Abstract
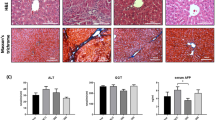
To examine the effect of the bcl-xs gene on the sequence from hepatic precancerous lesions, foci and neoplastic nodules, to hepatocellular carcinomas, Sprague–Dawley rats were given water containing 175 mg/l N-nitrosomorpholine (NNM) for 8 weeks. At weeks 1, 4 and 7, the left lobe of the rat liver was exposed and injected with the bcl-xs plasmid (pCR3.1-rat bcl-xs cDNA) or pCR3.1 encapsulated in cationic empty liposomes each at a dose of 80 μg plasmid/kg body weight. One minute later, low-field-strength, long-duration electric pulses were applied to the left lobe using a pincette electrode with circular poles 1 cm in diameter. The in vivo electroporation procedure significantly increased the transfer of the reporter gene chloramphenicol acetyl transferase (CAT) plasmid via empty liposomes. Thus, CAT mRNA was expressed not only at the sites of electrode contact but at sites 0.5–1.0 cm away from the electrode, and expression also increased with increasing doses of plasmid, meaning that in vivo electroporation enabled the expression of plasmid DNA throughout an extensive area of the rat liver. By week 11, the neoplastic nodules were significantly fewer and smaller in the bcl-xs group than in the pCR3.1 group at the two sites, one with and the other without electrode contact. No hepatocellular carcinomas were found in the rats that had received the bcl-xs plasmid, whereas these tumors were observed in 30% of the rats given pCR3.1. Moreover, overexpression of the bcl-xs protein was detected, and apoptotic activity was significantly increased in the neoplastic nodules, foci and hepatocytes adjacent to the hepatic lesions. These results indicate that the bcl-xs plasmid inhibits the occurrence and growth of rat hepatocellular carcinoma and may thus be effective for the prevention and treatment of human liver tumors.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout








Similar content being viewed by others
References
Chao DT, Korsmeyer SJ . Bcl-2 family: regulations of cell death Ann Rev Immunol 1998 16: 395–419
Yang E, Korsmeyer SJ . Molecular thanatopsis: a discourse on the bcl-2 family and cell death Blood 1996 88: 386–401
Desoize B . Anticancer drug resistance and inhibition of apoptosis Anticancer Res 1994 14: 2291–2294
Olopade OJ et al. Overexpression of bcl-x protein in primary breast cancer is associated with high tumor grade and nodal metastases Cancer J Sci Am 1997 3: 230–237
Paula A et al. Bcl-xL antisense treatment induced apoptosis in breast carcinoma cells Int J Cancer 2000 87: 582–590
Ziegler A et al. Induction of apoptosis in small-cell lung cancer cells by an antisense oligonucleotide targeting the bcl-2 coding sequence J Natl Cancer Inst 1997 89: 1027–1036
Baba M, Iishi H, Tatsuta M . In vivo electroporetic transfer of bcl-2 antisense oligonucleotide inhibits the development of hepatocellular carcinoma in rats Int J Cancer 2000 85: 260–266
Fiorentino M et al. High levels of bcl-2 messenger RNA detected by in situ hybridyzation in human hepatocellular carcinomas Dia Mol Pathol 1999 8: 189–194
Reed JC . Double identity for proteins of the bcl-2 family Nature 1997 387: 773–776
Boise LH et al. Bcl-x, a bcl-2-related gene that functions as a domant regulator of apoptotic cell death Cell 1993 74: 597–608
Fridman JS, Benedict MA, Maybaum J . Bcl-xs-induced cell death in 3T3 cells does not require or induce caspase activation Cancer Res 1999 59: 5999–6004
Fridman JS et al. Cytchrome c depletion upon expression of bcl-xs J Biol Chem 2001 276: 4205–4210
Clarke MF et al. A recombinant bcl-xs adenovirus selectively induces apoptosis in cancer cells but not in normal bone marrow cells Proc Natl Acad Sci USA 1995 92: 11024–11028
Sumantran VN et al. A bcl-xs adenovrus selectively induced apoptosis in transformed cells compared to normal mammary cells Neoplasia 2000 2: 251–260
Abou-Elella A, Gramlich T, Fritsch C, Gansler T . c-Myc amplification in hepatocellular carcinoma predicts unfavorable prognosis Mol Pathol 1996 9: 95–98
Pascale RM et al. c-Myc amplification in pre-malignant and malignant lesions induced in rat liver by the resistant hepatocyte model Int J Cancer 1996 68: 136–142
Macias Rodriguez MA et al. Risk factors for hepatocellular carcinoma in patients with liver cirrhosis Rev Esp Enferm Dig 2000 92: 458–469
Hanazaki K et al. Survival and recurrence after hepatic resection of 386 consecutive patients with hepatocellular carcinoma J Am Coll Surg 2000 191: 381–388
Moore MA, Mayer D, Bannasch P . The dose dependence and sequential appearance of putative preneoplastic populations induced in the rat liver by stop experiments with N-nitrosomorpholine Carcinogenesis 1982 3: 1429–1436
Dagle JM, Andracki ME, Devine RJ, Walder JA . Physical properties of oligonucleotides containing phosphoramidate modified internucleoside linkages Nucleic Acids Res 1991 19: 1805–1810
Kay MA, Liu D, Hoogerbrugge PM . Gene therapy Proc Natl Acad Sci USA 1991 94: 12744–12746
Jiao S et al. Direct gene transfer into nonhuman primate myofibers in vivo Hum Gene Ther 1992 3: 21–33
Ledley FD . Nonviral gene therapy: the promise of genes as pharmaceutical products Hum Gene Ther 1995 6: 1129–1144
Lee RJ, Huang L . Lipidic vector systems for gene transfer Crit Rev Ther Drug: Carr Syst 1997 14: 173–206
Niven R, Zhang Y, Smith J . Toward development of a non-viral gene therapeutic Adv Drug Del Rev 1997 26: 135–150
Rols M-P et al. In vivo electrically mediated protein and gene transfer in murine melanoma Nat Biotechnol 1998 16: 168–171
Suzuki T, Shin BC, Fujikura K, Matsuzaki T . Direct gene transfer into rat liver cells by in vivo electroporation FEBS Lett 1998 425: 436–440
Heller L et al. Electrically mediated plasmid DNA delivery to hepatocellular carcinomas in vivo Gene Therapy 2000 7: 826–829
Institute of Laboratory Animal Resources . Histologic typing of liver tumors of the rat J Natl Cancer Inst 1980 64: 177–207
Mir LM et al. High-efficiency gene transfer into skeletal muscle mediated by electric pulses Proc Natl Acad Sci USA 1999 96: 4262–4267
Baer A, Schubeler D, Bode J . Transcriptional properties of genomic transgene integration sites marked by electroporation or retroviral infection Biochemistry 2000 39: 7041–7049
Gavrieli Y, Sheman Y, Ben-Sasson SA . Identification of programmed cell death in situ via specific labeling of nuclear DNA fragmentation J Cell Biol 1992 119: 493–501
Acknowledgements
We thank Meiwa Shoji Company (Osaka, Japan) for lending a BTX 500 optimizer and a BTX 820 standard sequare wave electroporator (BTX, San Diego, CA, USA).
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Baba, M., Iishi, H. & Tatsuta, M. Transfer of bcl-xs plasmid is effective in preventing and inhibiting rat hepatocellular carcinoma induced by N-nitrosomorpholine. Gene Ther 8, 1149–1156 (2001). https://doi.org/10.1038/sj.gt.3301504
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/sj.gt.3301504
Keywords
This article is cited by
-
Bak but not Bax is essential for Bcl-xS-induced apoptosis
Cell Death & Differentiation (2005)
-
DNA electrotransfer: its principles and an updated review of its therapeutic applications
Gene Therapy (2004)
-
Prevention of liver cancer
Current Oncology Reports (2002)