nuclear targeting by the growth factor midkine. Mol Cell Biol 2002; 22: 6788–6796.
Davidson BL et al. A model system for in vivo gene transfer into the central nervous system using an adenoviral vector. Nat Genet 1993; 3: 219–223.
Ooboshi H et al. Augmented adenovirus-mediated gene transfer to atherosclerotic vessels. Arterioscler Thromb Vasc Biol 1997; 17: 1786–1792.
Ooboshi H et al. Adenovirus-mediated gene transfer to ischemic brain: ischemic flow threshold for transgene expression. Stroke 2001; 32: 1043–1047.
Yao H et al. Simplified model of krypton laser-induced thrombotic distal middle cerebral artery occlusion in spontaneously hypertensive rats. Stroke 1996; 27: 333–336.
Tsutsui J et al. A new family of heparin-binding factors: strong conservation of midkine (MK) sequences between the human and the mouse. Biochem Biophys Res Commun 1991; 176: 792–797.
Takei Y et al. 5′-,3′-inverted thymidine-modified antisense oligodeoxynucleotide targeting midkine – its design and application for cancer therapy. J Biol Chem 2002; 277: 23800–23806.
Acknowledgements
This work was supported in part by the research grant in aid from the Ministry of Health and Welfare Comprehensive Research on Aging and Health (H11-008), Japan and from the Ministry of Education, Science and Culture (14570604), Japan (HO). We would like to thank the University of Iowa Gene Transfer Vector Core, especially Maria Scheel, Kate Lamsey and Beverly L Davidson, for viral vector preparations.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Takada, J., Ooboshi, H., Ago, T. et al. Postischemic gene transfer of midkine, a neurotrophic factor, protects against focal brain ischemia. Gene Ther 12, 487–493 (2005). https://doi.org/10.1038/sj.gt.3302434
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/sj.gt.3302434
Keywords
This article is cited by
-
Loss of heparin-binding protein prevents necrotizing glomerulonephritis: first clues hint at plasminogen activator inhibitor-1
International Urology and Nephrology (2013)
-
IL4 gene delivery to the CNS recruits regulatory T cells and induces clinical recovery in mouse models of multiple sclerosis
Gene Therapy (2008)
-
Absence of an intrathecal immune reaction to a helper-dependent adenoviral vector delivered into the cerebrospinal fluid of non-human primates
Gene Therapy (2008)
-
Postischemic Gene Transfer of Soluble Flt-1 Protects against Brain Ischemia with Marked Attenuation of Blood—Brain Barrier Permeability
Journal of Cerebral Blood Flow & Metabolism (2007)
-
Human gene therapy and imaging in neurological diseases
European Journal of Nuclear Medicine and Molecular Imaging (2005)